The cancer biomarkers market to reach US$ 34,857.12 million by 2028, registering at a CAGR of 11.1% during 2022–2028, according to a new research study conducted by The Insight partners
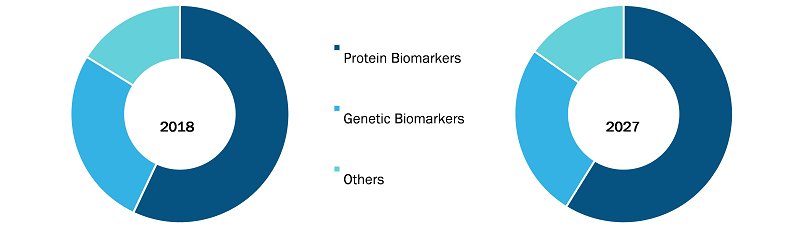
Protein Biomarker Segment to Hold Largest Market Share During 2019–2028
The report highlights the trends and drivers prevailing in the market.
Based on biomarker type, the cancer biomarkers market is segmented into genetic biomarkers, protein biomarkers, and others. The protein biomarker segment held the largest market share in 2022 and is anticipated to register the highest growth rate during forecast period. Based on profiling technology, the cancer biomarker market is segmented into omics technologies, imaging technologies, immunoassays, cytogenetics, and bioinformatics. In 2022, the omics technologies segment held the largest share of the market, and it is anticipated to register the highest growth rate during 2022–2028. By cancer type, the market is segmented into breast cancer, lung cancer, prostate cancer, leukemia, bladder cancer, and others. In 2022, the others segment held the largest share of the market. However, the market in the lung cancer segment is expected to grow at the fastest rate in the coming years. Based on application, the cancer biomarkers market is segmented into diagnostics, research and development, prognostics, and others. In 2022, the prognostics segment held the largest share of the market. In addition, the segment is expected to register the highest CAGR during the forecast period. Based on geography, the cancer biomarkers market is segmented into North America (the US, Canada, and Mexico), Europe (the UK, Germany, France, Italy, the Netherlands, and the Rest of Europe), Asia Pacific (China, Japan, India, Australia, Thailand, and the Rest of Asia Pacific), the Middle East & Africa (the UAE, Saudi Arabia, South Africa, and the Rest of Middle East & Africa), and South & Central America (Brazil, Argentina, and the Rest of South & Central America).
Cancer Biomarkers Market, by Biomarker Type – 2022 and 2028
Cancer Biomarkers Market Forecast, Trends, Scope by 2031
Download Free Sample
Cancer Biomarkers Market Size and Forecasts (2021 - 2031), Global and Regional Share, Trends, and Growth Opportunity Analysis Report Coverage: By Biomarker Type (Protein Biomarkers, Genetic Biomarkers, and Others), Profiling Technology (Omics Technologies, Imaging Technologies, Immunoassays, Cytogenetics, and Bioinformatics), Cancer Type (Breast Cancer, Lung Cancer, Prostate Cancer, Leukemia, Bladder Cancer, and Others), and Application (Prognostics, Diagnostics, Research and Development, and Others), and Geography (North America, Europe, Asia Pacific, and South and Central America)
Cancer Biomarkers Market Forecast, Trends, Scope by 2031
Download Free SampleCancer Biomarkers Market Size and Forecasts (2021 - 2031), Global and Regional Share, Trends, and Growth Opportunity Analysis Report Coverage: By Biomarker Type (Protein Biomarkers, Genetic Biomarkers, and Others), Profiling Technology (Omics Technologies, Imaging Technologies, Immunoassays, Cytogenetics, and Bioinformatics), Cancer Type (Breast Cancer, Lung Cancer, Prostate Cancer, Leukemia, Bladder Cancer, and Others), and Application (Prognostics, Diagnostics, Research and Development, and Others), and Geography (North America, Europe, Asia Pacific, and South and Central America)
Cancer Biomarker Insights-Future Trends
Inclination Toward Personalized Medicine to Become Emerging Trend in Cancer Biomarkers Market During Forecast Period
The demand for personalized medicine is expanding in the healthcare sector, and it is a relatively new approach. Personalized medicine enables healthcare professionals to select treatment based on a patient's genetic profile. The use of personalized medicine helps minimize side effects and leads to successful results within a short period of time.
The use of this medicine comprises the study of individual’s genetic that makes up for tumor growth; the study helps oncologist to customize the medication depending on the genetic mutations of patients. Mutations in KRAS, ALK, and EGFR lead to lung cancer, but lung cancer patient provides essential information for the kind of treatment for identifying the type of mutation.
All patients receive the similar treatment for each type of cancer before the launch of personalized medicine. The patient's tissue removed to carry out the genetic diagnosis process and sample is checked for gene mutations. Further, depending on the type of mutation, chemotherapy is carried out.
Hence, personalized medicine allows the right treatment at the right time, which reduces the mortality rate. Therefore, based on these advantages of tailoring the treatment on an individual's genetic profile, the personalized medicine approach is likely to become future trend for the cancer biomarkers market.
Thermo Fisher Scientific Inc; F. Hoffmann-La Roche Ltd; Illumina Inc; Bio-Rad Laboratories Inc; QIAGEN; bioMérieux, Inc; Creative Diagnostics Hologic Inc; Myriad Genetic Inc; and Quest Diagnostics are among the several players engaged in addressing the growing demands for cancer biomarkers.
Contact Us
Phone: +1-646-491-9876
Email Id: sales@theinsightpartners.com