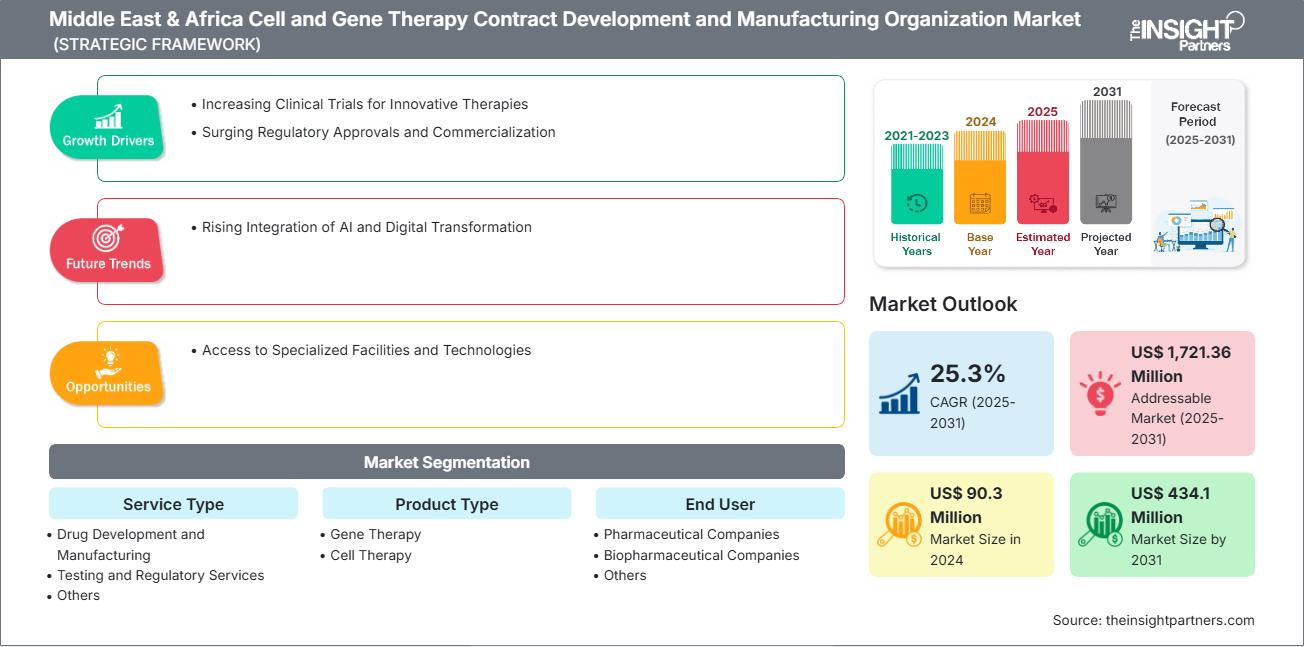
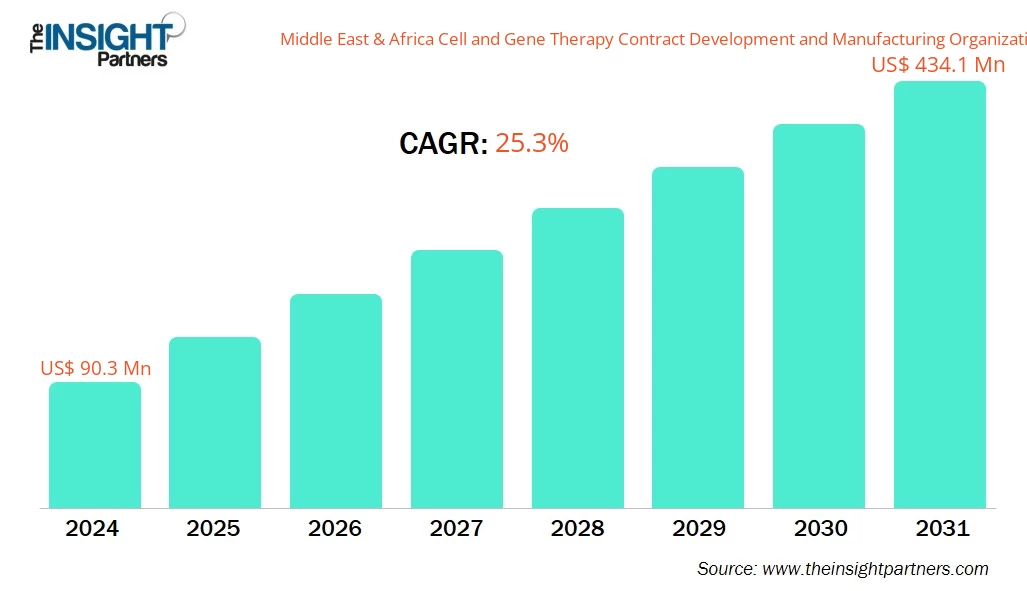
The Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market size is expected to reach US$ 434.1 Million by 2031 from US$ 90.3 Million in 2024. The market is estimated to record a CAGR of 25.3% from 2025 to 2031.
