Analytical Laboratory Services Market Demand, Share & Growth by 2034
Analytical Laboratory Services Market Size and Forecasts (2021 - 2034), Global and Regional Share, Trends, and Growth Opportunity Analysis Report Coverage: By Type (Bioanalytical Testing, Batch Release Testing, Stability Testing, Raw Material Testing, Physical Characterization, Method Validation, Microbial Testing, Environmental Monitoring); End User (Government, University and Research Institution, Hospital, Others), and Geography (North America, Europe, Asia Pacific, and South and Central America)
Historic Data: 2021-2024 | Base Year: 2025 | Forecast Period: 2026-2034- Status : Data Released
- Report Code : TIPRE00017615
- Category : Life Sciences
- No. of Pages : 150
- Available Report Formats :


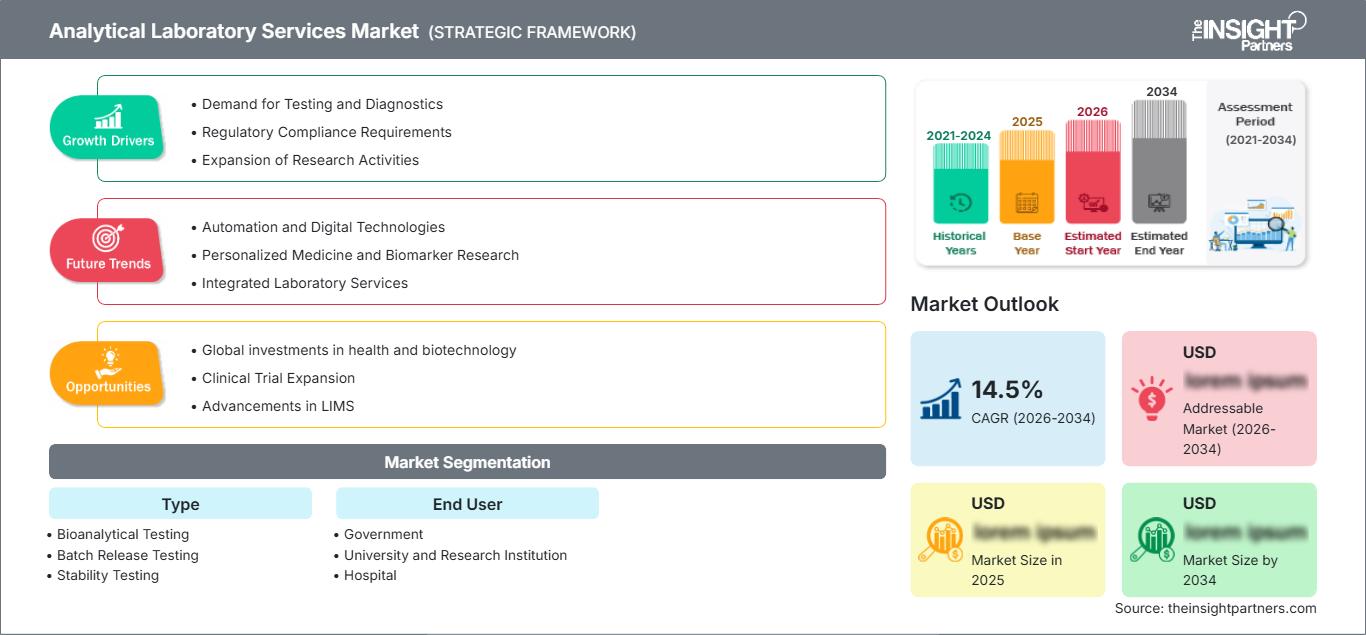
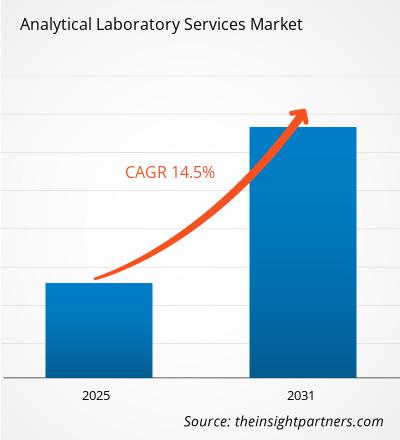
The global Analytical Laboratory Services Market size is projected to reach US$ 1505.22 million by 2034 from US$ 405.22 Million in 2025. The market is anticipated to register a CAGR of 16.04% during the forecast period 2026–2034
Key market dynamics include the intensifying regulatory scrutiny across the pharmaceutical and medical device sectors, the rapid proliferation of biologics and biosimilars requiring complex characterization, and a growing trend toward outsourcing non-core R&D activities to specialized contract laboratories. Additionally, the market is expected to benefit from the rise in global healthcare expenditure, advancements in high-throughput screening technologies, and the increasing necessity for rigorous environmental and safety testing in emerging industrial hubs.
