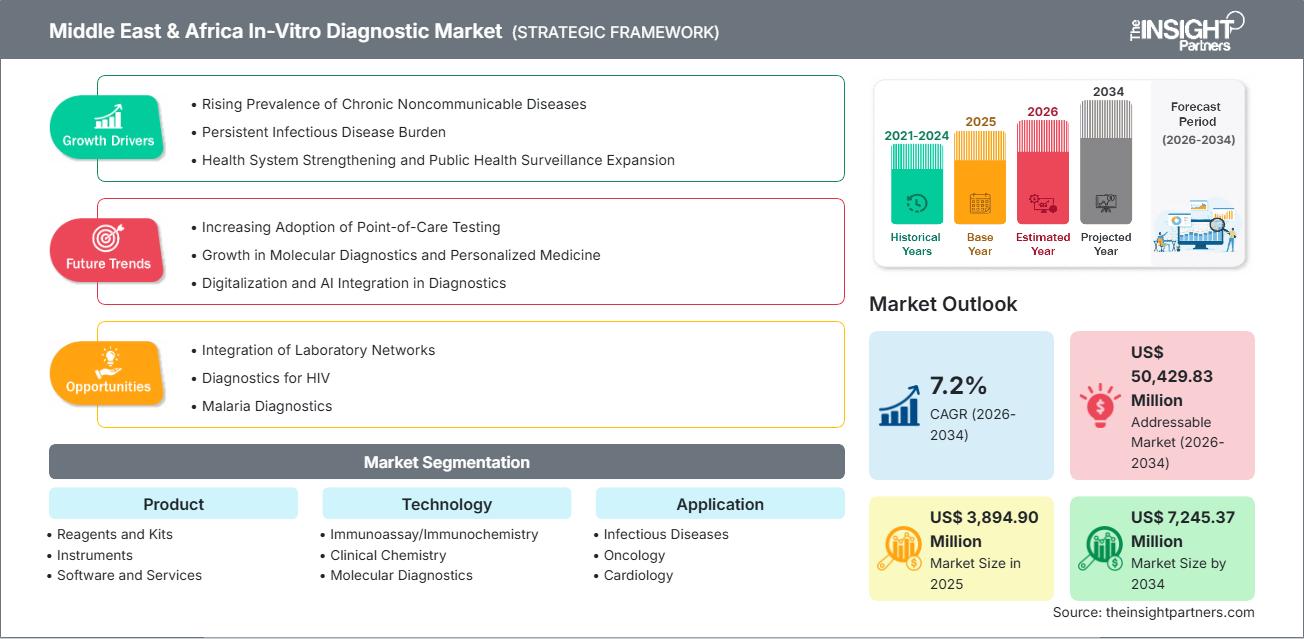
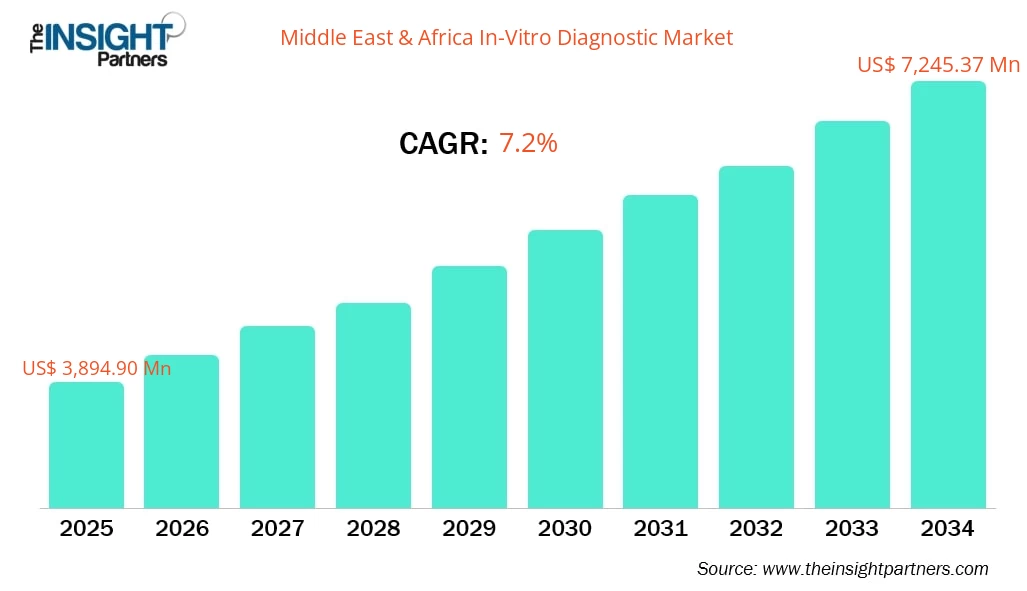
The MEA in-vitro diagnostics market size is projected to reach US$ 7,245.37 million by 2034 from US$ 3,894.90 million in 2025. The market is expected to register a CAGR of 7.2% from 2026 to 2034.
