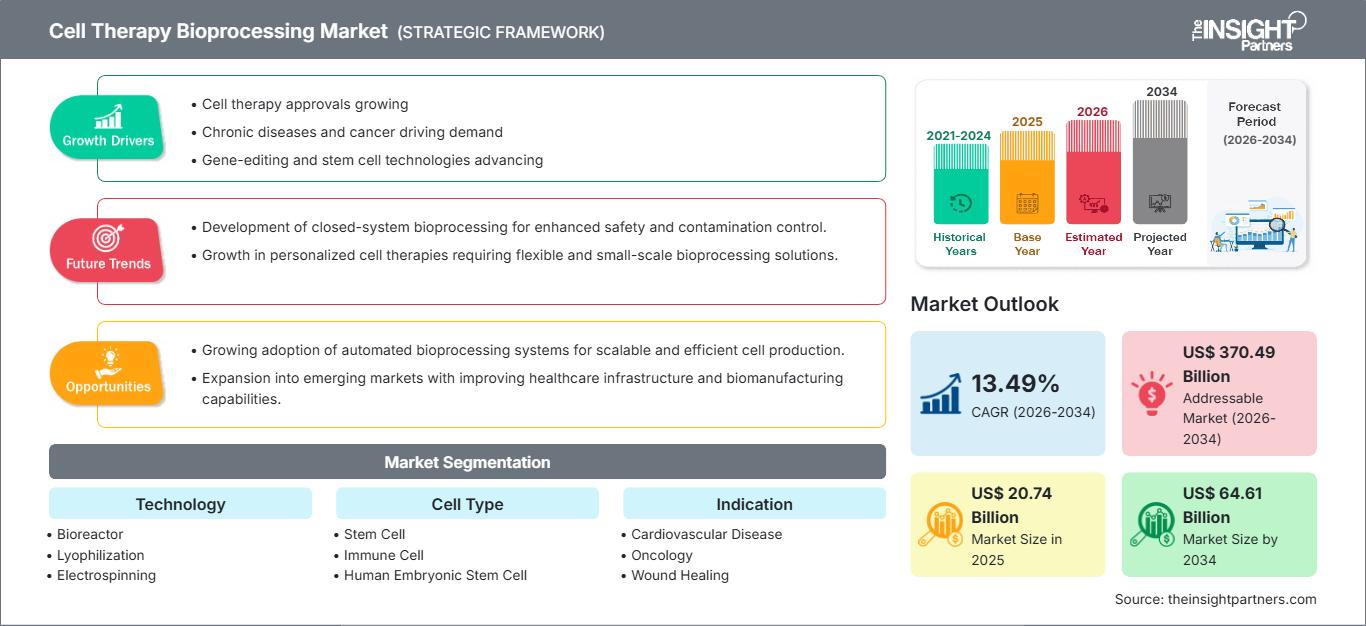
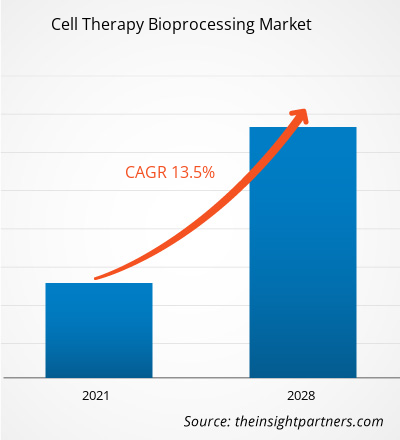
Der Markt für Zelltherapie-Bioprozesse wird bis 2034 voraussichtlich ein Volumen von 64,61 Milliarden US-Dollar erreichen, gegenüber 20,74 Milliarden US-Dollar im Jahr 2025. Es wird erwartet, dass der Markt im Prognosezeitraum 2026–2034 eine robuste durchschnittliche jährliche Wachstumsrate (CAGR) von 13,49 % verzeichnen wird.
