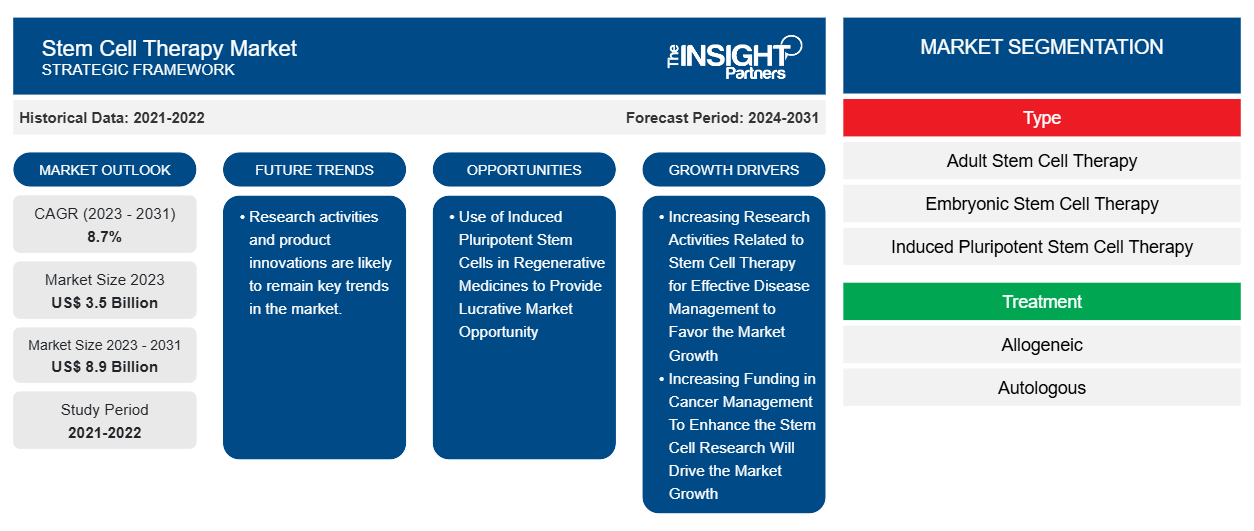
Le marché de la thérapie par cellules souches devrait atteindre 8,9 milliards de dollars d'ici 2031, contre 3,5 milliards de dollars en 2023. Le marché devrait enregistrer un TCAC de 8,7 % au cours de la période 2023-2031. Les activités de recherche et les innovations de produits devraient rester les principales tendances du marché.
Analyse du marché de la thérapie par cellules souches
Les activités de recherche et les innovations de produits devraient rester les principales tendances du marché. Un rapport d'EuroGCT révèle que la greffe de cellules souches sanguines s'avère efficace pour traiter les types de cancer du sang tels que la leucémie, le lymphome et le myélome, le syndrome myélodysplasique et les troubles myéloprolifératifs. De plus, les greffes de cellules souches sanguines ont cliniquement prouvé leur efficacité pour traiter le déficit immunitaire combiné sévère (DICS) en remplaçant les cellules immunitaires défectueuses par des cellules saines du donneur. Par conséquent, les activités de recherche et l'innovation de produits sont un facteur influent responsable de la croissance considérable du marché au cours de la période 2021-2031.
Aperçu du marché de la thérapie par cellules souches
L'augmentation du financement ou des investissements permettra d'améliorer la recherche sur les cellules souches. En 2023, la Fondation Lee Shau Kee, basée à Hong Kong, a accordé à l'Institut de régénération et de réparation (IRR) de l'université 10,82 millions de dollars américains pour stimuler la recherche sur les cellules souches et l'ingénierie tissulaire de l'université. Le fonds soutiendra le programme de collaboration internationale en biologie des cellules souches qui aidera l'IRR à lutter contre des maladies telles que le dysfonctionnement chronique des organes, les traumatismes et les maladies congénitales et génétiques. Par conséquent, l'augmentation du financement ou des investissements accélérera la demande de thérapie par cellules souches au cours de la prochaine décennie.