Analytical Laboratory Services Market Analysis
The analytical laboratory services market analysis shows a strategic pivot toward specialized high-end testing as manufacturers seek to mitigate the risks associated with drug failure and regulatory non-compliance. Procurement trends indicate that pharmaceutical giants are increasingly favoring long-term strategic partnerships with one-stop-shop laboratories that offer integrated suites from raw material testing to batch release. Strategic opportunities are emerging in the cell and gene therapy (CGT) space, where traditional analytical methods are insufficient, creating a high-margin niche for labs with advanced molecular diagnostic and viral clearance capabilities. The analysis also notes that market expansion is contingent upon the integration of Laboratory Information Management Systems (LIMS) and AI-driven data analytics to ensure data integrity and faster turnaround times. Competitive differentiation is now defined by a provider's ability to offer audit-ready transparency and specialized expertise in navigating diverse international regulatory frameworks such as the FDA, EMA, and NMPA.
Analytical Laboratory Services Market Overview
The analytical laboratory services market is transitioning from a decentralized support function to a critical pillar of the global life sciences supply chain. While historically focused on routine compendial testing, the market is expanding into sophisticated bioanalytical services, including pharmacokinetic (PK) and pharmacodynamic (PD) modeling for complex large molecules. Both large-scale international contract research organizations (CROs) and boutique regional laboratories are adapting to a landscape where precision and speed are paramount. The rising prevalence of chronic diseases and the subsequent surge in clinical trial volumes have bolstered the demand for specialized bioassays and microbial testing. While North America and Europe remain the primary hubs for high-value innovation, Asia-Pacific region is rapidly emerging as a preferred destination for cost-effective, high-volume analytical outsourcing, supported by significant government investments in biotechnology parks and research infrastructure. For instance, the market in the US represents a highly mature and technologically advanced landscape within the global analytical laboratory market. The market is characterized by a dense concentration of biopharmaceutical innovation hubs and a stringent regulatory environment that necessitates exhaustive validation and testing protocols. Growth is primarily sustained by the continuous flow of novel drug candidates and a robust domestic healthcare infrastructure that prioritizes diagnostic accuracy and patient safety.
Customize This Report To Suit Your Requirement
Get FREE CUSTOMIZATIONAnalytical Laboratory Services Market: Strategic Insights
-
Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
Analytical Laboratory Services Market Drivers and Opportunities
Market Drivers:
- Stringent Regulatory Standards and Compliance: Global health authorities are continuously updating guidelines for drug safety and quality. The mandatory requirement for detailed stability testing and impurity profiling under ICH guidelines compels companies to utilize professional analytical services to ensure market authorization.
- Increasing Outsourcing by Pharmaceutical and Biotech Firms: To optimize operational costs and focus on core drug discovery, many companies are shifting their internal testing loads to third-party providers. This trend is particularly strong among small-to-mid-sized biotech firms that lack the capital to invest in expensive analytical instrumentation.
- Rise of Biologics and Biosimilars: Unlike small molecules, biologics are highly complex and sensitive to manufacturing changes. This has led to an unprecedented demand for advanced physical characterization and bioanalytical testing to prove biosimilarity and ensure batch-to-batch consistency.
