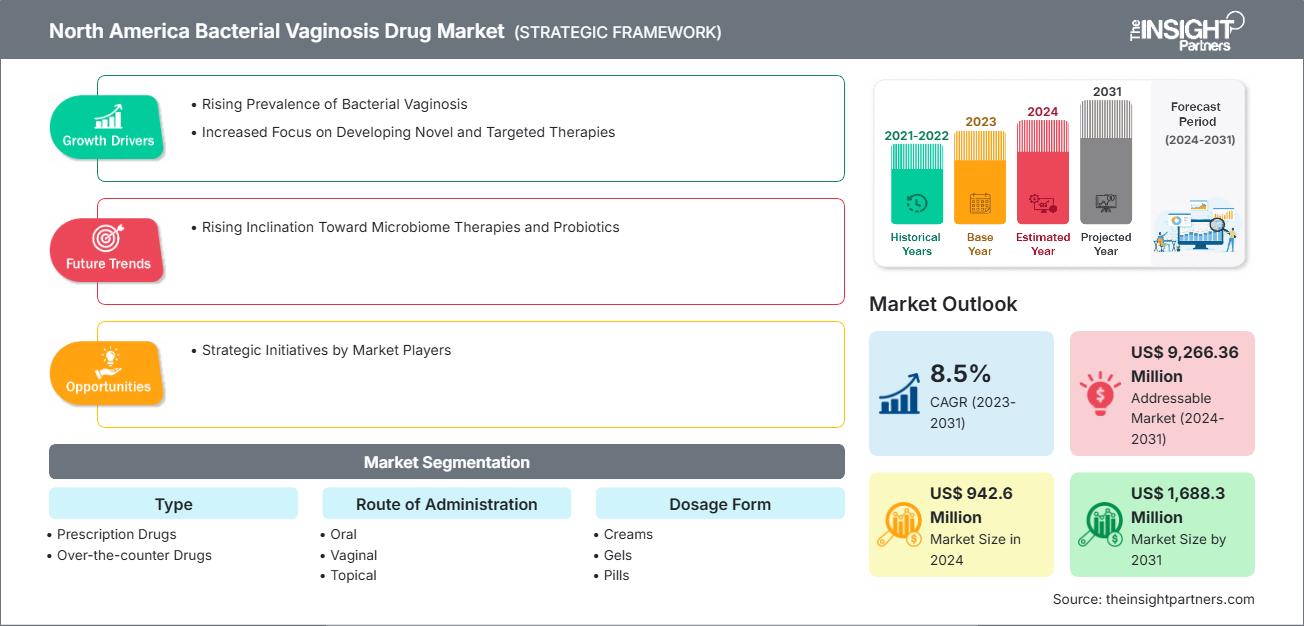
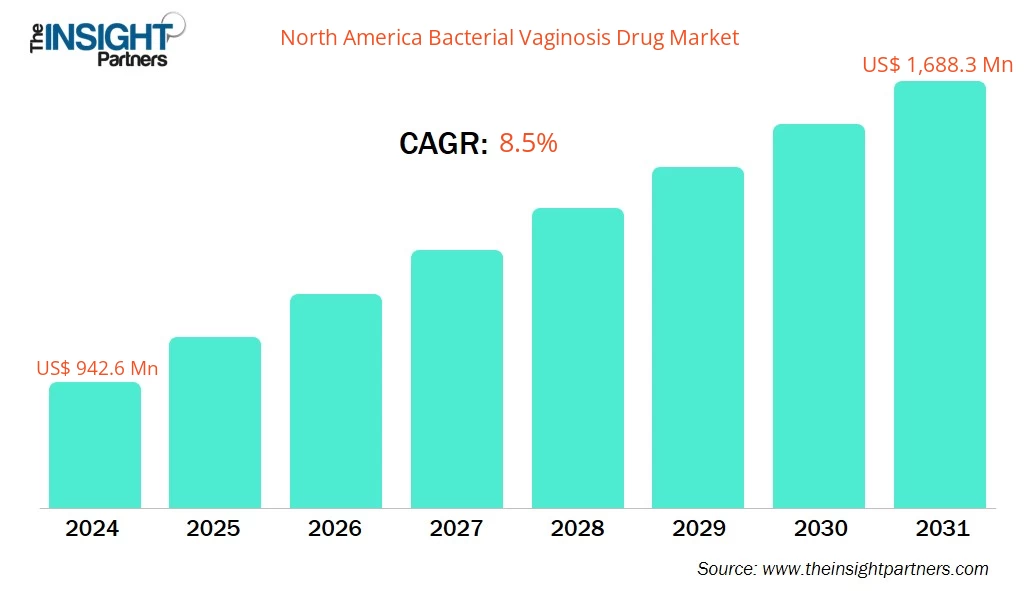
The North America Bacterial Vaginosis Drug Market size is expected to reach US$ 1,688.3 Million by 2031 from US$ 942.6 Million in 2024. The market is estimated to record a CAGR of 8.5% from 2023 to 2031.
