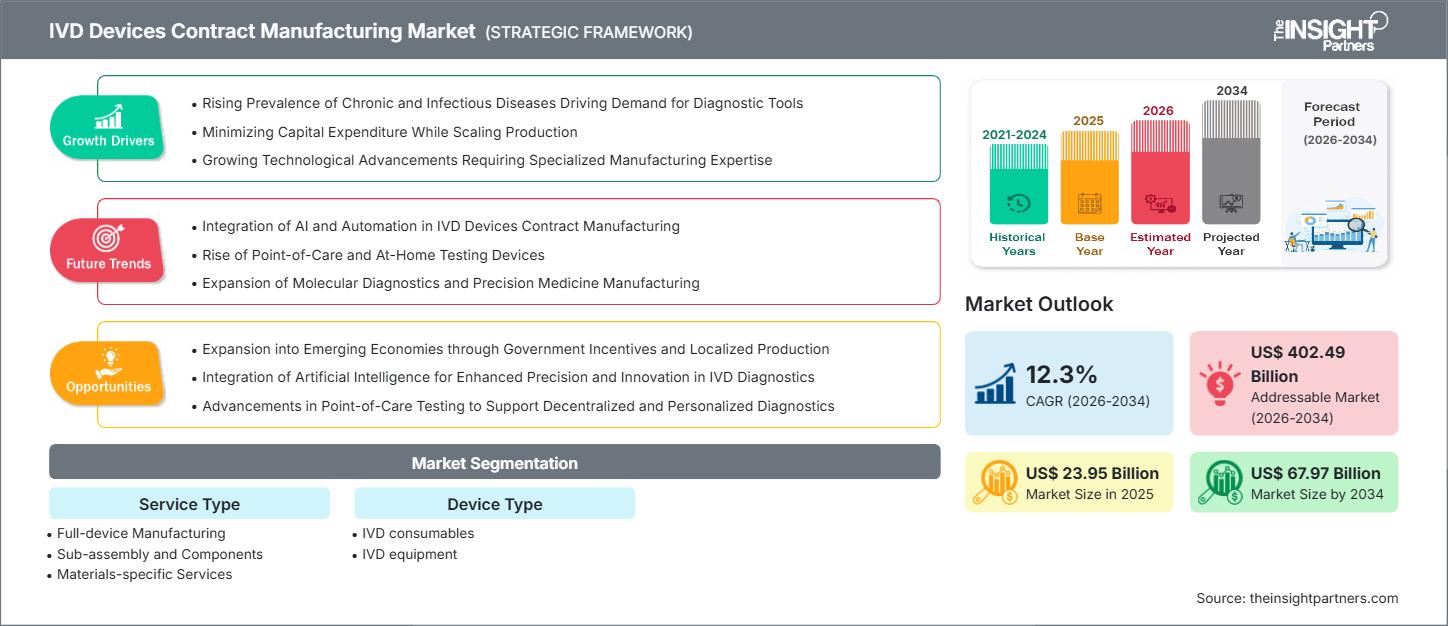
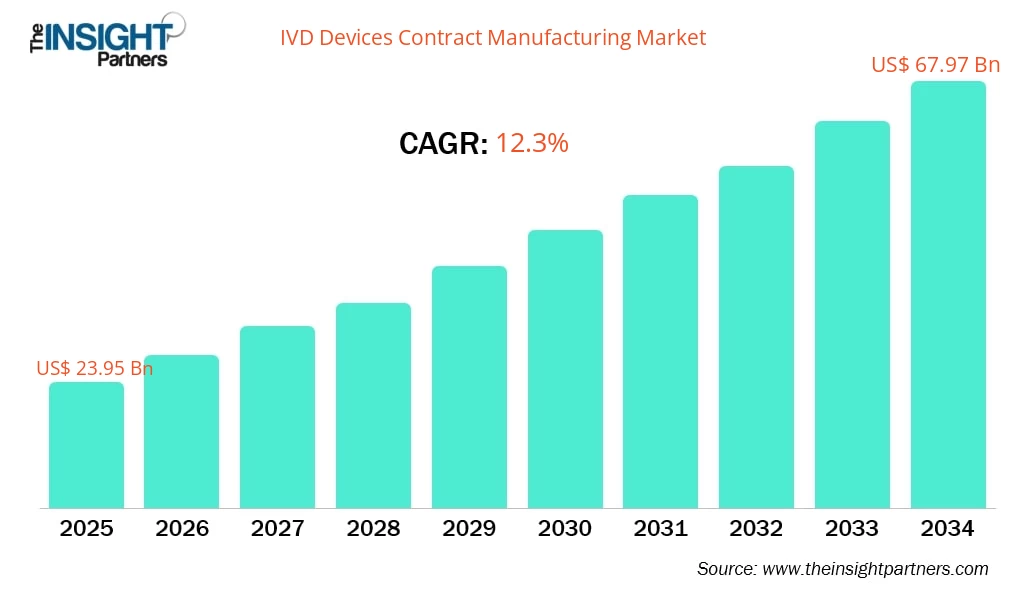
The IVD devices contract manufacturing market size is projected to reach US$ 67.79 billion by 2034 from US$ 23.95 billion in 2025. The market is expected to register a CAGR of 12.3% during 2026–2034.