Personnalisez ce rapport en fonction de vos besoins
Vous bénéficierez d'une personnalisation gratuite de n'importe quel rapport, y compris de certaines parties de ce rapport, d'une analyse au niveau des pays, d'un pack de données Excel, ainsi que de superbes offres et réductions pour les start-ups et les universités.
Marché de la thérapie par cellules souches : informations stratégiques
-
Obtenez les principales tendances clés du marché de ce rapport.Cet échantillon GRATUIT comprendra une analyse de données, allant des tendances du marché aux estimations et prévisions.
Moteurs et opportunités du marché de la thérapie par cellules souches
Augmenter les activités de recherche liées à la thérapie par cellules souches pour une gestion efficace des maladies afin de favoriser le marché
La thérapie par cellules souches a fait l'objet de nombreuses recherches à l'échelle mondiale, car les cellules souches sont principalement utilisées pour remplacer les cellules mourantes et reconstruire les tissus endommagés. Sur la base des résultats cliniques menés par les scientifiques ciblant les cellules souches, on affirme que ces cellules pourraient probablement être utilisées pour générer des traitements spécifiques pour plusieurs maladies telles que le cancer et les maladies cardiovasculaires. De plus, les thérapies par cellules souches développées impliquent le remplacement des cellules responsables de la maladie par des cellules souches. Les chercheurs étudient en outre l'utilisation de la thérapie par cellules souches pour traiter les maladies auto-immunes. Par conséquent, l'augmentation des activités de recherche liées à la thérapie par cellules souches pour une gestion efficace des maladies est l'un des principaux moteurs de la part de marché mondiale de la thérapie par cellules souches.
L'utilisation de cellules souches pluripotentes induites dans les médicaments régénératifs offre une opportunité de marché lucrative
Selon le rapport Frontiers Media SA 2023, les progrès réalisés dans le domaine des cellules souches pluripotentes induites (iPSC) ont ouvert de nouvelles voies pour la recherche en thérapeutique. Les iPSC sont principalement destinées à la recherche et aux études cliniques pour la modélisation des maladies, la médecine régénérative et les études de découverte de médicaments/cytotoxicité des médicaments. Par exemple, les iPSC peuvent être utilisées pour convertir des cellules somatiques différenciées en cellules souches multipotentes afin de générer différents types de cellules de tissus adultes. Par conséquent, les iPSC ont montré des résultats prometteurs en médecine régénérative, en particulier pour remplacer les cellules malades ou blessées. Par conséquent, des études telles que la découverte de médicaments impliquant le criblage de petites molécules et des tests de toxicité pour l'évaluation de la sécurité ont été révélées avec succès. Les résultats positifs basés sur les iPSC sont susceptibles de créer des opportunités lucratives pour les acteurs du marché de la thérapie par cellules souches dans les années à venir.
Analyse de segmentation du rapport sur le marché de la thérapie par cellules souches
Les segments clés qui ont contribué à l’élaboration de l’analyse du marché de la thérapie par cellules souches sont le type, le traitement, l’application et l’utilisateur final.
- En fonction du type, le marché de la thérapie par cellules souches est segmenté en thérapie par cellules souches adultes, thérapie par cellules souches embryonnaires, thérapie par cellules souches pluripotentes induites et autres thérapies par cellules souches. Le segment de la thérapie par cellules souches adultes détenait une part de marché plus importante en 2023.
- En termes de traitement, le marché est divisé en allogénique et autologue. Le segment allogénique détenait la plus grande part du marché en 2023.
- En termes d'application, le marché est segmenté en musculo-squelettique, dermatologie , cardiologie, découverte et développement de médicaments et autres applications. Le segment de la découverte et du développement de médicaments détenait la plus grande part du marché en 2023.