Executive Summary and Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market Analysis:
The cell and gene therapy contract development and manufacturing organization market in the Middle East & Africa is segmented into the UAE, Saudi Arabia, South Africa, and the Rest of Middle East & Africa. The market growth is driven by a combination of increasing healthcare investments, rising prevalence of chronic and genetic diseases, and growing interest in advanced therapeutic modalities. Governments in countries such as the UAE and Saudi Arabia are actively investing in biotechnology and life sciences as part of broader economic diversification strategies, like Saudi Vision 2030. These initiatives aim to reduce reliance on oil revenues by fostering innovation-driven sectors, including biotechnology and personalized medicine. As a result, there is a growing demand for local and regional expertise in the development and manufacturing of complex therapies like cell and gene therapies. Additionally, many organizations are increasingly investing in improving the manufacturing capabilities of cell and gene therapy. Academic and research institutions are also becoming more active in cell and gene therapy RandD, often collaborating with CDMOs to bridge the gap between discovery and commercialization.
Customize This Report To Suit Your Requirement
Get FREE CUSTOMIZATIONMiddle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market: Strategic Insights
-
Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market Segmentation Analysis:
- By Service Type, the Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market is segmented into Drug Development and Manufacturing, Testing and Regulatory Services, and Others. Drug Development and Manufacturing held the largest share of the market in 2024.
- By Product Type, the Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market is segmented into Gene Therapy and Cell Therapy. Gene Therapy held the largest share of the market in 2024.
- By End User, the Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market is segmented into Pharmaceutical Companies, Biopharmaceutical Companies, and Others. Biopharmaceutical Companies held the largest share of the market in 2024.
Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2024 | US$ 90.3 Million |
| Market Size by 2031 | US$ 434.1 Million |
| CAGR (2025 - 2031) | 25.3% |
| Historical Data | 2021-2023 |
| Forecast period | 2025-2031 |
| Segments Covered |
By Service Type
|
| Regions and Countries Covered |
Middle East & Africa
|
| Market leaders and key company profiles |
|
Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market Players Density: Understanding Its Impact on Business Dynamics
The Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market Outlook
The integration of AI, machine learning, and other digital technologies into manufacturing and production processes allows CDMOs to streamline operations, optimize efficiency, reduce costs, and maintain compliance with regulatory standards. With increasing complexities in cell and gene therapies, the use of these technologies is proving essential to meet the growing demand for scalable solutions and high-quality. AI and digital tools are revolutionizing various aspects of gene therapy manufacturing. For instance, in February 2025, NecstGen and Digi.Bio, collaborated to improve testing and development for Cell Therapies. This partnership utilises Digi.Bio's cutting-edge platform to deliver dynamic multiparametric single-cell immunoprofiling, aiming to accelerate the journey from concept to therapeutic. The increasing application of AI in process optimization and predictive analytics is expected to emerge as a major trend in the coming years. Machine learning algorithms are being used to monitor real-time data from temperature, pH levels, and cell viability to predict potential failures or deviations before actual occurrence. This predictive capability enables CDMOs to take corrective actions proactively, reducing downtime and improving the quality and yield of therapeutic products. For instance, in October 2024, OmniaBio opened its new commercial-ready manufacturing facility and AI centre of excellence in Hamilton, Ontario, Canada. At 100,000 sq. ft., It is one of the largest CDMO dedicated to cell and gene therapy. Similarly, in August 2024, Researchers at the Broad Institute of MIT and Harvard have developed a machine-learning approach that promises to speed up AAV engineering for gene therapy.
