Human Papillomavirus (HPV) Vaccine Market for 3 Dose Segment to Grow at Higher CAGR During 2022–2028According to our new market research study on “Human Papillomavirus (HPV) Vaccine Market Forecast to 2028 – COVID-19 Impact and Global Analysis – by Type, Dosage, Age, Application, and Distribution Channel,” the market is expected to grow from US$ 4,273.84 million in 2022 to US$ 5,730.50 million by 2028; it is estimated to grow at a CAGR of 5.0% from 2022 to 2028. The report highlights trends prevailing in the market and drivers and hindrances pertaining to the market growth.
Based on dosage, the human papillomavirus (HPV) vaccine market is segmented into 2 dose and 3 dose. In 2022, the 2 dose segment accounts for a larger market share. Pre-adolescent girls (9–15 years) can receive a 2 dose HPV vaccine series at either a 6 month or 1-year interval to protect from HPV 16, the most prevalent type associated with cervical cancers, and several other less prevalent types. This series of vaccinations is highly expected to protect her from HPV infection until she enters the routine screening program, whether primary HPV testing or a combination of HPV testing and cytology. The 2-dose program has been recommended by the World Health Organization (WHO) since 2015. 2 doses are generally administered to boys and girls aged between 9 to 15 years, and the doses are administered within a span of 12 months. The minimum interval is 5 months between the first and second dose. If the vaccination schedule is interrupted, vaccine doses do not need to be repeated (no maximum interval). Immunogenicity studies have shown that 2 doses of HPV vaccine given to 9–14 years-olds at least 6 months apart provided as good or better protection than 3 doses given to older adolescents or young adults.
3 dose vaccine offers excellent immunogenicity that induces high peak titers of antibodies, which persist for years. Therefore, owing to the advantages of the 3 doses, the market for this segment is likely to grow at a significant rate during the forecast period.
Based on type, the human papillomavirus (HPV) vaccine market is segmented into 9-valent HPV vaccine, quadrivalent HPV vaccine, and bivalent HPV vaccine. In 2022, the quadrivalent HPV vaccine segment holds the largest share of the market.
The global human papillomavirus (HPV) vaccine market, based on age, is segmented into 9 to 14 years and 15 to 45 years. The 9 to 14 years segment holds a larger market share in 2022 and is anticipated to register a higher CAGR during the forecast period.
In terms of application, the global human papillomavirus (HPV) vaccine market is segmented into HPV-attributable cancers and genital warts. The HPV-attributable cancers segment holds a larger share of the market in 2022, and the same segment is anticipated to register a higher CAGR during the forecast period.
On the basis of distribution channel, the global human papillomavirus (HPV) vaccine market is segmented into doctors office, community health clinics, school-based health centers, health departments, hospitals, and others. The hospitals segment holds the largest market share in 2022, and the school-based health centers segment is anticipated to register the highest CAGR in the market during the forecast period.
The cost of overall healthcare is surging significantly in North America. The US health system incurred a direct cost of US$ 5.3 billion in 2020. The healthcare system contributed US$ 60 billion in addition to the overall annual costs of the country in 2020. Over 40% of the population in North America canceled their appointments in 2020, and 13% of them reported that they needed care but did not schedule or receive care. The COVID-19 pandemic altered economic conditions and social behaviors in North American countries. Containment measures enacted by governments to mitigate the disease spread changed the pattern of healthcare service delivery in the US. According to the Department of Emergency Medicine, in many cities across the country, emergency department (ED) visits decreased by ~40% in 2020. Moreover, outpatient appointments and elective treatments were postponed or replaced by telemedicine practices.
In November 2021, Pfizer Inc. announced the investigational COVID-19 oral antiviral candidate, PAXLOVID. Based on an interim analysis of the Phase 2/3 Evaluation of Protease Inhibition for COVID-19 in High-Risk Patients (EPIC-HR), a randomized, double-blind study of non-hospitalized adult COVID-19 patients who were at a high risk of progressing to severe illness, the PAXLOVID vaccine reportedly reduced hospitalization and death. There were supply constraints related to vaccine production in the initial months of 2021. However, setting up new plants and promoting technological shifts are the strategies that support the production of booster doses in the US and Canada, along with their administration plans.