IVD Devices Contract Manufacturing Market Analysis
The market growth is driven by the rising prevalence of chronic and infectious diseases, driving demand for diagnostic tools, and rapid technological advancements requiring specialized manufacturing expertise. Expansion into emerging economies through government incentives and localized production, and integration of artificial intelligence for enhanced precision and innovation in IVD diagnostics, are expected to create ample market opportunities.
IVD Devices Contract Manufacturing Market Overview
The market for IVD devices contract manufacturing consists of companies that offer outsourced services for the design, development, and manufacturing of IVD products, as well as supportive services to IVD companies. The growth of this market is driven by the demand for early disease detection, the increasing prevalence of chronic and infectious diseases, and the increase in the number of point-of-care and molecular diagnostics used by healthcare organizations. Contract manufacturers provide companies with significant expertise in regulatory compliance, quality systems, and scalable manufacturing, enabling IVD companies to lower their costs and improve their time-to-market. Newer technologies such as automation, microfluidics, and digital diagnostics have made outsourcing a more viable option, especially since access to healthcare in emerging markets is increasing, creating additional growth opportunities.
Market Research Highlights
- North America dominated the market with 35.5% share in 2025.
- Asia Pacific is poised to grow at a CAGR of 12.8% over the forecast period.
- United States market is projected to grow at a CAGR of 12.6% over the forecast period.
- By Service Type, the Full-device Manufacturing segment accounted for the largest market share of 62.5% in 2025.
- By Device Type, the IVD Consumables segment is anticipated to witness the fastest growth, registering a CAGR of 12.5% over the forecast period
- The report profiles key industry players such as TE Connectivity Ltd, Jabil Inc, Nova Biomedical Corporation, Flex Ltd, Celestica Inc, Fujirebio Europe NV, Thermo Fisher Scientific Inc., Nemera, SMC Ltd, HiArc, while also analyzing key developments in novel ideas, disruptive products, and innovative services that could reshape the future market and reveal emerging themes across the industry.
Customize This Report To Suit Your Requirement
Get FREE CUSTOMIZATIONIVD Devices Contract Manufacturing Market: Strategic Insights
-
Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
IVD Devices Contract Manufacturing Market Drivers and Opportunities
Market Drivers:
- Rising Disease Prevalence: As the number of chronic and infectious diseases continues to increase, there is a greater demand for diagnostic tools. There is a significant opportunity for OEMs to support OEMs with scalable and compliant manufacturing to support increased testing across the globe.
- Cost Savings and Operational Efficiencies: Outsourcing provides IVD companies with the ability to reduce both capital and labor costs, increase operational efficiency, expedite time to market, and develop innovative solutions by using the specialised knowledge of contract manufacturers.
- Technological Advancements: As diagnostic technologies continue to develop rapidly, such as molecular and digital, OEMs will require specialised manufacturing capabilities and is expected to continue to depend on contract manufacturers to achieve the level of precision, scalability, and rapid commercialisation required.