Marktanalyse für Zelltherapie-Bioprozesse
Die Marktprognose für die Bioprozessierung in der Zelltherapie deutet auf ein robustes und vielschichtiges Wachstum hin, das primär durch den Übergang von Zell- und Gentherapien von Nischenbehandlungen hin zur breiten klinischen Anwendung getrieben wird. Diese Marktexpansion wird durch entscheidende technologische Fortschritte begünstigt, darunter die zunehmende Verwendung von Einweg-Bioreaktoren für mehr Flexibilität, die Integration von Automatisierung und Robotik zur Minimierung menschlicher Fehler und zur Skalierung der Produktion sowie der Einsatz KI-gestützter Analysen zur Echtzeit-Prozessüberwachung und -optimierung. Darüber hinaus beschleunigen steigende Investitionen in die regenerative Medizin in Verbindung mit einer verstärkten regulatorischen Unterstützung (wie beispielsweise Fast-Track-Zulassungen für neuartige Therapien) die Entwicklung therapeutischer Kandidaten durch klinische Studien und in die kommerzielle Produktion und stärken so die Marktentwicklung.
Marktübersicht Zelltherapie-Bioprozessierung
Die Bioprozessierung in der Zelltherapie umfasst den komplexen, durchgängigen Arbeitsablauf, der für die Entwicklung und Herstellung therapeutischer Zellen, darunter mesenchymale Stammzellen (MSCs), induzierte pluripotente Stammzellen (iPSCs) und verschiedene Immunzellen (z. B. CAR-T-Zellen) für die klinische Anwendung, erforderlich ist. Die Kernprozesse sind äußerst anspruchsvoll und müssen strikt den Richtlinien der Guten Herstellungspraxis (GMP) entsprechen. Der Bioprozessablauf umfasst typischerweise mehrere kritische Schritte: die Isolierung der Zellen aus dem Ausgangsmaterial, die großtechnische Zellvermehrung und -kultur zur Erreichung der therapeutischen Dosis, die Ernte und Aufreinigung des Endprodukts sowie die hochspezialisierte Konservierung und Kryolagerung für den Vertrieb. Der Markt befindet sich derzeit in einem bedeutenden Wandel von kostspieligen, patientenspezifischen autologen Therapien hin zu industrialisierten, kostengünstigen allogenen (gebrauchsfertigen) Therapien, was skalierbarere und standardisierte Produktionsplattformen erfordert.
Passen Sie diesen Bericht Ihren Anforderungen an.
Sie erhalten eine kostenlose Anpassung aller Berichte – einschließlich Teilen dieses Berichts, Länderanalysen und Excel-Datenpaketen – sowie attraktive Angebote und Rabatte für Start-ups und Universitäten.
Markt für Zelltherapie-Bioprozesse: Strategische Einblicke
-
Ermitteln Sie die wichtigsten Markttrends dieses Berichts.Diese KOSTENLOSE Probe beinhaltet eine Datenanalyse, die von Markttrends bis hin zu Schätzungen und Prognosen reicht.
Markttreiber und Chancen im Bereich der Zelltherapie-Bioprozessierung
Markttreiber:
-
Zunehmende Verbreitung chronischer und lebensbedrohlicher Krankheiten: Die weltweit steigende Inzidenz chronischer Krankheiten, insbesondere verschiedener Krebsarten (was die Nachfrage nach CAR-T- und adoptiven T-Zell-Therapien ankurbelt) und schwerer Herz-Kreislauf-Erkrankungen, führt zu einem dringenden und wachsenden Bedarf an innovativen therapeutischen Alternativen wie Zelltherapien.
-
Anstieg der F&E-Investitionen und der staatlichen Förderung: Erhebliche private und öffentliche Fördermittel, darunter groß angelegte staatliche Initiativen zur Unterstützung der regenerativen Medizin und neuartiger Therapieprodukte (ATMPs), treiben die Forschung, klinische Studien und den notwendigen Infrastrukturausbau für Bioprozesstechnologien voran.
-
Technologischer Wandel hin zu geschlossenen und automatisierten Systemen: Der Übergang zu automatisierten, geschlossenen Bioprozesssystemen, die das Kontaminationsrisiko verringern und die Prozesseffizienz und -standardisierung verbessern, ist entscheidend für die Erreichung der für die Kommerzialisierung erforderlichen Skalierbarkeit.
Marktchancen:
-
Expansion in Schwellenländer und CDMO-Dienstleistungen: Das Wachstum in Entwicklungsländern (wie beispielsweise im asiatisch-pazifischen Raum) mit ausgebauter Gesundheitsinfrastruktur und steigendem Patientenbewusstsein bietet riesige, bisher unerschlossene Märkte. Darüber hinaus eröffnet die zunehmende Abhängigkeit von spezialisierten Auftragsentwicklungs- und Produktionsunternehmen (CDMOs) für die Produktion im kommerziellen Maßstab bedeutende Chancen im Dienstleistungsbereich.