The COVID-19 pandemic is likely to have longer-term consequences on the HPV vaccine business. Many primary care clinics in the US have continued to operate at a reduced capacity to abide by social distancing measures and adopt stringent cleaning protocols. A reduction in in-person office visits limits opportunities for providers of vaccine to discuss and deliver vaccines. Barriers in transportation are likely to harm HPV vaccination; pharma and biotech companies traditionally find it difficult to market vaccines among the masses because of resistance received from parents and the absence of widespread awareness about school entry requirements that support other adolescent vaccines. The available evidence indicates that the COVID-19 pandemic has dramatically disrupted the delivery of HPV vaccines in 2020 and 2021. Early reports by CDC, suggest that ordering and billing dropped by >70% in March 2020. Furthermore, a 25–50% decrease in HPV vaccine orders continued in June 2020, in contrast to other early childhood-vaccine orders that resurged to near pre-pandemic levels. Although the revival of the HPV vaccine market is in progress, available data suggest that a short-term decline in HPV vaccination coverage will be substantial, and the demand for HPV vaccines will grow in North America for long term.
GSK; Innovax; Walvax; Serum Institute of India Pvt., Ltd.; Vaccitech; Innovio Pharmaceuticals; Merck & Co., Inc.; 2A Pharm; ChengDu Institute of Biological Products Co., Ltd.; Sanofi; R-Pharm; Shanghai Bowei; and BioLeader are among the leading companies operating in the global human papillomavirus (HPV) vaccine market.
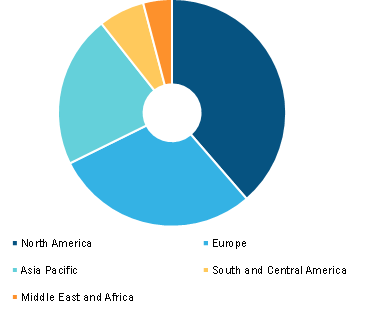
Global Human Papillomavirus (HPV) Vaccine Market, by Region, 2022 (%)
Human Papillomavirus (HPV) Vaccine Market Size & Demand 2034
Download Free Sample
Human Papillomavirus (HPV) Vaccine Market Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Type (9-Valent HPV Vaccine, Quadrivalent HPV Vaccine, Bivalent HPV Vaccine); Dosage (2 Dose, 3 Dose); Age (9 to 14 Years, 15 to 45 Years); Application (HPV-Attributable Cancer, Genital Warts); and Geography
By geography, the human papillomavirus (HPV) vaccine market is segmented into North America (the US, Canada, and Mexico), Europe (the UK, Germany, France, Italy, Spain, Russia, Belarus, and the Rest of Europe), Asia Pacific (China, Japan, India, Australia, Southeast Asia, New Zealand, South Korea, and the Rest of Asia Pacific), the Middle East & Africa (the UAE, Saudi Arabia, South Africa, Iran, Turkey, Kuwait, Bahrain, Egypt, and the Rest of Middle East & Africa), and South & Central America (Brazil, Argentina, Chile, Guatemala, Peru, Colombia, and the Rest of South & Central America).
Contact Us
Phone: +1-646-491-9876
Email Id: sales@theinsightpartners.com
The global human papillomavirus (HPV) vaccine market, based on age, is segmented into 9 to 14 years and 15 to 45 years. The 9 to 14 years segment holds a larger market share in 2022 and is anticipated to register a higher CAGR during the forecast period.
The COVID-19 pandemic is likely to have longer-term consequences on the HPV vaccine business. Many primary care clinics in the US have continued to operate at a reduced capacity to abide by social distancing measures and adopt stringent cleaning protocols. A reduction in in-person office visits limits opportunities for providers of vaccine to discuss and deliver vaccines. Barriers in transportation are likely to harm HPV vaccination; pharma and biotech companies traditionally find it difficult to market vaccines among the masses because of resistance received from parents and the absence of widespread awareness about school entry requirements that support other adolescent vaccines. The available evidence indicates that the COVID-19 pandemic has dramatically disrupted the delivery of HPV vaccines in 2020 and 2021. Early reports by CDC, suggest that ordering and billing dropped by >70% in March 2020. Furthermore, a 25–50% decrease in HPV vaccine orders continued in June 2020, in contrast to other early childhood-vaccine orders that resurged to near pre-pandemic levels. Although the revival of the HPV vaccine market is in progress, available data suggest that a short-term decline in HPV vaccination coverage will be substantial, and the demand for HPV vaccines will grow in North America for long term.
Human Papillomavirus (HPV) Vaccine Market Size & Demand 2034
Download Free SampleHuman Papillomavirus (HPV) Vaccine Market Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Type (9-Valent HPV Vaccine, Quadrivalent HPV Vaccine, Bivalent HPV Vaccine); Dosage (2 Dose, 3 Dose); Age (9 to 14 Years, 15 to 45 Years); Application (HPV-Attributable Cancer, Genital Warts); and Geography
Phone: +1-646-491-9876
Email Id: sales@theinsightpartners.com