Market Opportunities:
- Advancements in Personalized Medicine: The shift toward targeted therapies creates significant opportunities for labs to develop and offer specialized companion diagnostic assays and genomic testing services that require high levels of analytical precision.
- Integration of Artificial Intelligence and Automation: There is a substantial opportunity for service providers to adopt AI-driven predictive modeling for stability testing and automated sample processing. These technologies can reduce human error, lower costs, and significantly shorten the time-to-market for new products.
- Expansion into Environmental and Food Safety Testing: Beyond healthcare, analytical labs can diversify into monitoring chemical contaminants and microbial pathogens in the food supply chain and environment, driven by rising consumer awareness and stricter global safety regulations.
Analytical Laboratory Services Market Report Segmentation Analysis
The Analytical Laboratory Services Market share is analyzed across various segments to provide a clearer understanding of its structure, growth potential, and emerging trends. Below is the standard segmentation approach used in most industry reports:
By Type:
- Bioanalytical Testing: Focuses on the quantitative measurement of drugs and their metabolites in biological matrices, playing a crucial role in clinical and preclinical trials.
- Batch Release Testing: A vital final step in the manufacturing process, ensuring every product batch meets the specified quality standards before entering the market.
- Stability Testing: Evaluates how the quality of a drug substance or product varies with time under the influence of environmental factors like temperature and humidity.
- Raw Material Testing: Ensures that all ingredients and components used in the production process meet the necessary purity and identity specifications.
- Physical Characterization: Involves the analysis of the physical properties of substances, such as particle size and surface area, which are critical for drug formulation.
- Method Validation: The process of proving that an analytical procedure is suitable for its intended use, a mandatory requirement for regulatory submissions.
- Microbial Testing: Used to detect and identify microbial contaminants in products, ensuring they are sterile and safe for human consumption.
- Environmental Monitoring: Involves the continuous assessment of cleanrooms and production areas to prevent contamination during the manufacturing process.
By End User:
- Government: Public health agencies and regulatory bodies that utilize analytical services for surveillance, standard-setting, and public safety monitoring.
- University and Research Institution: Academic centers that require high-precision testing to support basic research, drug discovery, and early-stage development projects.
- Hospital: Medical facilities that utilize analytical labs for specialized patient diagnostics, therapeutic drug monitoring, and clinical research support.
- Others: Includes pharmaceutical and biopharmaceutical companies, medical device manufacturers, and forensic laboratories.
By Geography:
- North America
- Europe
- Asia Pacific
- South & Central America
- Middle East & Africa
Analytical Laboratory Services Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2025 | US$ 405.22 Million |
| Market Size by 2026-2034 | US$ 1505.22 Million |
| Global CAGR (2026 - 2034) | 16.04% |
| Historical Data | 2021-2024 |
| Forecast period | 2026-2034 |
| Segments Covered |
By Type
|
| Regions and Countries Covered |
North America
|
| Market leaders and key company profiles |
|
Analytical Laboratory Services Market Players Density: Understanding Its Impact on Business Dynamics
The Analytical Laboratory Services Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
Analytical Laboratory Services Market Share Analysis by Geography
North America is expected to maintain its dominant position in the coming years due to its robust R&D ecosystem. Emerging markets in Asia-Pacific and the Middle East also have many untapped opportunities for laboratory service providers to expand as local manufacturing capabilities for generics and vaccines continue to scale.
The analytical laboratory services market is undergoing a significant transformation, moving from traditional routine testing to high-complexity molecular and bioanalytical analysis. Growth is driven by the rise in clinical trials, the complexity of modern therapeutic modalities, and the global push for standardized quality control. Below is a summary of market share and trends by region:
North America
- Market Share: Holds the largest global share, supported by the presence of major pharmaceutical headquarters and a sophisticated network of independent contract laboratories.
- Key Drivers:
- High concentration of biotechnology and medical device innovators.
- Strict FDA oversight requiring comprehensive analytical validation.
- Growing adoption of personalized medicine and molecular diagnostics.
- Trends: Increasing investment in automated laboratory workflows and the expansion of specialized testing facilities for cell and gene therapy products.
