Mechanical Ventilation Segment Held Largest Share of Acute Lung Injury Market
According to our new research study on "Acute Lung Injury Market Forecast to 2028 – COVID-19 Impact and Analysis – by Therapy and End User," the market is expected to reach US$ 741.98 million by 2028 from US$ 539.61 million in 2021; it is estimated to register a CAGR of 4.7% from 2021 to 2028. The report highlights trends prevailing in the market and drivers and hindrances pertaining to the market growth. Factors such as increasing prevalence of respiratory diseases and rising geriatric population are the key factors driving the market growth. However, the high cost of therapies hinders market growth.
Based on therapy, the acute lung injury market is segmented into mechanical ventilation, fluid management, pharmacotherapy, and adjunctive procedures. In 2021, the mechanical ventilation segment accounted for the largest market share in the global acute lung injury market. The segment's growth is attributed to the fact that mechanical ventilation in acute lung injury (ALI) aims to maintain oxygen while avoiding its toxicity and complications associated with the ventilation.
GlaxoSmithKline plc.; Stemedica Cell Technologies, Inc; Histocell; APEPTICO Forschung und Entwicklung GmbH; Windtree Therapeutics, Inc.; ReAlta Life Sciences, Inc.; Apeiron Biologics AG; Qx Therapeutics, Inc.; Angion; and Asklepion Pharmaceuticals, LLC are among the leading companies operating in the acute lung injury market. These companies are adopting inorganic and organic growth strategies to expand their global footprints and product portfolio to meet the rising demands for acute lung injury treatment. For instance, in March 2021, Qx Therapeutics Inc. announced that the US Food and Drug Administration (FDA) cleared investigational new drug application (IND) to investigate its lead drug product, QXT-101, for a Phase 2 clinical trial for its safety and efficacy in hospitalized patients with severe or critical COVID-19. QXT-101 is a first-in-class treatment for patients who suffer from acute lung injury (ALI). Thus, continuous efforts and investments by the market players to address the rising demands by boosting the production and sales of their products in the domestic and international markets are driving the acute lung injury market.
.
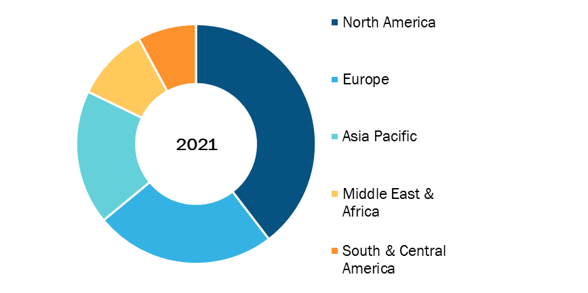
Acute Lung Injury Market, by Region, 2021(%)
Acute Lung Injury Market Size, Share & Demand 2034
Download Free Sample
Acute Lung Injury Market Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Therapy (Mechanical Ventilation, Fluid Management, Pharmacotherapy, and Adjunctive Procedures) and End User (Hospitals, Ambulatory Surgery Centers, and Others) and Geography
Based on therapy, the acute lung injury market is segmented into mechanical ventilation, fluid management, pharmacotherapy, and adjunctive procedures. In terms of end user, the acute lung injury market is segmented into hospitals, ambulatory surgery centers, and others. Geographically, the acute lung injury market is segmented into North America (the US, Canada, and Mexico), Europe (France, Germany, the UK, Spain, Italy, and the Rest of Europe), Asia Pacific (China, India, Japan, Australia, South Korea, and the Rest of APAC), Middle East & Africa (Saudi Arabia, the UAE, South Africa, and the Rest of MEA), and South and Central America (Brazil, Argentina, and the Rest of SCAM). Acute Lung Injury Market Size, Share & Demand 2034
Download Free SampleAcute Lung Injury Market Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Therapy (Mechanical Ventilation, Fluid Management, Pharmacotherapy, and Adjunctive Procedures) and End User (Hospitals, Ambulatory Surgery Centers, and Others) and Geography
Contact Us
Phone: +1-646-491-9876
Email Id: sales@theinsightpartners.com