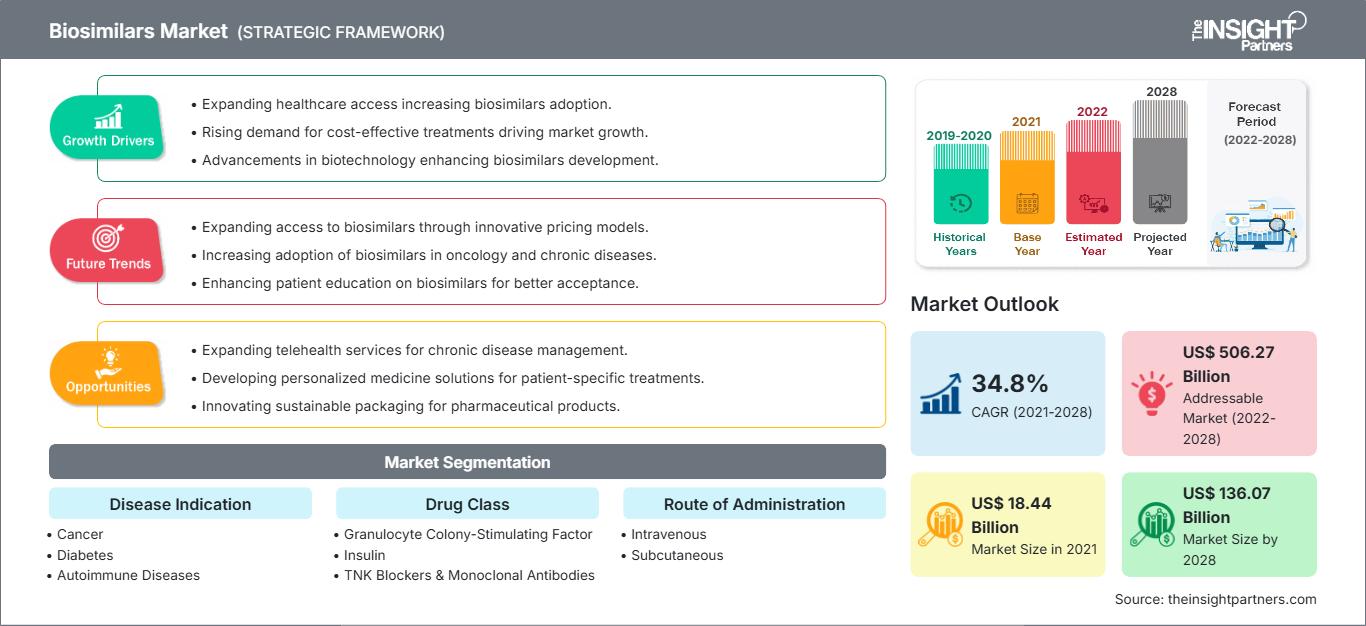
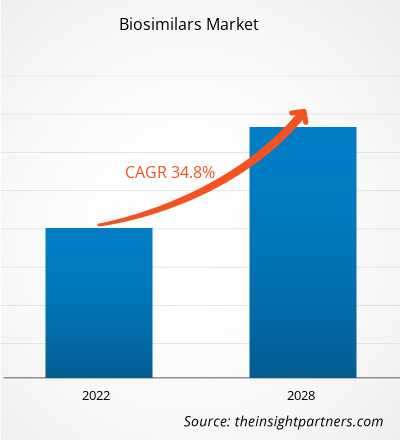
[Forschungsbericht]Der Markt für Biosimilars soll von 18.435,89 Millionen US-Dollar im Jahr 2021 auf 136.069,53 Millionen US-Dollar im Jahr 2028 wachsen; von 2022 bis 2028 wird eine durchschnittliche jährliche Wachstumsrate (CAGR) von 34,8 % erwartet.