- En termes d'utilisateur final, le marché est segmenté en hôpitaux et cliniques spécialisées et en instituts universitaires et de recherche. Le segment des instituts universitaires et de recherche détenait la plus grande part du marché en 2023.
Analyse des parts de marché de la thérapie par cellules souches par zone géographique
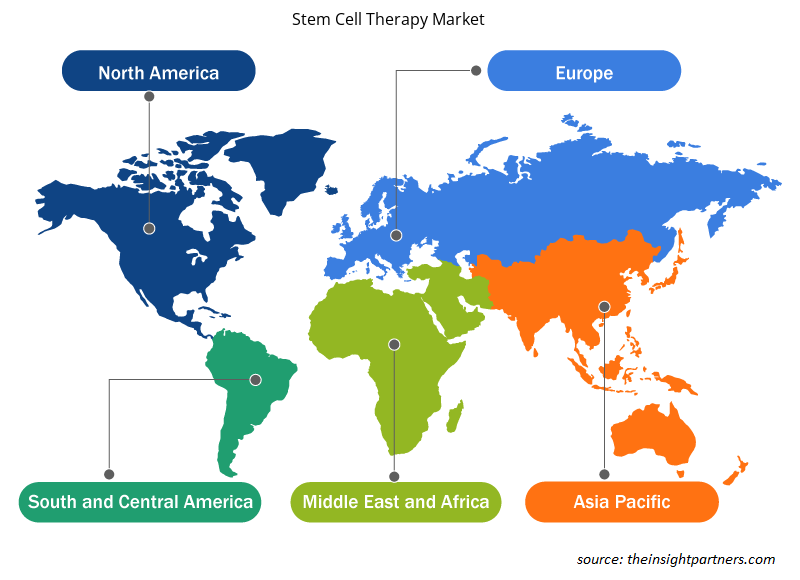
La portée géographique du rapport sur le marché de la thérapie par cellules souches est principalement divisée en cinq régions : Amérique du Nord, Asie-Pacifique, Europe, Moyen-Orient et Afrique, et Amérique du Sud et centrale.
L'Amérique du Nord domine le marché. En Amérique du Nord, les États-Unis représentent la plus grande part du marché de la thérapie par cellules souches. La présence des principaux fabricants dans la région et les activités de recherche et développement (R&D) élevées sont les facteurs contribuant à la domination du marché. L'Asie-Pacifique devrait connaître le TCAC le plus élevé dans les années à venir.
Aperçu régional du marché de la thérapie par cellules souches
Les tendances et facteurs régionaux influençant le marché de la thérapie par cellules souches tout au long de la période de prévision ont été expliqués en détail par les analystes d’Insight Partners. Cette section traite également des segments et de la géographie du marché de la thérapie par cellules souches en Amérique du Nord, en Europe, en Asie-Pacifique, au Moyen-Orient et en Afrique, ainsi qu’en Amérique du Sud et en Amérique centrale.
- Obtenez les données régionales spécifiques au marché de la thérapie par cellules souches
Portée du rapport sur le marché de la thérapie par cellules souches
| Attribut de rapport | Détails |
|---|---|
| Taille du marché en 2023 | 3,5 milliards de dollars américains |
| Taille du marché d'ici 2031 | 8,9 milliards de dollars américains |
| Taux de croissance annuel composé mondial (2023-2031) | 8,7% |
| Données historiques | 2021-2022 |
| Période de prévision | 2024-2031 |
| Segments couverts |
Par type
|
| Régions et pays couverts |
Amérique du Nord
|
| Leaders du marché et profils d'entreprises clés |
|
Densité des acteurs du marché de la thérapie par cellules souches : comprendre son impact sur la dynamique commerciale
Le marché de la thérapie par cellules souches connaît une croissance rapide, tirée par la demande croissante des utilisateurs finaux en raison de facteurs tels que l'évolution des préférences des consommateurs, les avancées technologiques et une plus grande sensibilisation aux avantages du produit. À mesure que la demande augmente, les entreprises élargissent leurs offres, innovent pour répondre aux besoins des consommateurs et capitalisent sur les tendances émergentes, ce qui alimente davantage la croissance du marché.