-
Integration der Prozessanalysetechnologie (PAT): Die Einbeziehung von künstlicher Intelligenz und digitalen Zwillingen für die Prozessmodellierung, die vorausschauende Wartung und die Echtzeit-Qualitätskontrolle (QC) bietet erhebliche Möglichkeiten zur Optimierung der Erträge, zur Reduzierung der Ausfallraten und zur Senkung der gesamten Herstellungskosten (COGS).
-
Standardisierung allogener Produktionsplattformen: Die Industrialisierung allogener Therapien erfordert hochskalierbare, robuste Bioprozesslösungen, wodurch sich für Hersteller eine große Chance ergibt, standardisierte Produktionsplattformen mit hohem Durchsatz zu entwickeln und zu vermarkten.
Marktbericht Zelltherapie-Bioprozessierung: Segmentierungsanalyse
Der Markt wird anhand mehrerer kritischer Segmente analysiert, die die Branchenstruktur und die Investitionsschwerpunkte definieren. Nachfolgend ist der in den meisten Branchenberichten verwendete Standard-Segmentierungsansatz dargestellt:
Durch Technologie:
-
Bioreaktor
-
Lyophilisierung
-
Elektrospinnen
-
Durchflusszentrifugation
-
Ultraschalllyse
-
Genomeditierungstechnologie
-
Zellimmortalisierungstechnologie
-
Virale Vektortechnologie
Nach Zelltyp:
- Stammzellen
- Immunzelle
- Menschliche embryonale Stammzelle
- Pluripotente Stammzelle
- Hämatopoetische Stammzellen
Nach Indikation:
-
Herz-Kreislauf-Erkrankungen
-
Onkologie
-
Wundheilung
-
Orthopädisch
Vom Endbenutzer:
-
Zentren für regenerative Medizin
-
Akademische Institute und Forschungsinstitute
-
Krankenhäuser und Kliniken
-
Diagnostische Zentren
Nach Geographie:
-
Nordamerika
-
Europa
-
Asien-Pazifik
-
Süd- und Mittelamerika
-
Naher Osten und Afrika
Regionale Einblicke in den Markt für Zelltherapie-Bioprozessierung
Die regionalen Trends und Einflussfaktoren auf den Markt für Zelltherapie-Bioprozessierung im gesamten Prognosezeitraum wurden von den Analysten von The Insight Partners eingehend erläutert. Dieser Abschnitt behandelt außerdem die Marktsegmente und die geografische Verteilung des Zelltherapie-Bioprozessierungsmarktes in Nordamerika, Europa, Asien-Pazifik, dem Nahen Osten und Afrika sowie Süd- und Mittelamerika.
Umfang des Marktberichts zur Zelltherapie-Bioprozessierung
| Berichtattribute | Details |
|---|---|
| Marktgröße im Jahr 2025 | 20,74 Milliarden US-Dollar |
| Marktgröße bis 2034 | 64,61 Milliarden US-Dollar |
| Globale durchschnittliche jährliche Wachstumsrate (2026 - 2034) | 13,49 % |
| Historische Daten | 2021-2024 |
| Prognosezeitraum | 2026–2034 |
| Abgedeckte Segmente |
Durch Technologie
|
| Abgedeckte Regionen und Länder |
Nordamerika
|
| Marktführer und wichtige Unternehmensprofile |
|
Marktdichte der Akteure im Bereich der Zelltherapie-Bioprozessierung: Auswirkungen auf die Geschäftsdynamik verstehen
Der Markt für Zelltherapie-Bioprozesse wächst rasant, angetrieben durch die steigende Nachfrage der Endverbraucher. Gründe hierfür sind unter anderem sich wandelnde Verbraucherpräferenzen, technologische Fortschritte und ein wachsendes Bewusstsein für die Vorteile des Produkts. Mit steigender Nachfrage erweitern Unternehmen ihr Angebot, entwickeln innovative Lösungen, um den Kundenbedürfnissen gerecht zu werden, und nutzen neue Trends, was das Marktwachstum zusätzlich beflügelt.
