Device Segment Drives Impulse Oscillometry Market Growth
According to our new research study named "Impulse Oscillometry Market Forecast to 2034 – Global Analysis – by Product, Application, End User, and Geography," the market was valued at US$ 95.85 million in 2025 and is projected to reach US$ 156.19 million by 2034; it is expected to register a CAGR of 5.7% during 2026–2034. The rising prevalence of respiratory diseases, technological advancements in diagnostic devices, and growing awareness and adoption in pediatrics drive the adoption of impulse oscillometry. Increased focus on small airway dysfunction (SAD) detection and integration of AI and machine learning for enhanced analysis are projected to drive impulse oscillometry market trends in the coming years.
Impulse Oscillometry (IOS) is a noninvasive pulmonary function test measuring airway resistance and reactance during normal breathing using sound waves. It helps diagnose and monitor asthma, COPD, and other respiratory disorders. Rising prevalence of respiratory diseases such as asthma and COPD, increasing air pollution, and growing geriatric population are driving impulse oscillometry market growth. Additionally, demand for noninvasive diagnostic techniques and technological advancements in pulmonary devices is driving growth in the impulse oscillometry market size. Expanding healthcare infrastructure and heightened awareness of early lung function assessment and preventive care are contributing to the rising impulse oscillometry market size.
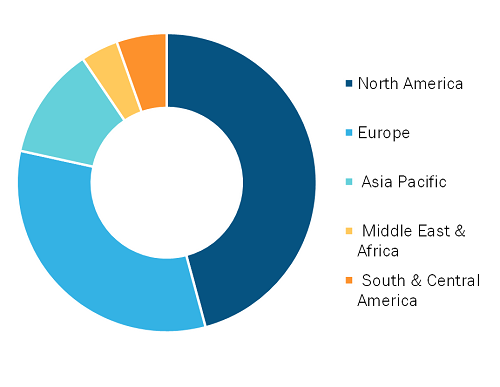
Impulse Oscillometry Market, by Region, 2025 (%)
Impulse Oscillometry Market Share, Size & Demand by 2034
Download Free Sample
Impulse Oscillometry Market Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Product (Device and Accessories), Application (Asthma, COPD, and Other Applications), End User (Hospitals and Clinics, Diagnostic Laboratories, and Other End Users), and Geography (North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America)
Impulse Oscillometry Market Share, Size & Demand by 2034
Download Free SampleImpulse Oscillometry Market Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Product (Device and Accessories), Application (Asthma, COPD, and Other Applications), End User (Hospitals and Clinics, Diagnostic Laboratories, and Other End Users), and Geography (North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America)
Source: The Insight Partners Analysis
Impulse Oscillometry Market Analysis Based on Segmental Evaluation:
Based on product, the impulse oscillometry market is bifurcated into devices and accessories. The device segment held a larger impulse oscillometry market share in 2025. The segment is growing due to increasing respiratory disorders such as asthma and chronic obstructive pulmonary disease (COPD). Rising demand for noninvasive, patient-friendly diagnostic tools, technological advancements in pulmonary function testing equipment, expanding geriatric populations, and greater adoption in hospitals and pulmonary clinics drive device sales.
The geographical scope of the impulse oscillometry market report encompasses an assessment of the market performance in North America, Europe, Asia Pacific, South America, Central America, the Middle East, and Africa. North America dominated the impulse oscillometry market share in 2025.
The increasing need for advanced pulmonary diagnostic tools originates from rising air pollution levels, smoking rates, and occupational exposure to respiratory irritants. The aging population in the US and Canada creates a higher demand for noninvasive diagnostic technologies, which include impulse oscillometry, because they are more susceptible to chronic respiratory diseases. The development of pulmonary function testing equipment reached new heights because technological innovations brought better testing equipment, which combines higher accuracy with easier portable devices and simpler testing systems, boosting impulse oscillometry market growth.
Growing awareness among healthcare providers about the benefits of IOS—such as minimal patient effort requirement and suitability for pediatric and geriatric populations—supports market expansion. The North American healthcare system combines favorable reimbursement policies with advanced medical infrastructure and rising healthcare spending to accelerate the adoption of new diagnostic technologies. The region's medical device manufacturers conduct clinical research and early disease detection programs, which together drive impulse oscillometry market growth through product innovation and market introduction.
A 2024 review in BMJ Open Respiratory Research demonstrated how IOS testing results relate to standard pulmonary function assessments used in asthma and COPD diagnosis, while showing its value for assessing small airway damage through its artificial intelligence diagnostic capabilities. A 2025 study in BMC Pulmonary Medicine demonstrated that IOS testing helped detect small airway dysfunction among patients with preserved ratio impaired spirometry and COPD through its ability to identify R5-R20 parameters, which functioned as effective disease diagnostic cutoffs. A 2025 study in Diagnostics confirmed that IOS testing showed strong connections with body plethysmography results within obstructive groups, which confirmed its ability to function as a complementary diagnostic tool for medical practice.
A 2025 study in Clinical and Translational Allergy examined IOS's role in COPD and asthma-COPD overlap through its use in tracking acute exacerbations with R5-R20 measurements across mouse models and human study groups. A 2025 study in Respiratory Medicine showed that IOS testing results and airway oscillometry measurements produced matching resistance ratio results among asthma and COPD patients, which demonstrated the need for device-specific assessment standards in testing small airway dysfunction. A 2025 CHEST review showed that IOS testing functions as a diagnostic tool for small airway dysfunction in asthma patients who have treatable traits, with support from the Mexican National Institute of Respiratory Diseases. The new technologies enable the population base to use IOS testing for better identification of respiratory diseases and their associated health risks. Hence, the above-mentioned factors collectively support the growth of the global impulse oscillometry market.
Chest MI Inc, THORASYS Thoracic Medical Systems Inc., Jaeger Medical GmbH, GANSHORN Medizin Electronic GmbH, Morgan Scientific Inc, Lothar Medical GmbH, Cognita Labs LLC, COSMED Srl, RESTECH srl, and Vitalograph Ltd. are among the leading companies profiled in the impulse oscillometry market report.
Based on product, the impulse oscillometry market is bifurcated into device and accessories. By application, the market is classified into asthma, COPD, and other applications. Based on end user, the market is segmented into hospitals and clinics, diagnostic laboratories, and other end users. Geographically, the impulse oscillometry market is segmented into North America (the US, Canada, and Mexico), Europe (France, Germany, the UK, Spain, Italy, and the Rest of Europe), Asia Pacific (China, Japan, India, South Korea, Australia, and the Rest of Asia Pacific), the Middle East and Africa (Saudi Arabia, South Africa, the UAE, and the Rest of Middle East and Africa), and South and Central America (Brazil, Argentina, and the Rest of South and Central America).
Contact Us
Phone: +1-646-491-9876
Email Id: sales@theinsightpartners.com