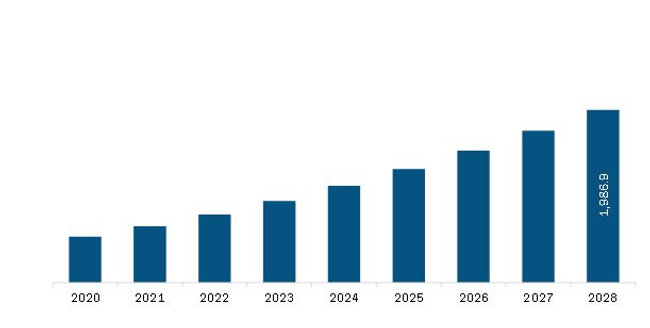
The Europe 3D printing medical devices market is expected to reach US$ 1,986.9 million by 2028 from US$ 642.6 million in 2021. The market is estimated to grow at a CAGR of 17.5% from 2021–2028.
3D printers are used to manufacture a variety of medical devices, including those with complex geometry or features that match a patient’s unique anatomy. A few devices are printed from a standard design, and then multiple identical copies of the same device are made. Other devices, called patient-matched or patient-specific devices, are created from the patient-specific imaging data. The choice of technology used for 3D printing depends on many factors, including the intended use of printed products and the simplicity of the printer, among others. Powder bed fusion is the most common technology used for the 3D printing of medical devices. This technique is compatible with various materials used in medical devices, such as titanium and nylon. With 3D printing, creating patient-specific, tactile reference models from CT and MRI scans is both affordable and simple. These models provide an additional perspective that helps physicians better prepare for surgeries, resulting in drastically reduced time and cost of an actual procedure performed within an operating room. This may benefit patients through greater satisfaction, lowered anxiety, and shortened recovery time. In addition to this, the launch of new biocompatible medical 3D printing materials helps in the development of new surgical tools and techniques with the ultimate goal of improving the clinical experience during surgery. The tools that can be printed using 3D printing techniques include sterilizable fixation trays, contouring templates, and implant sizing models, which can be used to size implants in the operating room before the first cut, allowing surgeons to save time and improve accuracy during complex procedures.
Due to the coronavirus outbreak, the European Commission decided to push back the transition from MDD to MDR. This permits the medical device sector and Notified Bodies (NBs) to concentrate on the current issue and devote all of their resources to combatting the coronavirus. However, there has been no indication on whether the In Vitro Diagnostic Regulation (IVDR) 2017/246, which is set to take effect in May 2021 would be postponed. This adds to the strain on IVD makers, who must stay on track with the improvements required to move to IVDR. They must also deal with the increased demand for key diagnostic tests as well as the difficulty of kit manufacture and distribution during the pandemic. The European Commission has temporarily made the export of certain medical gadgets, such as personal protective equipment (PPE) and other similar protective gear, subject to authorization. Medical device companies that are contractually obligated to supply devices to nations outside of the EU may find this problematic. Norway, Lichtenstein, and Switzerland are exempt from this authorization.
With the new features and technologies, vendors can attract new customers and expand their footprints in emerging markets. This factor is likely to drive the 3D printing medical devices market. The Europe 3D printing medical devices market is expected to grow at a good CAGR during the forecast period.
Europe 3D Printing Medical Devices Market Revenue and Forecast to 2028 (US$ Million)
- This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
Europe 3D Printing Medical Devices Market Segmentation
By Component
- Software and Services
- Equipment
- 3D Printers
- 3D Bioprinters
- Materials
- Plastics Material
- Metal and Metal Alloys
- Bioprinting Biomaterials
- Wax
- Others
By Technology
- Laser Beam Melting
- Direct Metal Laser Sintering
- Selective Laser Sintering
- Selective Laser Melting
- LaserCusing
- Photopolymerization
- Stereolithography
- Others
- Droplet Deposition/Extrusion Based Technologies
- Fused Deposition Modelling
- Multiphase Solidification
- Low Temperature Deposition Manufacturing
- Electron Beam melting
By Application
- Custom Prosthetics and Implants
- Craniomaxillofacial Implants
- Custom Dental Prosthetics and Implants
- Custom Orthopaedic Implants
- Surgical Guides
- Dental Guides
- Orthopaedic Guides
- Craniomaxillofacial Guides
- Spinal Guides
- Tissue Engineering Products
- Bone and Cartilage Scaffolds
- Ligaments and Tendons Scaffolds
- Surgical Instruments
- Surgical Fasteners
- Scalpels
- Retractors
- Hearing Aids
- Wearable Medical Devices
- Standard Prosthetic and Implants
- Others
By End-User
- Hospitals and Surgical Centres
- Dental and Orthopaedic Centres
- Medical Device Companies
- Pharmaceutical and Biotechnology Companies
- Academic and Research Institutes
- Others
By Country
- Europe
- Germany
- UK
- France
- Spain
- Italy
- Rest of Europe
Companies Mentioned
- EOS GmbH Electro Optical Systems
- Renishaw PLC
- Stratasys Ltd.
- 3D Systems, Inc.
- EnvisionTech, Inc.
- Concept Laser Gmbh (General Electric)
- Proadways Group
- SLM Solution Group AG
- CELLINK
- 3T RPD Ltd
Europe 3D Printing Medical Devices Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2021 | US$ 642.6 Million |
| Market Size by 2028 | US$ 1,986.9 Million |
| CAGR (2021 - 2028) | 17.5% |
| Historical Data | 2019-2020 |
| Forecast period | 2022-2028 |
| Segments Covered |
By Component
|
| Regions and Countries Covered |
Europe
|
| Market leaders and key company profiles |
|
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Recent Reports
Testimonials
Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends







 Get Free Sample For
Get Free Sample For