Cardiovascular Medical Devices Segment to Register Highest CAGR During 2021–2028
According to our latest study on “Reprocessed Medical Devices Market Forecast to 2028 – COVID-19 Impact and Global Analysis – by Product and End-User,” and in the global market is expected to grow from US$ 2,087.36 million in 2021 to US$ 5,653.87 million by 2028; it is estimated to grow at a CAGR of 15.3% from 2021 to 2028. The report highlights the key factors driving the market growth and prominent players with their developments in the market.
Based on product type, the reprocessed medical devices market is segmented into cardiovascular medical devices, gastroenterology biopsy forceps, orthopedic external fixation devices, laparoscopic medical devices, general medical surgery, non-invasive devices, and others. The cardiovascular medical devices segment is further sub-segmented into blood pressure cuffs/tourniquet cuffs, cardiac stabilization and positioning devices, diagnostic electrophysiology catheters, and electrophysiology cables. The laparoscopic medical devices segment is bifurcated into endoscopic trocars and components and harmonic scalpels. The general medical surgery is bifurcated into balloon inflation devices and infusion pressure bags. The non-invasive devices segment is sub segmented into DVT garments, ECG leads, pulse oximeter probes, air-assisted transfer mats, ultrasonic clothing, extraction balloons and baskets, circuit accessories, optical lens, and other non-invasive devices. In 2021, cardiovascular medical devices held the largest market due to increasing cardiovascular devices in the region. Moreover, the same segment is expected to register faster growth owing to low-cost resulting in high adoption of the devices in the region.
By end-user, the reprocessed medical devices market is segmented into hospitals and clinics, ambulatory surgery centers, medical laboratories and others. The hospitals and clinics segment held a considerable share of the market in 2021, and it is likely to continue its dominance in the market during the forecast period.
The reprocessed medical devices market has experienced significant growth opportunities during the pandemic. The rapid spread of the COVID-19 has provided significant momentum to the reprocessed medical device industry in 2020. Following a severe shortage of medical supplies, regulatory agencies such as the Association of Medical Device Reprocessors (AMDR) promoted the reprocessing of healthcare equipment. According to AMDR, the expansion of reprocessing programmes could assist healthcare providers manage COVID-19-related costs, better regulate supply chains, and better plan for future hazards. While the COVID-19 pandemic continues to disrupt economies around the world, the growing volume of medical waste is a huge worry. This emphasizes the need of efficient reprocessing, which can not only help meet supply shortages but also enhance healthcare sustainability. However, with the onset of COVID-19 pandemic, physicians and patients became more concerned about their safety, hence, raising apprehensions about the reuse of medical devices. This negatively affected demand for reprocessed medical devices. Thus, to overcome these safety concerns, reassuring steps such as sterilization and microbial testing of reprocessed medical devices were made mandatory by various regulatory authorities in the region. Also, the risk of infection in surgeries and inappropriate sterilization of the medical devices can increase the potential outbreaks which hinders the market growth. In addition, the R&D of reprocessing services has put on hold due to diversion of professionals to satisfy the demand of pandemic related products. Thus, owing to the above–mentioned factors, the pandemic has shown negative impact on the reprocessed medical devices market.
Reprocessed Medical Device Market: Competition Landscape and Key Developments
Medline Industries, LP, Arjo Medical Devices, Currie Medical Specialties, Inc., Stryker Corporation, Teleflex Incorporated, Johnson and Johnson Services, Inc., 3M, STERIS, Cardinal Health, and Vanguard AG are among the leading companies operating in the reprocessed medical device market.
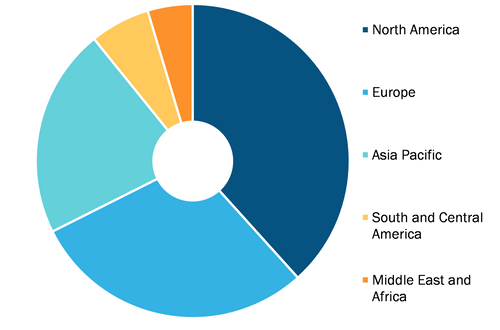
Reprocessed Medical Device Market, by region, 2021 (%)
Reprocessed Medical Devices Market Forecast and Size by 2028
Download Free Sample
Reprocessed Medical Devices Market Forecast to 2028 - Analysis By Product (Cardiovascular Medical Devices, General Surgery Medical Devices, Laparoscopic Medical Devices, Orthopedic External Fixation Devices, Gastroenterology Biopsy Forceps, Non-Invasive Devices, And Others); and End User (Hospitals & Clinics, Ambulatory Surgery Centers, Medical Laboratories, And Others)
Reprocessed Medical Devices Market Forecast and Size by 2028
Download Free SampleReprocessed Medical Devices Market Forecast to 2028 - Analysis By Product (Cardiovascular Medical Devices, General Surgery Medical Devices, Laparoscopic Medical Devices, Orthopedic External Fixation Devices, Gastroenterology Biopsy Forceps, Non-Invasive Devices, And Others); and End User (Hospitals & Clinics, Ambulatory Surgery Centers, Medical Laboratories, And Others)
The reprocessed medical device market have been geographically segmented as follows:
Based on geography, the market is segmented into North America, Europe, Asia Pacific (APAC), Middle East & Africa (MEA), and South & Central America (SCAM). The reprocessed medical device market in North America is sub-segmented into the US, Canada, and Mexico. The market in Europe is further segmented into France, Germany, the UK, Spain, Italy, and the Rest of Europe. The reprocessed medical device market in Asia Pacific (APAC) is further segmented into China, India, Japan, Australia, South Korea, and the Rest of Asia Pacific. The market in the Middle East & Africa (MEA) is segmented Into Saudi Arabia, the UAE, South Africa, and the Rest of Middle East & Africa. The reprocessed medical device market in South and Central America (SCAM) is sub-segmented into Brazil, Argentina, and the Rest of South and Central America
Contact Us
Phone: +1-646-491-9876
Email Id: sales@theinsightpartners.com