MEA In-Vitro Diagnostics Market Analysis
The increasing prevalence of infectious and chronic diseases propels the demand for disease testing during the early stages of diagnosis. African nations face challenges because their hospital systems lack sufficient capacity. Their laboratories require better performance, which drives the use of point-of-care and decentralized diagnostic methods. Market expansion is supported by government healthcare system improvement projects, together with growing investments in private healthcare and advancements in laboratory automation and molecular diagnostic technology. People are becoming more conscious about preventive health methods. Remote testing services are becoming more widely available. Digital health systems with data management tools are being progressively adopted.
MEA In-Vitro Diagnostics Market Overview
Centralized laboratory testing and new point-of-care diagnostics provide quick results to solve accessibility problems. The focus of testing infectious diseases includes HIV and tuberculosis and malaria and hepatitis, and COVID-19. The need for clinical chemistry tests and immunoassays, and molecular diagnostics tests is increasing to manage chronic diseases. Gulf Cooperation Council (GCC) countries are leading the adoption due to higher healthcare spending, advanced laboratory infrastructure, and strong government support. African markets are gradually expanding through public health programs and international funding. The diagnostic industry operates through a divided market, which international companies and regional distributors, and local manufacturers use to create diagnostic solutions that enhance medical testing across the region.
Customize This Report To Suit Your Requirement
Get FREE CUSTOMIZATIONMiddle East & Africa In-Vitro Diagnostic Market: Strategic Insights
-
Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
MEA In-Vitro Diagnostics Market Drivers and Opportunities
Market Drivers:
- Rising Prevalence of Chronic Noncommunicable Diseases: The rising number of people with diabetes, cardiovascular diseases, cancer, and kidney disorders throughout the MEA drives the IVD demand. The GCC countries experience rising demand for clinical chemistry, immunoassays, and molecular diagnostics, driven by urbanization, changing lifestyles, and population aging, which require regular health assessments.
- Persistent Infectious Disease Burden: The region shows high rates of infectious disease transmission because people now have better access to healthcare services than they did before. The ongoing presence of diseases creates a strong need for testing through serology and molecular and rapid diagnostic tests, which Sub-Saharan Africa requires to achieve early disease detection and essential population screenings for disease management.
- Health System Strengthening and Public Health Surveillance Expansion: National governments and worldwide organizations direct their funding toward laboratory development projects and national testing initiatives, and disease monitoring systems. The healthcare sector in public hospitals speeds up the implementation of automated IVD solutions through initiatives that strengthen diagnostic skills and outbreak response, and public health analysis using data.
Market Opportunities:
- Integration of Laboratory Networks: Laboratory network digitization enables test efficiency improvements through enhanced data-sharing capabilities and better-quality control processes. The establishment of centralized reference laboratories, peripheral testing facilities, and point-of-care testing centers enable healthcare systems to achieve better service delivery through reduced service times and more efficient use of available resources, which is essential for low- and middle-income nations.