La densité des acteurs du marché fait référence à la répartition des entreprises ou des sociétés opérant sur un marché ou un secteur particulier. Elle indique le nombre de concurrents (acteurs du marché) présents sur un marché donné par rapport à sa taille ou à sa valeur marchande totale.
Les principales entreprises opérant sur le marché de la thérapie par cellules souches sont :
- MÉDIPOST
- RichSource
- BioTime, Inc
- Mésoblast Limitée
- Holostem Thérapie Avancé Srl
- Cellules souches américaines, Inc.
Avis de non-responsabilité : les sociétés répertoriées ci-dessus ne sont pas classées dans un ordre particulier.

- Obtenez un aperçu des principaux acteurs du marché de la thérapie par cellules souches
Actualités et développements récents du marché de la thérapie par cellules souches
Le marché de la thérapie par cellules souches est évalué en collectant des données qualitatives et quantitatives après des recherches primaires et secondaires, qui comprennent des publications d'entreprise importantes, des données d'association et des bases de données. Quelques-uns des développements du marché de la thérapie par cellules souches sont répertoriés ci-dessous :
- Allogene Therapeutics, Inc. a annoncé que la Food and Drug Administration (FDA) des États-Unis a accordé la désignation Fast Track à « ALLO-605 ». Le produit nouvellement accordé est la thérapie AlloCAR T de nouvelle génération de la société pour le traitement du myélome multiple récidivant ou réfractaire. La FDA a accordé la désignation Fast Track en raison du potentiel d'ALLO-605 à répondre aux besoins non satisfaits des patients qui ont échoué aux autres thérapies standard contre le myélome multiple. (Source : Allogene Therapeutics, Inc., site Web de la société, juin 2021)
Rapport sur le marché de la thérapie par cellules souches : couverture et livrables
Le rapport « Taille et prévisions du marché de la thérapie par cellules souches (2021-2031) » fournit une analyse détaillée du marché couvrant les domaines ci-dessous :
- Taille et prévisions du marché de la thérapie par cellules souches aux niveaux mondial, régional et national pour tous les segments de marché clés couverts par le champ d'application
- Tendances du marché de la thérapie par cellules souches ainsi que dynamique du marché telles que les moteurs, les contraintes et les opportunités clés
- Analyse détaillée des cinq forces de PEST/Porter et SWOT
- Analyse du marché de la thérapie par cellules souches couvrant les principales tendances du marché, le cadre mondial et régional, les principaux acteurs, les réglementations et les développements récents du marché
- Analyse du paysage industriel et de la concurrence couvrant la concentration du marché, l'analyse de la carte thermique, les principaux acteurs et les développements récents pour le marché de la thérapie par cellules souches
- Profils d'entreprise détaillés
- Analyse historique (2 ans), année de base, prévision (7 ans) avec TCAC
- Analyse PEST et SWOT
- Taille du marché Valeur / Volume - Mondial, Régional, Pays
- Industrie et paysage concurrentiel
- Ensemble de données Excel
Rapports récents
Rapports connexes
Témoignages
Raison d'acheter
- Prise de décision éclairée
- Compréhension de la dynamique du marché
- Analyse concurrentielle
- Connaissances clients
- Prévisions de marché
- Atténuation des risques
- Planification stratégique
- Justification des investissements
- Identification des marchés émergents
- Amélioration des stratégies marketing
- Amélioration de l'efficacité opérationnelle
- Alignement sur les tendances réglementaires






















 Obtenez un échantillon gratuit pour - Marché de la thérapie par cellules souches
Obtenez un échantillon gratuit pour - Marché de la thérapie par cellules souches