Analystenperspektive
Die wichtigsten Wachstumsfaktoren des Biosimilars-Marktes sind die zunehmende Zahl chronischer Erkrankungen wie Krebs. Die zunehmende Krebsbelastung und die damit verbundenen Todesfälle schaffen den Bedarf an erschwinglichen Behandlungen und fördern somit das Wachstum des Biosimilars-Marktes. Die wichtigsten Marktteilnehmer erwarten im Prognosezeitraum zudem Marktwachstum durch verschiedene strategische Aktivitäten wie Produkteinführungen, Fusionen und Übernahmen. Die zunehmende Prävalenz von Autoimmunerkrankungen wie Morbus Bechterew und rheumatoider Arthritis treibt das Wachstum des Biosimilars-Marktes voran. Laut einem 2020 im „Scandinavian Journal of Rheumatology“ veröffentlichten Artikel mit dem Titel „Prävalenz der ankylosierenden Spondylitis in Spanien“ wiesen beispielsweise etwa 7,3 % der Bevölkerung ein positives Screening auf Morbus Bechterew auf. Biosimilars wie Infliximab-axxq (Avsola), Infliximab-qbtx (Ixifi), Infliximab-dyyb (Inflectra) und Infliximab-abda (Renflexis) werden zur Behandlung chronischer Schmerzen bei Arthritis eingesetzt.
Marktübersicht
Ein Biosimilar ist ein biologisches Arzneimittel, das einem anderen, bereits zugelassenen biologischen Arzneimittel (dem „Referenzarzneimittel“) sehr ähnlich ist. Biosimilars werden nach denselben Standards für pharmazeutische Qualität, Sicherheit und Wirksamkeit zugelassen, die für alle biologischen Arzneimittel gelten. Biosimilars sind sichere und wirksame Behandlungsoptionen für viele Krankheiten, wie chronische Haut- und Darmerkrankungen (wie Psoriasis, Reizdarmsyndrom, Morbus Crohn und Colitis), Arthritis, Nierenerkrankungen und Krebs. Biosimilars verbessern den Zugang zu lebensrettenden Medikamenten zu potenziell geringeren Kosten. Die Haupttreiber des Biosimilars-Marktes sind die zunehmende Zahl chronischer Krankheiten.
Passen Sie diesen Bericht Ihren Anforderungen an
Sie erhalten kostenlos Anpassungen an jedem Bericht, einschließlich Teilen dieses Berichts oder einer Analyse auf Länderebene, eines Excel-Datenpakets sowie tolle Angebote und Rabatte für Start-ups und Universitäten.
Biosimilars-Markt: Strategische Einblicke
-
Holen Sie sich die wichtigsten Markttrends aus diesem Bericht.Dieses KOSTENLOSE Beispiel umfasst Datenanalysen, die von Markttrends bis hin zu Schätzungen und Prognosen reichen.
Markttreiber
Steigende Zulassungen von Biosimilars treiben das Wachstum des globalen Biosimilar-Marktes voran
Die Food and Drug Administration (FDA) genehmigt Biosimilar-Produkte und bietet die wissenschaftliche und regulatorische Beratung, die erforderlich ist, um sichere und wirksame Biosimilars auf den Markt zu bringen. Die Zulassung von Biosimilar-Produkten kann die Patientenversorgung verbessern, indem sie die Anzahl der Medikamentenoptionen zu potenziell geringeren Kosten erhöht.
Einige kürzlich erfolgte Zulassungen von Biosimilar-Produkten sind in der folgenden Tabelle aufgeführt.
Name des Biosimilars |
Zulassungsdatum |
Referenzprodukt |
Alymsys (Bevacizumab-Maly) |
April 2022 |
Avastin (Bevacizumab) |
Cimerli (Ranibizumab-Eqrn) |
August 2022 |
Lucentis (Ranibizumab) |
Fylnetra (Pegfilgrastim-pbbk) |
Mai 2022 |
Neulasta (pegfilgrastim) |
Stimufend (pegfilgrastim-fpgk) |
September 2022 |
Neulasta(pegfilgrastim) |
Vegzelma (Bevacizumab-adcd) |
September 2022 |
Avastin (Bevacizumab) |
Idacio (Adalimumab-aacf) |
Dezember 2022 |
Humira (Adalimumab) |
Byooviz (Ranibizumab-nuna) |
September 2021 |
Lucentis (Ranibizumab) |
Rezvoglar (Insulin glargine-aglr) |
Dezember 2021 |
Lantus (Insulin glargin) |
Semglee (Insulin glargin-yfgn) |
Juli 2021 |
Lantus (Insulin glargin) |
Yusimry (adalimumab-aqvh) |
Dezember 2021 |
Humira (adalimumab) |
Hulio (adalimumab-fkjp) |
Juli 2020 |
Humira (Adalimumab) |
Riabni (Rituximab-arrx) |
Dezember 2020 |
Rituxan (Rituximab) |
Nyvepria (Pegfilgrastim-apgf) |
Juni 2020 |
Neulasta (Pegfilgrastim) |
Die steigenden Zulassungen von Biosimilars treiben das Wachstum des Biosimilars-Marktes voran.