- Diagnostics for HIV: HIV diagnostics remain critical in MEA, as global funding programs and national elimination strategies provide essential support. The ongoing need for early infant diagnosis and viral load testing, together with rapid screening assays, creates extensive demand for molecular and point-of-care HIV diagnostic technologies.
- Malaria Diagnostics: Rapid diagnostic tests and microscopy solutions experience ongoing demand because malaria remains an endemic disease throughout multiple African nations. The region will experience persistent growth in malaria diagnostics, driven by public health programs, donor-funded initiatives, and improved access to healthcare in rural areas.
MEA In-Vitro Diagnostics Market Report Segmentation Analysis
The MEA In-Vitro Diagnostics market is divided into different segments to give a clearer view of how it works, its growth potential, and the latest trends. Below is the standard segmentation approach used in industry reports:
By Product:
- Reagents and Kits: Medical testing facilities use reagents and kits to meet their high testing needs for detecting infectious diseases, including HIV, malaria, tuberculosis, hepatitis, diabetes, and cardiovascular diseases. Public health programs and screening programs, and hospital laboratories create ongoing demand for assay kits and controls, and reagents which support clinical chemistry and immunoassay, and molecular diagnostics.
- Instruments: IVD instruments support diagnostic testing across centralized laboratories and point-of-care settings through their analyzers, which include clinical chemistry and hematology and immunoassay, and molecular diagnostics components. GCC countries experience rising adoption rates because of their financial commitment to developing advanced laboratory facilities. African markets grow through funding from donors and their ongoing shift to automated and semi-automated systems, which enhance accuracy and throughput.
- Software and Services: Software and services include middleware and remote diagnostics and maintenance services. Public health surveillance and integrated laboratory networks across the region receive support from digital solutions, which provide improved data management and connectivity, and quality assurance capabilities.
By Technology:
- Immunoassay/Immunochemistry
- Clinical Chemistry
- Molecular Diagnostics
- Microbiology
- Urinalysis
- Coagulation and Hemostasis
- Hematology
- Blood Gas Analyzers
- Glucose Monitoring
- Others
By Application:
- Infectious Diseases
- Oncology
- Cardiology
- Nephrology
- Diabetes
- Genetic Diseases
- Blood Grouping and Testing
- Allergy Diagnostics
- Gastrointestinal Diseases
- Immune System Disorders
- Others
By Usability:
- Disposable Devices
- Reusable Equipment
By Site of Testing:
- Laboratory Tests
- Point of Care Tests
By Specimen:
- Blood, Serum, and Plasma
- Saliva Specimens
- Urine Specimens
- Others
By End User
- Hospitals
- Clinical Laboratories
- Pharmaceutical and Biopharmaceutical Companies
- Home Care
- Others
By Geography:
- Middle East & Africa
Middle East & Africa In-Vitro Diagnostic Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2025 | US$ 3,894.90 Million |
| Market Size by 2034 | US$ 7,245.37 Million |
| CAGR (2026 - 2034) | 7.2% |
| Historical Data | 2021-2024 |
| Forecast period | 2026-2034 |
| Segments Covered |
By Product
|
| Regions and Countries Covered |
Middle East and Africa
|
| Market leaders and key company profiles |
|
Middle East & Africa In-Vitro Diagnostic Market Players Density: Understanding Its Impact on Business Dynamics
The Middle East & Africa In-Vitro Diagnostic Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
MEA In-Vitro Diagnostics Market Share Analysis by Geography
The MEA in-vitro diagnostics market grows differently in each nation due to the increasing prevalence of infectious & chronic disorders, surging government initiatives, and rising inclination toward in-vitro diagnostics. Below is a summary of market share and trends by region:
1. Middle East & Africa
- Market Share: Saudi Arabia holds a significant portion of the Middle East & Africa market
- Key Drivers: Rapidly aging population, rising prevalence of chronic diseases, and integration of laboratory networks
- Trends: Digital Integration and AI and Expansion of Laboratory Networks
MEA In-Vitro Diagnostics Market Players Density: Understanding Its Impact on Business Dynamics
High Market Density and Competition
Competition is intense due to the presence of established players such as Roche Diagnostics, Abbott Diagnostics, and Thermo Fisher Scientific. Regional and niche players add to the competitive landscape across regions.