- Verschaffen Sie sich einen Überblick über die wichtigsten Akteure im Markt für Zelltherapie-Bioprozessierung
Marktanteilsanalyse für die Zelltherapie-Bioprozessierung nach Regionen
Der Markt weist in den verschiedenen Regionen unterschiedliche Reifegrade und Wachstumsraten auf, die maßgeblich von regulatorischen Rahmenbedingungen, öffentlicher Förderung und der Konzentration von Biotechnologie-Expertise beeinflusst werden. Nachfolgend finden Sie eine Zusammenfassung der Marktanteile und Trends nach Region:
-
Nordamerika
-
Marktanteil: Besitzt den größten Marktanteil, was auf ein etabliertes, fortschrittliches Ökosystem im Gesundheitswesen und in der Forschung und Entwicklung zurückzuführen ist.
-
Wichtigste Einflussfaktoren: Hohe Konzentration großer Pharma- und Biotech-Unternehmen, starkes regulatorisches Umfeld (z. B. beschleunigte Zulassungsverfahren der FDA) und erhebliche Risikokapitalfinanzierung für Zelltherapie-Startups.
-
Trends: Schnelle Einführung vollautomatisierter und digitalisierter Bioprozesslösungen und ein wachsendes Netzwerk spezialisierter CDMOs, die sich auf die Herstellung von CAR-T-Zellen im kommerziellen Maßstab konzentrieren.
-
-
Europa
-
Marktanteil: Besitzt einen bedeutenden Marktanteil, gestützt durch starke öffentliche Fördermittel und konsolidierte Biotech-Zentren.
-
Wichtigste Einflussfaktoren: Günstige Förderprogramme der Europäischen Union (EU) (z. B. Horizon Europe), vereinfachte regulatorische Rahmenbedingungen für ATMPs (Arzneimittel für neuartige Therapien) und ein äußerst aktives akademisches Forschungsumfeld.
-
Trends: Zunehmender Fokus auf die Einrichtung harmonisierter, standortübergreifender Produktionsnetzwerke zur Abwicklung grenzüberschreitender klinischer Studien und der kommerziellen Versorgung in den EU-Mitgliedstaaten.
-
-
Asien-Pazifik (APAC)
-
Marktanteil: Voraussichtlich die am schnellsten wachsende Region, angeführt von China und Indien.
-
Wichtigste Einflussfaktoren: Rasch zunehmende staatliche Initiativen zur Förderung der heimischen Life-Science- und Biotech-Sektoren, Ausbau der Gesundheitsinfrastruktur, eine große und schnell alternde Patientenpopulation sowie Kostenvorteile in der Fertigung.
-
Trends: Hohe Investitionen in den Aufbau von GMP-konformen Produktionsanlagen, starker Fokus auf die Einführung modernster, kosteneffizienter Automatisierungstechnologien und Beschleunigung der lokalen klinischen Studienaktivitäten.
-
-
Süd- und Mittelamerika sowie Naher Osten und Afrika (SCA & MEA)
-
Marktanteil: Aufstrebende Märkte mit erheblichem Wachstumspotenzial.
-
Wichtigste Einflussfaktoren: Steigende Investitionen in die Modernisierung der Gesundheitsinfrastruktur, wachsendes Bewusstsein und erste regulatorische Schritte zur Einführung von Zelltherapien sowie öffentlich-private Partnerschaften mit Schwerpunkt auf medizinischer Innovation.
-
Trends: Frühe Einführung leicht verfügbarer, skalierbarer Technologien wie geschlossener Einwegsysteme, um schnell erste Bioprozesskapazitäten aufzubauen.
-
Marktdichte der Akteure im Bereich der Zelltherapie-Bioprozessierung: Auswirkungen auf die Geschäftsdynamik verstehen
Hohe Marktdichte und starker Wettbewerb
Der Markt ist durch einen intensiven Wettbewerb zwischen etablierten Anbietern von Werkzeugen für die Biowissenschaften und spezialisierten Anbietern von Zelltherapielösungen gekennzeichnet.
Führende Unternehmen wie Thermo Fisher Scientific Inc., Sartorius AG und Lonza nutzen ihre umfassende Expertise im Bereich Bioprozessanlagen und CDMO-Dienstleistungen, um integrierte Lösungen entlang der gesamten Wertschöpfungskette der Zelltherapie anzubieten. Der Wettbewerb treibt die kontinuierliche Innovation voran, wobei sich die Anbieter auf Folgendes konzentrieren:
-
Entwicklung geschlossener Systeme: Schaffung hocheffizienter, geschlossener und automatisierter Systeme zur Reduzierung des Kontaminationsrisikos und des manuellen Arbeitsaufwands.