Traditional cell culture techniques for gene therapies are labor-intensive and time-consuming, often requiring constant monitoring. However, AI-based systems can automate aspects of this process, including cell growth monitoring, optimizing nutrient delivery, and adjusting environmental conditions. This automation speeds up production timelines and reduces human error, which is crucial in gene therapy where precision is key. For instance, BioMarin Pharmaceutical has implemented AI systems to automate cell culture monitoring in their gene therapy production processes, resulting in improvements in productivity and scalability.
Gene therapy manufacturing involves vast amounts of data that must be recorded and analyzed for regulatory purposes. AI and digital tools enable seamless data integration, ensuring that all necessary documentation is complete and accurate for regulatory review. This integration is important as the FDA and EMA continue to maintain the stringent standards for gene therapy manufacturing. CDMOs that leverage AI and digital platforms can better manage the complexities of regulatory submissions and ensure compliance with evolving standards.
Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market Country Insights
By country, the Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market is segmented into Saudi Arabia, South Africa, the United Arab Emirates, and the Rest of Middle East & Africa. South Africa held the largest share in 2024.
South Africa's established pharmaceutical industry, exemplified by companies such as Aspen Pharmacare, provides a strong foundation for the growth of CDMO services in the cell and gene therapy sector. Aspen's extensive experience in manufacturing biologics and active pharmaceutical ingredients positions it as a potential leader in the contract manufacturing space for advanced therapies. The company's existing infrastructure and expertise can be leveraged to support the production of cell and gene therapies, meeting both local and international demands.
According to the article Rare is everywhere : Embracing rare disease in South Africa, published by Illumina in February 2022, more than 4.1 million individuals in South Africa were living with a rare disease, and one in 15 people will contract a rare disease in the country at some point in their lives. These conditions underscore the necessity for advanced therapeutic solutions, including gene therapies. CDMOs play a significant role in advancing the treatment of rare diseases through innovations in cell and gene therapies by providing specialized manufacturing infrastructure, regulatory support, and clinical trial services to companies. In conclusion, the combination of a strong pharmaceutical industry, growing disease prevalence, and supportive government policies favors South Africa's growth in cell and gene therapy CDMO landscape.
Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market Company Profiles
Some of the key players operating in the market include Charles River Laboratories International Inc, Catalent Inc, Lonza Group AG, WuXi Biologics Inc, Pluri Inc, Aenova Holding GmbH, Takara Bio Inc, FUJIFILM Holdings Corp, AGC Biologics AS, Thermo Fisher Scientific Inc., Minaris Advanced Therapies, and SK pharmteco Inc
These players are adopting various strategies such as expansion, product innovation, and mergers and acquisitions to provide innovative products to their consumers and increase their market share.
Middle East & Africa Cell and Gene Therapy Contract Development and Manufacturing Organization Market Research Methodology
The following methodology has been followed for the collection and analysis of data presented in this report:
Secondary Research
The research process begins with comprehensive secondary research, utilizing internal and external sources to gather qualitative and quantitative data for each market. Commonly referenced secondary research sources include, but are not limited to:
- Company websites, annual reports, financial statements, broker analyses, and investor presentations
- Industry trade journals and other relevant publications
- Government documents, statistical databases, and market reports
- News articles, press releases, and webcasts specific to companies operating in the market
Note: All financial data included in the Company Profiles section has been standardized to US$. For companies reporting in other currencies, figures have been converted to US$ using the relevant exchange rates for the corresponding year.
Primary Research
The Insight Partners conducts a significant number of primary interviews each year with industry stakeholders and experts to validate its data analysis and gain valuable insights. These research interviews are designed to:
- Validate and refine findings from secondary research
- Enhance the expertise and market understanding of the analysis team
- Gain insights into market size, trends, growth patterns, competitive dynamics, and future prospects
Primary research is conducted via email interactions and telephone interviews, encompassing various markets, categories, segments, and sub-segments across different regions. Participants typically include:
- Industry stakeholders: Vice Presidents, Business Development Managers, Market Intelligence Managers, and National Sales Managers
- External experts: Valuation specialists, research analysts, and key opinion leaders with industry-specific expertise
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Recent Reports
Testimonials
Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends







 Get Free Sample For
Get Free Sample For