Segmentanalyse
Basierend auf der Krankheitsindikation ist der Biosimilars-Markt in Krebs, Diabetes, Autoimmunerkrankungen und andere Krankheitsindikationen. Das Segment Krebs hatte 2021 den größten Marktanteil und Autoimmunerkrankungen werden im Prognosezeitraum (2022–2028) voraussichtlich die höchste CAGR von 36,1 % verzeichnen. Basierend auf der Medikamentenklasse ist der Biosimilars-Markt in Granulozyten-Kolonie-stimulierende Faktoren, menschliches Wachstumshormon, Insulin, TNF-Blocker und monoklonale Antikörper sowie Sonstige (Osteoporose usw.) segmentiert. Das Medikamentensegment der Granulozyten-Kolonie-stimulierenden Faktoren hatte 2021 den größten Marktanteil. Darüber hinaus wird für das Segment der sonstigen Medikamentenklassen im Prognosezeitraum das höchste CAGR-Wachstum erwartet. Basierend auf der Anwendung ist der globale Biosimilars-Markt in intravenöse, subkutane und sonstige Anwendungen unterteilt. Das intravenöse Segment hatte 2021 den größten Marktanteil und wird im Prognosezeitraum voraussichtlich das höchste CAGR-Wachstum erzielen. Der Biosimilars-Markt ist nach Endverbraucher in Krankenhäuser, Fachkliniken, häusliche Pflege und andere Endverbraucher unterteilt. Das Krankenhaussegment hatte 2021 den größten Marktanteil, und das Segment häusliche Pflege wird im Prognosezeitraum (2022–2028) voraussichtlich die höchste durchschnittliche jährliche Wachstumsrate (CAGR) von 36,6 % verzeichnen.
Regionale Analyse
Der nordamerikanische Biosimilars-Markt wurde 2021 auf 5.479,84 Millionen US-Dollar geschätzt und soll bis 2028 47.746,80 Millionen US-Dollar erreichen; im Prognosezeitraum wird mit einer CAGR von 37,3 % gerechnet. Der nordamerikanische Biosimilars-Markt ist in die USA, Kanada und Mexiko unterteilt. Die USA hatten 2019 den größten Anteil am nordamerikanischen Biosimilars-Markt. Zunehmende Fälle von Diabetes und Unfruchtbarkeit sowie eine steigende Produktentwicklung auf dem Biosimilars-Markt. Laut dem NIH Autoimmune Diseases Coordinating Committee litten 2019 mehr als 24 Millionen Amerikaner an Autoimmunerkrankungen. Acht Millionen Menschen haben Autoantikörper. Das sind Blutmoleküle, die das Risiko einer Person anzeigen, an Autoimmunerkrankungen zu erkranken. Aus unbekannten Gründen sind immer mehr Menschen von Autoimmunerkrankungen betroffen. Laut der Clinical Research Branch des National Institute of Environmental Health Sciences (NIEHS) ist im Jahr 2020 ein signifikanter Anstieg der Prävalenz antinukleärer Antikörper (ANA) zu verzeichnen, des häufigsten Biomarkers für Autoimmunität in den USA. Die Studie ist die erste, die ANA-Veränderungen in einer repräsentativen Stichprobe der US-Bevölkerung im Zeitverlauf untersucht. Sie umfasst Männer, nicht-hispanische Weiße, Erwachsene über 50 und Jugendliche. In den USA werden Biosimilars zur Behandlung von Patienten mit Krebs, Nierenerkrankungen, Diabetes und anderen Autoimmunerkrankungen wie rheumatoider Arthritis und Morbus Crohn eingesetzt. Laut Cardinal Health wurden in den USA insgesamt 33 Biosimilars von der FDA zugelassen und 21 sind im Handel erhältlich. Von den 21 auf dem Markt befindlichen Biosimilars werden 17 zur Behandlung von Krebserkrankungen eingesetzt, drei zur Behandlung von Autoimmunerkrankungen und eines zur Behandlung von Diabetes.