Market Opportunities:
- Expansion into Emerging Economies: Expansion into emerging economies allows IVD contract manufacturers to use government incentives, localized production, and the increasing amount spent on healthcare to provide affordable and scalable diagnostic solutions in areas with inadequate access.
- Integration of Artificial Intelligence: AI integration allows manufacturers to increase diagnostic accuracy, decrease the time needed for assay development, optimize manufacturing processes, and provide next-generation, data-driven diagnostic tools for detecting both infectious and chronic diseases.
- Advancements in Point-of-Care Testing: Recent advances in POC diagnostic devices present contract manufacturers with the opportunity to produce portable, rapid, and personalized diagnostic devices to support decentralized healthcare delivery and real-time clinical decision-making.
IVD Devices Contract Manufacturing Market Report Segmentation Analysis
The IVD devices contract manufacturing market is divided into different segments to give a clearer view of how it works, its growth potential, and the latest trends. Below is the standard segmentation approach used in most industry reports:
By Device Type:
- IVD Consumables: The market for contract manufacturing is driven by in vitro diagnostic consumables (IVD consumables), which constitute a large portion of recurring orders for reagents, consumable test kits (IVD Equipment), and other disposable materials. Manufacturers of IVD consumables also benefit from lower costs, increased scalability, increased regulatory alignment, and faster time-to-market.
- IVD Equipment: The company offers very high-quality instruments with advanced engineering and design capabilities for producing complex IVD diagnostic equipment to provide OEMs with reduced development costs, a chance to access advanced engineering, ensure regulatory compliance, and easily expand their business in the production of high-precision analyzers.
By Service Type:
- Full-device Manufacturing
- Sub-assembly and Components
- Materials-specific Services
Each end user in the market has distinct handling, safety, and regulatory needs, shaping equipment selection and operational protocols.
By Geography:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
The IVD devices contract manufacturing market in Asia Pacific is expected to witness the fastest growth. The cost-effective production, abundant skilled labor, and expanding healthcare infrastructure are likely to drive the market.
IVD Devices Contract Manufacturing Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2025 | US$ 23.95 Billion |
| Market Size by 2034 | US$ 67.97 Billion |
| Global CAGR (2026 - 2034) | 12.3% |
| Historical Data | 2021-2024 |
| Forecast period | 2026-2034 |
| Segments Covered |
By Service Type
|
| Regions and Countries Covered |
North America
|
| Market leaders and key company profiles |
|
IVD Devices Contract Manufacturing Market Players Density: Understanding Its Impact on Business Dynamics
The IVD Devices Contract Manufacturing Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
IVD Devices Contract Manufacturing Market Share Analysis by Geography
Asia Pacific is expected to grow at the fastest rate during the forecast period. Emerging markets in Latin America, the Middle East, and Africa also have many untapped opportunities for IVD devices contract manufacturing providers to expand.
The IVD devices contract manufacturing market grows differently across regions, as economic growth and rising healthcare expenditure. Below is a summary of market share and trends by region:
1. North America
- Market Share: Holds a significant portion of the global market
- Key Drivers:
- Developed healthcare systems & infrastructure, heavy investment in research and development, strong regulatory support, and increasing market demand for precision diagnostics
- Trends: Increase in point-of-care testing (POCT) and improved molecular biology testing technologies
2. Europe
- Market Share: Substantial share due to early adoption of IVD devices contract manufacturing
- Key Drivers:
- The establishment of research and development, increasing demand for personalized & high-quality diagnostics, generates consistently growing markets.
- Trends: The growth of personalised diagnostics, as well as the need for production that meets government standards
3. Asia Pacific
- Market Share: Fastest-growing region with rising market share every year
- Key Drivers:
- Investments in healthcare infrastructure, increased spending on healthcare, and established manufacturing capabilities in cost-competitive areas
- Trends: Due to an increase in investments into the healthcare sector and the availability of affordable manufacturing options
4. South and Central America
- Market Share: Steadily progressing market share
- Key Drivers:
- Growing healthcare access, increased burden associated with chronic disease, and increasing demand for local manufacturing contribute to the market growth.
- Trends: Emerging diagnostic technologies and an increase in healthcare investments
5. Middle East and Africa
- Market Share: Small market share, growing at a rapid pace
- Key Drivers:
- Recovery of healthcare access and investments by the government to improve infrastructure and mitigate the impact of infectious diseases.
