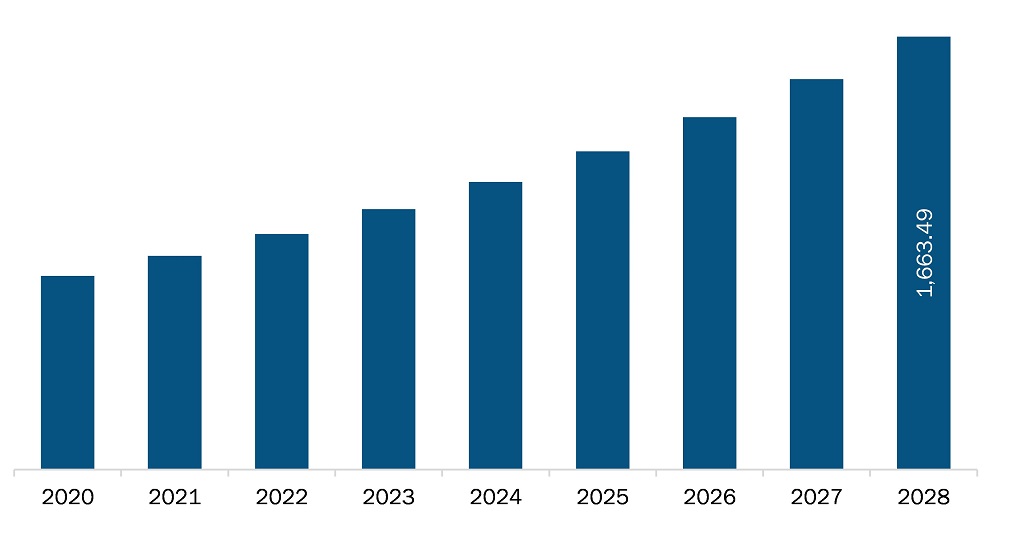
The cryopreservation equipment market in Asia Pacific is expected to grow from US$ 821.1 million in 2021 to US$ 1,663.5 million by 2028; it is estimated to grow at a CAGR of 10.6% from 2021 to 2028.
Cryopreservation has become an important aspect of the manufacturing process for many cellular therapies, as it occasionally comes before cell culture (by conserving the starting cellular material before moving forward with large-scale manufacturing) and generally follows cell expansion therapies. The ability to store cells at different points during the manufacturing process allows for the creation of a customized workflow. Cryopreservation can be employed in both types of transplant manufacturing processes—a patient-specific therapy, such as autologous therapy, and/or an allogeneic therapy. The efficacy of immune cell-based cancer therapies depends on the injection of highly viable cells after thawing; the injected cells can effectively detect and destroy target tumour cells. Adopting best practices during manufacturing such as the selection of GMP-grade or suitable cryoprotective agents (CPA) and controlled-rate freezing equipment with appropriate cooling profiles for immune cells helps generate high-quality cells. This guarantees the consistency of products and effective functioning of cells. The cryopreservation of cell-based therapeutic products also aids in quarantining donor cells and final products, allowing for more extensive microbiological testing. Coordinating and transporting the final product in accordance with the patient's treatment plan and extending the final product's shelf life prior to patient administration. Thus, the importance of cryopreservation in the successful delivery of cell-based therapies boosts the use of cryopreservation equipment.
Many measures have been implemented to contain the spread of COVID-19, that have resulted in significant operational disruption for many companies including in healthcare industry. Moreover, medical device companies are starting to forecast large sales declines in their Chinese markets because people are staying at home. Many big medical device companies such as Philips, GE Healthcare, and Hill-Rom amongst others have predicated the decline in sales in China. On the other hand, restricted access to family planning services, economic uncertainty and recession, and reduced work-life balance led to increasing number of egg and embryo freezing during the pandemic. Asia-Pacific region and beyond have taken unprecedented steps to curb the spread of the virus and provide care for the many infected individuals. Crucially, the impact of the pandemic on fertility is associated with huge uncertainty. Thus, the rising use of cryopreservation equipment during the pandemic is likely to pose a positive impact on the market.
With the new features and technologies, vendors can attract new customers and expand their footprints in emerging markets. This factor is likely to drive the cryopreservation equipment market. The Asia Pacific Cryopreservation equipment market is expected to grow at a good CAGR during the forecast period.
- This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
Asia Pacific Cryopreservation Equipment Market Segmentation
By Type
- Freezers
- Sample Preparation Systems
- Accessories
- Freezer Boxes
- Freezer Racks
- Cryopreservation Vials
- Others
By Cryogen Type
- Liquid Nitrogen
- Oxygen
- Liquid Helium
- Argon
- Others
By Application
- Cord Blood Stem Cells
- Sperms Semen and Testicular Tissues
- Embryos and Oocytes
- Cell and Gene Therapies
- Others
By End User
- Stem Cell Banks
- Biotechnology and Pharmaceutical Organizations
- Stem Cell Research Laboratories
- Others
By Country
- Asia Pacific
- China
- India
- Japan
- South Korea
- Australia
- South-eastern Asia
- Rest of Asia Pacific
Companies Mentioned
- Thermo Fisher Scientific Inc.
- Gold Sim Cellular Science Llc
- Brooks Automation, Inc
- Avantor, Inc.
- Hamilton Company
- PHC Holdings Corporation
- General Electric Company
- Cryoport Systems, LLC.
- Antech Group Inc.
- BioLife Solutions
- ZhongkeMeiling Cryogenics Company Limited
Asia Pacific Cryopreservation Equipment Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2021 | US$ 821.1 Million |
| Market Size by 2028 | US$ 1,663.5 Million |
| CAGR (2021 - 2028) | 10.6% |
| Historical Data | 2019-2020 |
| Forecast period | 2022-2028 |
| Segments Covered |
By Type
|
| Regions and Countries Covered |
Asia-Pacific
|
| Market leaders and key company profiles |
|
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Recent Reports
Testimonials
Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends







 Get Free Sample For
Get Free Sample For