Biologika sind in den USA die teuersten Medikamente; die Kosten belaufen sich pro Patient und Jahr auf mehrere zehntausend Dollar. Biosimilars werden voraussichtlich 15 bis 30 Prozent günstiger sein als ihr Referenzprodukt. Allein im Jahr 2020 wurden durch Biosimilars 7,9 Milliarden US-Dollar eingespart, und diese Einsparungen werden in den nächsten Jahren voraussichtlich deutlich steigen, da mehr Biosimilars auf den Markt kommen. Laut Cardinal Health wird erwartet, dass Biosimilars die Arzneimittelausgaben in den USA bis 2025 um 133 Milliarden US-Dollar senken werden. In den USA haben Biosimilars also ein enormes Potenzial, die Kosten für biologische Arzneimittel zu senken, die Versorgung für Patienten zugänglicher zu machen und neue Innovationen und wissenschaftliche Durchbrüche hervorzubringen, wodurch das Wachstum des Biosimilar-Marktes in dieser Region vorangetrieben wird.
Analyse der wichtigsten Akteure
Die Analyse des Biosimilar-Marktes umfasst Akteure wie Amgen Inc, Celltrion Inc, Sanofi SA, Biocon Ltd, Pfizer Inc, Samsung Bioepis Co Ltd, Coherus BioSciences Inc, Eli Lilly and Co, Sandoz AG, Teva Pharmaceutical Industries Ltd und Dr. Reddy's Laboratories Ltd. Unter den Akteuren auf dem Biosimilar-Markt sind Pfizer Inc. und Novartis, Inc aufgrund ihres diversifizierten Produktportfolios die beiden größten Akteure.
Biosimilars
Regionale Einblicke in den Biosimilars-MarktDie Analysten von The Insight Partners haben die regionalen Trends und Faktoren, die den Biosimilars-Markt im Prognosezeitraum beeinflussen, ausführlich erläutert. In diesem Abschnitt werden auch die Marktsegmente und die geografische Verteilung von Biosimilars in Nordamerika, Europa, dem asiatisch-pazifischen Raum, dem Nahen Osten und Afrika sowie Süd- und Mittelamerika erörtert.
Umfang des Biosimilars-Marktberichts
| Berichtsattribut | Einzelheiten |
|---|---|
| Marktgröße in 2021 | US$ 18.44 Billion |
| Marktgröße nach 2028 | US$ 136.07 Billion |
| Globale CAGR (2021 - 2028) | 34.8% |
| Historische Daten | 2019-2020 |
| Prognosezeitraum | 2022-2028 |
| Abgedeckte Segmente |
By Krankheitsindikation
|
| Abgedeckte Regionen und Länder |
Nordamerika
|
| Marktführer und wichtige Unternehmensprofile |
|
Dichte der Marktakteure für Biosimilars: Auswirkungen auf die Geschäftsdynamik verstehen
Der Markt für Biosimilars wächst rasant. Die steigende Nachfrage der Endverbraucher ist auf Faktoren wie veränderte Verbraucherpräferenzen, technologische Fortschritte und ein stärkeres Bewusstsein für die Produktvorteile zurückzuführen. Mit der steigenden Nachfrage erweitern Unternehmen ihr Angebot, entwickeln Innovationen, um den Bedürfnissen der Verbraucher gerecht zu werden, und nutzen neue Trends, was das Marktwachstum weiter ankurbelt.