Europe
- Market Share: A major regional player characterized by a strong emphasis on quality assurance and a well-established regulatory framework under the EMA.
- Key Drivers:
- Expansion of the biosimilars market in countries like Germany and France.
- High demand for environmental monitoring and food safety analysis.
- Robust government funding for clinical research and healthcare infrastructure.
- Trends: A move toward Green Chemistry in laboratory practices and the rising consolidation of smaller labs into large-scale pan-European service providers.
Asia-Pacific
- Market Share: The fastest-growing region, increasingly serving as a global hub for outsourced analytical services, particularly in India and China.
- Key Drivers:
- Rapid growth of the generic drug manufacturing industry.
- Government incentives to establish world-class biotechnology parks.
- An increasing number of clinical trials are being conducted in emerging Asian economies.
- Trends: Heavy adoption of cloud-based laboratory management systems and a strategic focus on achieving international laboratory accreditation (e.g., ISO/IEC 17025).
South and Central America
- Market Share: An emerging market with a growing focus on improving local pharmaceutical manufacturing standards in Brazil and Argentina.
- Key Drivers:
- Rising healthcare awareness and government efforts to improve drug accessibility.
- Modernization of local laboratories to meet international export standards.
- Increasing prevalence of infectious and chronic diseases requiring diagnostic support.
- Trends: Expansion of public-private partnerships to enhance analytical testing capabilities for regional vaccine production.
Middle East and Africa
- Market Share: A developing market with a focus on reducing import reliance and establishing domestic quality control infrastructure.
- Key Drivers:
- Strategic investments in Smart Labs and advanced diagnostic centers in the GCC region.
- Growing demand for shelf-stable pharmaceutical products in diverse climates.
- Rising government focus on food security and environmental safety standards.
- Trends: Implementation of mobile testing units for rural areas and the formalization of national laboratory networks to support public health surveillance.
High Market Density and Competition
Competition is intensifying due to the presence of established leaders such as Eurofins Scientific, Labcorp (Laboratory Corporation of America), and SGS SA. Regional experts and niche players like Charles River Laboratories, Intertek Group, and WuXi AppTec, alongside innovators such as PPD (Thermo Fisher Scientific), contribute to a highly competitive and technically demanding market landscape.
This competitive environment pushes vendors to differentiate through:
- Technological Superiority: Investing in next-generation sequencing (NGS) and high-resolution mass spectrometry to handle the most complex biological samples.
- Global Footprint and Scalability: Maintaining a network of accredited labs worldwide to provide consistent service to multinational clients.
- Regulatory Expertise: Offering consultative services that help clients navigate the complexities of global regulatory filings and data integrity requirements.
- Strategic Outsourcing Models: Providing flexible service agreements, ranging from fee-for-service to full-time equivalent (FTE) models, to suit various client budgets.
Opportunities and Strategic Moves
- Strategic Mergers and Acquisitions: Acquiring boutique labs with specialized capabilities in high-growth areas like toxicology or genomics to provide a comprehensive service portfolio.
- Digital Transformation: Implementing end-to-end digital platforms that allow clients real-time access to their testing data, enhancing transparency and project management.
Major Companies operating in the Analytical Laboratory Services Market are:
- Agenzia Italiana del Farmaco
- Central Drugs Standard Control Organization
- China Food and Drug Administration
- European Medicines Agency
- Federal Institute for Drugs and Medical Devices
- Pharmaceuticals and Medical Devices Agency
- The Spanish Medicines and Health Products Agency
- U.S. Food and Drug Administration
- Agence francaise de securite sanitaire des produits de sante
- SOLVIAS AG
Disclaimer: The companies listed above are not ranked in any particular order.
Analytical Laboratory Services Market News and Recent Developments
- In November 2025, Labcorp, a global leader in innovative and comprehensive laboratory services, and Parkview Health, one of the fastest-growing health systems in the Midwest, announced a strategic agreement for Labcorp to acquire select assets of the health system's outreach analytical laboratory services that provided high-quality testing across Indiana and northwest Ohio.