This high level of competition urges companies to stand out by offering:
- Advanced Products
- Value-added services such as customization and sustainable solutions
- Competitive pricing models
- Regulatory compliance
Opportunities and Strategic Moves
- Through national screening programs that detect chronic diseases and cancers, as well as infectious diseases such as HIV, tuberculosis, and hepatitis, countries in the GCC, specifically Saudi Arabia, the UAE, and Qatar, aim to reduce healthcare expenses and disease burden by developing new diagnostic capabilities.
- The expansion of diagnostic services will take place through investments in laboratory infrastructure, the development of point-of-care testing, and the implementation of digital laboratory systems, which national health transformation agendas, public-private partnerships, and funding from global health organizations will use to improve access, particularly in underserved and rural regions.
- Companies are forming partnerships while implementing localization strategies that include manufacturing reagents, establishing distribution agreements, and transferring technology to expand their market reach and maintain dependable supply networks across Africa and the Middle East.
- Health systems are focusing on integration, quality, and workforce training to strengthen laboratory networks and surveillance systems. Public health authorities, hospitals, laboratories, and IVD manufacturers use automation, connectivity, and data-driven diagnostics to create systems that enhance disease monitoring and population health outcomes.
Other companies analyzed during the course of research:
- bioMérieux SA
- QIAGEN NV
- Bio-Rad Laboratories, Inc.
- Quidel Corporation
- Ortho Clinical Diagnostics
- PerkinElmer Inc.
- Agilent Technologies Inc.
- Hologic, Inc.
- Mindray Bio-Medical Electronics Co., Ltd.
- Autobio Diagnostics Co., Ltd.
- Randox Laboratories Ltd.
- Spectrum Diagnostics
MEA In-Vitro Diagnostics Market News and Recent Developments
- In December 2022, F. Hoffmann-La Roche Ltd. announced that it had secured ISO27001 Information Security certification, in recognition of its commitment to protecting critical patient data and building trust across the region. It is the first company in the Middle East & Africa to achieve this prestigious certification.
- In September 2022, Nanōmix Corporation announced that it had entered into a distribution agreement with IPS Genomix s.a.l., a medical diagnostics distributor in the Middle East. Under the distribution agreement, IPS would market and distribute the Nanōmix eLab system, including the Nanōmix S1 Panel assay and other future test products for the eLab system, in multiple countries throughout the Middle East.
- In March 2022, Audere entered a partnership with Medical Diagnostech, headquartered in South Africa, a developer and manufacturer of lateral flow rapid diagnostic test kits. This partnership will pair Medical Diagnostech's MD SARS-CoV-2 antigen device with Audere's HealthPulse digital companion app.
MEA In-Vitro Diagnostics Market Report Coverage and Deliverables
The " MEA In-Vitro Diagnostics Market Size and Forecast (2021–2034)" report provides a detailed analysis of the market covering below areas:
- In-vitro diagnostics market size and forecast at global, regional, and country levels for all the segments covered under the scope
- In-vitro diagnostics market trends, as well as dynamics such as drivers, restraints, and key opportunities
- Detailed PEST and SWOT analysis
- In-vitro diagnostics market analysis covering key trends, global and regional framework, major players, regulations, and recent developments
- Industry landscape and competition analysis covering market concentration, heat map analysis, prominent players, and recent developments for the market
- Detailed company profiles
Frequently Asked Questions
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Recent Reports
Testimonials
Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends







 Get Free Sample For
Get Free Sample For