- Trends: Investment into building up the healthcare sector and expanding production capabilities through outsourcing
IVD Devices Contract Manufacturing Market Players Density: Understanding Its Impact on Business Dynamics
High Market Density and Competition
Competition is strong due to the presence of established players, such as Jabil Inc, Flex Ltd, and TE Connectivity Ltd, which are also adding to the competitive landscape.
This high level of competition urges companies to stand out by offering:
- Advanced security features
- Value-added services such as analytics and predictive maintenance, real‑time operational analytics, and installation
- Competitive pricing models
- Strong customer support and easy integration
Opportunities and Strategic Moves
- Outsourcing trends, POCT demand, and tech integration, such as AI, drive the market. Up to 40% of IVD firms outsource to cut costs and speed innovation, with over 100 FDA approvals for outsourced products in recent years. This aligns with your prior analyses of medical device markets, emphasizing scalable production amid rising diagnostics needs.
- Rising POCT and telehealth integration create demand for portable, connected IVD equipment, as portable devices surged 22% in innovations per NIH data. Environmental and infectious disease concerns boost rapid testing needs, while regional hubs mitigate supply chain risks from geopolitics. Flexible manufacturing for customized solutions, supported by 30% investment growth in such tech, suits emerging markets such as Asia Pacific.
Other companies analysed during the course of research:
- Sanmina Corporation
- Celestica Inc.
- Thermo Fisher Scientific Inc.
- Savyon Diagnostics Ltd.
- West Pharmaceutical Services, Inc.
- KMC Systems, Inc.
- Cenogenics Corporation
- Nova Biomedical
- Cone Bioproducts
- Invetech (Danaher Corporation)
- AVIOQ Inc.
- Meridian Bioscience Inc.
- Kimball Electronics, Inc.
- Nemera
- Phillips Medsize Corporation
- Nolato GW, Inc.
- Stratec SE
- Fujirebio
- Biokit S.A.
- Affinity Lifesciences, Inc.
- Sekisui
- Prestige Diagnostics UK Ltd.
- Argonaut Manufacturing Services Inc.
- Bio Techne Corp.
- CorDx
- Genemed Biotechnologies Inc.
- HDA Technology Inc.
- HU Group Holdings Inc.
- Jena Bioscience GmbH
- JSR Corp.
- More Diagnostics Inc.
- Neogen Corp.
- PHC Holdings Corp.
- TCS Biosciences Ltd.
- LRE Medical
- Maxim Biomedical, Inc.
- Coris BioConcept
- In-Vitro Diagnostics Developers Inc.
- Cone Bioproducts
- Cenogenics Corporation
- Merck KGaA
- Biofortuna Inc.
- Medix Biochemica
- Diaclone Inc.
- TE Connectivity Ltd.
IVD Devices Contract Manufacturing Market News and Recent Developments
- DCN Dx Acquires IVD Vision, a Developer of Diagnostics Instruments and Software: In June 2021, DCN Dx completed its acquisition of IVD Vision, Inc., a developer of custom readers, sample collection and handling devices, and software for diagnostics systems.
IVD Devices Contract Manufacturing Market Report Coverage and Deliverables
The "IVD Devices Contract Manufacturing Market Size and Forecast (2021–2034)" report provides a detailed analysis of the market covering below areas:
- IVD Devices Contract Manufacturing Market size and forecast at global, regional, and country levels for all the key market segments covered under the scope
- IVD Devices Contract Manufacturing Market trends, as well as market dynamics such as drivers, restraints, and key opportunities
- Detailed PEST and SWOT analysis
- IVD Devices Contract Manufacturing Market analysis covering key market trends, global and regional framework, major players, regulations, and recent market developments
- Industry landscape and competition analysis covering market concentration, heat map analysis, prominent players, and recent developments for the IVD Devices Contract Manufacturing Market
- Detailed company profiles
Frequently Asked Questions
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Global, Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Recent Reports
Testimonials
Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends
Unlock Exclusive Report Discounts
Enquire Now









 Get Free Sample For
Get Free Sample For