Product Segment to Bolster Human Microbiome Market Growth During 2023–2031
According to our new research study on "Human Microbiome Market Forecast to 2031 – Global Analysis – by Type, Disease Type, and Application," the market is expected grow US$ 4.84 billion by 2031 from US$ 1.07 billion in 2023; the market is estimated to register a CAGR of 20.8% during 2023–2031. The human microbiome market report emphasizes the trends prevalent in the global market, along with drivers and deterrents affecting its growth.
Significant investments by key market players and government initiatives are contributing to the human microbiome market growth. However, challenges during commercialization hamper the market growth. The emergence of precision medicine is expected to bring new human microbiome market trends in the coming years.
Human Microbiome Market Share, by Region, 2023 (%)
Human Microbiome Market Size and Forecast (2021 - 2031), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Type (Product and Software and Services), Application (Therapeutics, Diagnostics, and Research), Disease Type (Obesity, Diabetes, Autoimmune Disorder, Cancer, GIT Disorders, and Others), and Geography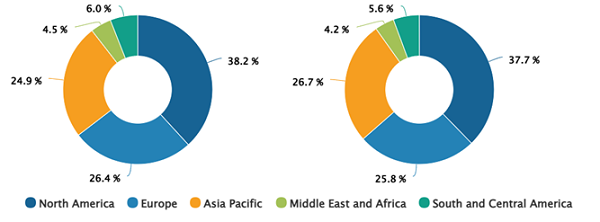
Human Microbiome Market Key Findings and Share by 2031
Download Free Sample
Significant Investments by Key Market Players Bolster Human Microbiome Market
The human microbiome industry's growth, innovation, and market dynamics are greatly influenced by the investments made by major players in the market. Major players are engaged in making significant investments in research and development to create novel probiotics, diagnostics, and treatments based on the microbiome. These expenditures stimulate innovation, identifying new strains, modes of action, and medicinal uses. Clinical trials receive a large amount of funding, which is essential for demonstrating the effectiveness and safety of products based on the microbiome. The market can grow if new products are approved due to successful trials. For example, a pioneer in microbiome mapping technology, Kanvas Biosciences, announced a US$ 12 million Pre-Series A funding round in June 2023. The company's proprietary platform technology, which has the potential to transform drug development for illnesses linked to the microbiome, will be further developed with the help of the investment. Additionally, regulating frameworks tailored to human microbiome-based products may be established or improved by governments, which would shorten the time and expense involved in introducing novel treatments to the market. Well-defined regulations guarantee the safety and efficacy of products, thereby increasing consumer trust and market uptake.
The human microbiome market analysis has been carried out by considering the following segments: type, application, disease type, and geography. The human microbiome market, based on type, is segmented into product and software and services. The product segment held a larger share of the human microbiome market in 2023, and it is expected to register a higher CAGR during 2023–2031. The human microbiome includes all microorganisms in and on the human body. These microbes have potential for use in disease management. Microbiome products show promise in treating severe conditions and personalizing therapy. The microbiota has been linked to neurological diseases, inflammatory diseases, and cancer.
Based on application, the market is segmented into therapeutics, diagnostics, and research. The therapeutics segment accounted for the largest share of the human microbiome market in 2023. The significant metabolic potential of gut microbes and their role in maintaining human health is emerging, with new possibilities to utilize them as therapeutic agents for addressing human disorders. In recent years, microbiome-based therapeutics, also known as microbiome modulators, have made rapid advances. Several promising candidates have moved forward through the clinical pipeline. For instance, in April 2023, Ferring Pharmaceuticals' VOWST, the first fecal microbiota product taken orally, was approved for preventing recurrence of Clostridioides difficile (C. difficile) infection in patients 18 years and older, following antibiotic treatment for recurrent infection.
Based on disease type, the human microbiome market is segmented into obesity, diabetes, autoimmune disorders, cancer, GIT disorders, and others. The GIT disorders segment held the largest share of the human microbiome market in 2023.
The geographic scope of the human microbiome market report includes the assessment of the market performance in North America, Europe, Asia Pacific, the Middle East and Africa, and South and Central America. North America accounted for the largest human microbiome market share in 2023. The market growth in North America is characterized by robust research activities on the human microbiome, increased funding for biotechnology, a rise in health consciousness among the population, and an increase in the demand for probiotics and prebiotics to treat chronic diseases. Moreover, an increase in focus on the development of microbiome therapeutics, the rise in the number of conferences in this region, and the presence of local key players are likely to stimulate growth and contribute to exceptional revenue generation for the human microbiome market in North America.
In the US, product launches by various market players and product approvals by regulatory authorities are increasing the demand for human microbiome therapeutics. For instance, in November 2022, Ferring Pharmaceuticals obtained US Food and Drug Administration (FDA) approval for REBYOTA (fecal microbiota, live-jslm). This is a novel first-in-class microbiota-based live biotherapeutic indicated to prevent the recurrence of Clostridioides difficile infection in individuals 18 years of age and older, following antibiotic treatment for recurrent C. difficile infection.
Contact Us
Phone: +1-646-491-9876
Email Id: sales@theinsightpartners.com