- In October 2024, Eurofins Scientific reached an agreement with SYNLAB to acquire its clinical diagnostics operations in Spain, a move that significantly expanded Eurofins' footprint in highly specialized analytical laboratory services. The transaction, which remained subject to customary regulatory approvals, was expected to close in 2025, further strengthening the company's regional presence in molecular clinical diagnostics and in vitro diagnostic products.
Analytical Laboratory Services Market Report Coverage and Deliverables
The Analytical Laboratory Services Market Size and Forecast (2021–2034) report provides a detailed analysis of the market covering below areas:
- Analytical Laboratory Services Market size and forecast at global, regional, and country levels for all the key market segments covered under the scope
- Analytical Laboratory Services Market trends, as well as market dynamics such as drivers, restraints, and key opportunities
- Detailed PEST and SWOT analysis
- Analytical Laboratory Services Market analysis covering key market trends, global and regional framework, major players, regulations, and recent market developments
- Industry landscape and competition analysis covering market concentration, heat map analysis, prominent players, and recent developments in the Analytical Laboratory Services Market.
- Detailed company profiles
Frequently Asked Questions
Mrinal is a seasoned research analyst with over 8 years of experience in Life Sciences Market Intelligence and Consulting. With a strategic mindset and unwavering commitment to excellence, she has built deep expertise in pharmaceutical forecasting, market opportunity assessment, and developing industry benchmarks. Her work is anchored in delivering actionable insights that empower clients to make informed strategic decisions.
Mrinal’s core strength lies in translating complex quantitative datasets into meaningful business intelligence. Her analytical acumen is instrumental in shaping go-to-market (GTM) strategies and uncovering growth opportunities across the pharmaceutical and medical device sectors. As a trusted consultant, she consistently focuses on streamlining workflow processes and establishing best practices, thereby driving innovation and operational efficiency for her clients.
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Global, Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Recent Reports
Testimonials
The Insight Partners' SCADA System Market report is comprehensive, with valuable insights on current trends and future forecasts. The team was highly professional, responsive, and supportive throughout. We are very satisfied and highly recommend their services.
RAN KEDEM Partner, Reali Technologies LTDsI requested a report on a very specific software market and the team produced the report in a few days. The information was very relevant and well presented. I then requested some changes and additions to the report. The team was again very responsive and I got the final report in less than a week.
JEAN-HERVE JENN Chairman, Future AnalyticaWe worked with The Insight Partners for an important market study and forecast. They gave us clear insights into opportunities and risks, which helped shape our plans. Their research was easy to use and based on solid data. It helped us make smart, confident decisions. We highly recommend them.
PIYUSH NAGPAL Sr. Vice President, High Beam GlobalThe Insight Partners delivered insightful, well-structured market research with strong domain expertise. Their team was professional and responsive throughout. The user-friendly website made accessing industry reports seamless. We highly recommend them for reliable, high-quality research services
YUKIHIKO ADACHI CEO, Deep Blue, LLC.This is the first time I have purchased a market report from The Insight Partners.While I was unsure at first, I visited their web site and felt more comfortable to take the risk and purchase a market report.I am completely satisfied with the quality of the report and customer service. I had several questions and comments with the initial report, but after a couple of dialogs over email with their analyst I believe I have a report that I can use as input to our strategic planning process.Thank you so much for taking the extra time and making this a positive experience.I will definitely recommend your service to others and you will be my first call when we need further market data.
JOHN SUZUKI President and Chief Executive Officer, Board Director, BK TechnologiesI wish to appreciate your support and the professionalism you displayed in the course of attending to my request for information regarding to infectious disease IVD market in Nigeria. I appreciate your patience, your guidance, and the fact that you were willing to offer a discount, which eventually made it possible for us to close a deal. I look forward to engaging The Insight Partners in the future, all thanks to the impression you have created in me as a result of this first encounter.
DR CHIJIOKE ONYIA MANAGING DIRECTOR, PineCrest Healthcare Ltd.Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends





 Get Free Sample For
Get Free Sample For