Executive Summary and North America Bacterial Vaginosis Drug Market Analysis:
The bacterial vaginosis drug market in North America is segmented into the US, Canada, and Mexico. The increasing prevalence of bacterial vaginosis (BV), rising awareness about women's health, and the ongoing development of innovative treatments favor the growth of the market. Government initiatives in the US contribute to promoting and spreading awareness of vaginosis in the region. In addition, increasing healthcare access, rising disposable incomes, and an expanding range of treatment options are propelling the growth of the market during the forecast period
Customize This Report To Suit Your Requirement
Get FREE CUSTOMIZATIONNorth America Bacterial Vaginosis Drug Market: Strategic Insights
-
Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
North America Bacterial Vaginosis Drug Market Segmentation Analysis:
- By Type, the North America Bacterial Vaginosis Drug Market is segmented into Prescription Drugs (Rx) and Over-the-counter Drugs (OTC). Prescription Drugs (Rx) held the largest share of the market in 2024.
- By Route of Administration, the North America Bacterial Vaginosis Drug Market is segmented into Oral, Vaginal, and Topical. Oral held the largest share of the market in 2024.
- By Dosage Form, the North America Bacterial Vaginosis Drug Market is segmented into Pills, Creams, Gel, Solution or Washes, and Others. Pills held the largest share of the market in 2024.
- By Distribution Channel, the North America Bacterial Vaginosis Drug Market is segmented into Hospital Pharmacies, Online Pharmacies, and Retail Pharmacies. Hospital Pharmacies held the largest share of the market in 2024.
North America Bacterial Vaginosis Drug Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2024 | US$ 942.6 Million |
| Market Size by 2031 | US$ 1,688.3 Million |
| CAGR (2023 - 2031) | 8.5% |
| Historical Data | 2021-2022 |
| Forecast period | 2024-2031 |
| Segments Covered |
By Type
|
| Regions and Countries Covered |
North America
|
| Market leaders and key company profiles |
|
North America Bacterial Vaginosis Drug Market Players Density: Understanding Its Impact on Business Dynamics
The North America Bacterial Vaginosis Drug Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
North America Bacterial Vaginosis Drug Market Outlook
Bacterial vaginosis (BV) is a common condition experienced by women of childbearing age, which is generally caused by changes in the normal bacterial flora of the vagina. BV is characterized by a decrease in Lactobacillus species and an increase in anaerobic bacteria such as Urea plasma urealyticum, Gardnerella vaginalis, Mobiluncus species, Mycoplasma hominis, and Prevotella species. Activities such as unprotected sexual intercourse and frequent douching further increase the risk of BV. Common symptoms of the condition include abnormal vaginal discharge, odor, and itching. According to the European Journal of Obstetrics & Gynecology and Reproductive Biology article published in ScienceDirect Journal in 2023, the global prevalence of BV ranges from 23% to 29%, and in pregnant women, it ranges from 11% to 49%. According to a report by Medscape in 2024, approximately one-third of adult women, i.e., ~22 million, in the US are affected by BV every year, and 10 million women visit clinics for vaginal discharge. BV poses risks such as preterm labor, miscarriage, and increased susceptibility to HIV and other sexually transmitted infections. The increasing incidence of BV, coupled with potential complications, encourages women to seek medical attention and pharmaceutical interventions, creating significant demand for effective drugs. The high chances of recurrence of BV lead to the development of treatment regimens to prevent relapses. As healthcare access improves globally, especially in emerging markets, a larger number of women are now seeking treatment for BV, driving the growth of the bacterial vaginosis drugs market.
