Fingertip Pulse Oximeters Segment to Contribute Larger Share to Market during 2020–2027
According to the latest study on “Pulse Oximeters Market Forecast to 2027 –COVID-19 Impact and Global Analysis – by Type, Sensor Type, and End User,” the market was valued at US$ 1,892.00 million in 2019 and is projected to reach US$ 3,693.92 million by 2027; it is expected to grow at a CAGR of 8.6% during 2020–2027. The report highlights trends prevailing in the global pulse oximeters market, and drivers and restraints pertaining to the market growth.
The growth of the pulse oximeters market is attributed to the increasing prevalence of respiratory diseases and growing preference for remote patient monitoring. Further, the strategic developments by manufacturers offer lucrative opportunities to the global pulse oximeters market players. However, the inaccuracies in pulse oximeter readings due to several factors hinder the market growth.
On the basis of type, the pulse oximeters market is sub-segmented into fingertip pulse oximeters, handheld pulse oximeters, wrist-worn pulse oximeters, and pediatric pulse oximeters. In 2019, the fingertip pulse oximeters segment accounted for the largest share of the market. The growth of the market for this segment is attributed to the rising number of product launches and increasing innovations in pulse oximeters devices.
Nonin; Koninklijke Philips N.V.; Vyaire Medical, INC; Smiths Medical; Hill Rom Holding Inc.; Edwards Lifesciences Corporation; Medtronic; Masimo; Beurer GmbH; and Maxtec are among the key players operating in the pulse oximeters market. These companies focus on partnerships, product launches, and collaborations, among other business strategies, to sustain their market positions. For instance, in July 2020, Nonin Medical has received 510(k) clearance for its flagship product — the CO-Pilot wireless handheld multi-parameter system. The product is meant for EMS, firefighters and military personnel to measure key vital signs following a cardiac arrest, traumatic injury, carbon monoxide or smoke inhalation.
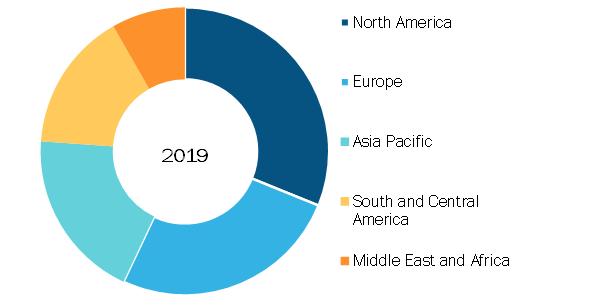
Pulse Oximeters Market, by Region, 2019 (%)
Pulse Oximeters Market Size, Share, Scope Analysis Report 2027
Download Free Sample
Pulse Oximeters Market Forecast to 2027 - Analysis By Type (Fingertip Pulse Oximeters, Handheld Pulse Oximeters, Wrist-worn Pulse Oximeters, Pediatric Pulse Oximeters); End User (Hospitals, Clinics, Others) and Geography
Pulse Oximeters Market Size, Share, Scope Analysis Report 2027
Download Free SamplePulse Oximeters Market Forecast to 2027 - Analysis By Type (Fingertip Pulse Oximeters, Handheld Pulse Oximeters, Wrist-worn Pulse Oximeters, Pediatric Pulse Oximeters); End User (Hospitals, Clinics, Others) and Geography
The report segments the global pulse oximeters market as follows:
By Type
- Fingertip Pulse Oximeters
- Handheld Pulse Oximeters
- Wrist-Worn Pulse Oximeters
- Pediatric Pulse Oximeters
By Sensor Type
- Reusable
- Disposable
By End User
- Hospitals
- Clinics
- Others
By Geography
- North America
- US
- Canada
- Mexico
- Europe
- France
- Germany
- Italy
- UK
- Russia
- Asia Pacific
- China
- India
- South Korea
- Japan
- Australia
- Middle East and Africa
- South Africa
- Saudi Arabia
- UAE
- South and Central America
- Brazil
- Argentina
Phone: +1-646-491-9876
Email Id: sales@theinsightpartners.com