- Holen Sie sich die Biosimilars-Markt Übersicht der wichtigsten Akteure
Anorganische und organische Strategien wie Fusionen und Übernahmen sowie Produkteinführungen werden von Unternehmen im Biosimilars-Markt stark genutzt. Nachfolgend sind einige wichtige Marktentwicklungen der letzten Zeit aufgeführt:
- Im Januar 2022 schloss Biocon Biologics, eine Tochtergesellschaft von Biocon Ltd., die Übernahme des globalen Biosimilars-Geschäfts von Viatris ab. Durch die Übernahme erhält Biocon Biologics direkte kommerzielle Kapazitäten und eine unterstützende Infrastruktur in den fortgeschrittenen Märkten und mehreren Schwellenmärkten und rückt so näher an Patienten, Kunden und Kostenträger heran. Mit dieser Übernahme wird Biocon Biologics zu einem weltweit führenden Biosimilars-Anbieter mit acht kommerzialisierten Produkten.
- Im Oktober 2022 lizenziert Biocon Biologics zwei Biosimilar-Produkte an Yoshindo zur Vermarktung in Japan. Im Rahmen dieser Vereinbarung erhält Yoshindo die exklusiven Vermarktungsrechte in Japan für bUstekinumab und bDenosumab, die von Biocon Biologics entwickelt und hergestellt werden, und damit ein adressierbares Marktpotenzial von 700 Millionen US-Dollar.
- Im Dezember 2022 gab Celltrion USA die Einreichung des Biologics License Application (BLA) für die neuartige subkutane Formulierung von CT-P13 bei der FDA bekannt. Eine subkutane Formulierung hat das Potenzial, die Behandlungsmöglichkeiten für die Verwendung des Medikaments Infliximab zu verbessern, indem sie eine hohe Konsistenz der Medikamentenexposition und eine bequeme Verabreichungsmethode bietet.
- Im September 2022 erhielt Celltrion USA die US-amerikanische FDA-Zulassung für sein onkologisches Biosimilar Vegzelma zur Behandlung von sechs Krebsarten wie metastasiertem Dickdarmkrebs, rezidivierendem oder metastasiertem nicht-plattenepithelialem nicht-kleinzelligem Lungenkrebs (nsNSCLC), rezidivierendem Glioblastom, metastasiertem Nierenzellkarzinom, persistierendem, rezidivierendem oder metastasiertem Gebärmutterhalskrebs; und epithelialem Eierstockkrebs, Eileiterkrebs oder primärem Peritonealkrebs. Vegzelma ist Celltrions drittes onkologisches Biosimilar, das die Zulassung der US-amerikanischen FDA erhalten hat.
- Im Mai 2022 bringen Biocon Biologics und Viatris Abevmy auf den Markt. Biocon Biologics Ltd., eine Tochtergesellschaft von Biocon Ltd., und Viatris Inc. gaben bekannt, dass Abevmy (bBevacizumab) in Kanada erhältlich ist. Abevmy, gemeinsam von Biocon Biologics und Viatris entwickelt, ist ein Biosimilar zu Avastin (Bevacizumab) von Roche und wurde von Health Canada für vier onkologische Indikationen zugelassen.
- Historische Analyse (2 Jahre), Basisjahr, Prognose (7 Jahre) mit CAGR
- PEST- und SWOT-Analyse
- Marktgröße Wert/Volumen – Global, Regional, Land
- Branchen- und Wettbewerbslandschaft
- Excel-Datensatz
Aktuelle Berichte
Verwandte Berichte
Erfahrungsberichte
Grund zum Kauf
- Fundierte Entscheidungsfindung
- Marktdynamik verstehen
- Wettbewerbsanalyse
- Kundeneinblicke
- Marktprognosen
- Risikominimierung
- Strategische Planung
- Investitionsbegründung
- Identifizierung neuer Märkte
- Verbesserung von Marketingstrategien
- Steigerung der Betriebseffizienz
- Anpassung an regulatorische Trends






















 Kostenlose Probe anfordern für - Biosimilars-Markt
Kostenlose Probe anfordern für - Biosimilars-Markt