North America Bacterial Vaginosis Drug Market Country Insights
By country, the North America Bacterial Vaginosis Drug Market is segmented into the United States, Canada, and Mexico. The United States held the largest share in 2024.
In the US, the bacterial vaginosis drug market is experiencing significant growth due to the increasing prevalence of bacterial vaginosis (BV), rising awareness about women's health, and the ongoing development of innovative treatments. BV is one of the most common vaginal infections in women, particularly affecting those of reproductive age. According to a report by Medscape in 2024, approximately one-third of adult women in the US, i.e., ~22 million females, are affected by BV annually, and 10 million women visit clinics for abnormal vaginal discharge. It is often linked to factors such as sexual activity, douching, and hormonal changes, although its exact cause remains complex. The primary therapeutic options available for treating BV include antibiotics approved by the US Food and Drug Administration (FDA), such as metronidazole and clindamycin. Metronidazole, often available in oral and topical forms, is considered the first-line treatment for BV. Clindamycin is also commonly available in both oral and topical formulations. Newer drugs and formulations, such as probiotics and phage therapy, are also emerging to help restore the natural vaginal flora, thereby reducing the recurrence of BV.
North America Bacterial Vaginosis Drug Market Company Profiles
Some of the key players operating in the market include Bayer AG, Pfizer Inc., Viatris Inc., Sanofi SA, Sun Pharmaceutical Industries Ltd, Lupin Ltd, Glenmark Pharmaceuticals Ltd, Organon & Co., Starpharma Holdings Limited, and Duchesnay Inc., Canada.
These players are adopting various strategies such as expansion, product innovation, and mergers and acquisitions to provide innovative products to their consumers and increase their market share.
North America Bacterial Vaginosis Drug Market Research Methodology
The following methodology has been followed for the collection and analysis of data presented in this report:
Secondary Research
The research process begins with comprehensive secondary research, utilizing internal and external sources to gather qualitative and quantitative data for each market. Commonly referenced secondary research sources include, but are not limited to:
- Company websites, annual reports, financial statements, broker analyses, and investor presentations
- Industry trade journals and other relevant publications
- Government documents, statistical databases, and market reports
- News articles, press releases, and webcasts specific to companies operating in the market
Note:
All financial data included in the Company Profiles section has been standardized to US$. For companies reporting in other currencies, figures have been converted to US$ using the relevant exchange rates for the corresponding year.Primary Research
The Insight Partners conducts a significant number of primary interviews each year with industry stakeholders and experts to validate its data analysis and gain valuable insights. These research interviews are designed to:
- Validate and refine findings from secondary research
- Enhance the expertise and market understanding of the analysis team
- Gain insights into market size, trends, growth patterns, competitive dynamics, and future prospects
Primary research is conducted via email interactions and telephone interviews, encompassing various markets, categories, segments, and sub-segments across different regions. Participants typically include:
- Industry stakeholders: Vice Presidents, Business Development Managers, Market Intelligence Managers, and National Sales Managers
- External experts: Valuation specialists, research analysts, and key opinion leaders with industry-specific expertise
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Recent Reports
Testimonials
Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends







 Get Free Sample For
Get Free Sample For