-
Strategische Partnerschaften: Zusammenarbeit mit Biotech-Unternehmen im klinischen Stadium zur Sicherung langfristiger Lieferverträge für Reagenzien und Ausrüstung.
-
Anlagenerweiterungen: Steigerung der globalen Produktionskapazität, insbesondere bei spezialisierten viralen Vektorproduktions- und Zellverarbeitungsdienstleistungen (CDMOs).
Wichtige Unternehmen, die auf dem Markt für Zelltherapie-Bioprozessierung tätig sind
-
Thermo Fisher Scientific Inc.
-
Sartorius AG
-
Lonza
-
Merck KGaA
-
Cytiva
-
Corning Incorporated
-
Fresenius Kabi AG
-
Asahi Kasei Corporation
-
Repligen Corporation
Hinweis: Die oben aufgeführten Unternehmen sind nicht in einer bestimmten Reihenfolge geordnet.
Neuigkeiten und aktuelle Entwicklungen auf dem Markt für Bioprozesse in der Zelltherapie
-
Thermo Fisher hat OpTmizer One SFM vorgestellt, ein spezielles serumfreies Medium, das entwickelt wurde, um die Expansion von T-Zellen signifikant zu steigern und den frühen Gedächtnis-T-Zell-Phänotyp zu erhalten – ein entscheidendes Qualitätsmerkmal für effektive Zelltherapien.
-
Im Oktober 2025 kündigte Miltenyi Biotec an, seine lentiviralen Vektoren (LVV) für Immatics' anzutresgene autoleucel (anzu-cel, IMA203) kommerziell zu liefern und demonstrierte damit seine wachsende Rolle als CDMO und seine zuverlässige Lieferkette für kritische Rohstoffe.
-
Lonza hat seine Expertise im Bereich der Zell- und Gentherapie durch ein umfassendes, durchgängiges Prozessentwicklungsangebot gestärkt. Dieses deckt den gesamten therapeutischen Lebenszyklus ab, von der Unterstützung bei der Einreichung von Prüfpräparatenanträgen (IND) in der frühen Phase bis hin zur kommerziellen GMP-konformen Herstellung.
Marktbericht zur Bioprozessierung in der Zelltherapie: Abdeckung und Ergebnisse
Der Bericht „Marktgröße und Prognose für die Zelltherapie-Bioprozessierung (2021–2034)“ bietet eine detaillierte Analyse des Marktes und deckt die folgenden Bereiche ab:
-
Marktgröße und Prognose für die Zelltherapie-Bioprozessierung auf globaler, regionaler und Länderebene für alle wichtigen Marktsegmente, die in den Geltungsbereich fallen.
-
Trends im Markt für Zelltherapie-Bioprozessierung sowie Marktdynamiken wie Treiber, Hemmnisse und wichtige Chancen.
-
Detaillierte PEST- und SWOT-Analyse.
-
Analyse des Marktes für Zelltherapie-Bioprocessing mit Fokus auf wichtige Markttrends, globale und regionale Rahmenbedingungen, Hauptakteure, regulatorische Bestimmungen und aktuelle Marktentwicklungen.
-
Branchenlandschaft und Wettbewerbsanalyse mit Fokus auf Marktkonzentration, Heatmap-Analyse, prominente Akteure und aktuelle Entwicklungen im Markt für Zelltherapie-Bioprozessierung.
-
Detaillierte Unternehmensprofile.
- Historische Analyse (2 Jahre), Basisjahr, Prognose (7 Jahre) mit CAGR
- PEST- und SWOT-Analyse
- Marktgröße Wert/Volumen – Global, Regional, Land
- Branchen- und Wettbewerbslandschaft
- Excel-Datensatz
Erfahrungsberichte
Grund zum Kauf
- Fundierte Entscheidungsfindung
- Marktdynamik verstehen
- Wettbewerbsanalyse
- Kundeneinblicke
- Marktprognosen
- Risikominimierung
- Strategische Planung
- Investitionsbegründung
- Identifizierung neuer Märkte
- Verbesserung von Marketingstrategien
- Steigerung der Betriebseffizienz
- Anpassung an regulatorische Trends















Exklusive Berichtsrabatte freischalten
Jetzt anfragen






 Kostenlose Probe anfordern für - Markt für Zelltherapie-Bioprozesse
Kostenlose Probe anfordern für - Markt für Zelltherapie-Bioprozesse