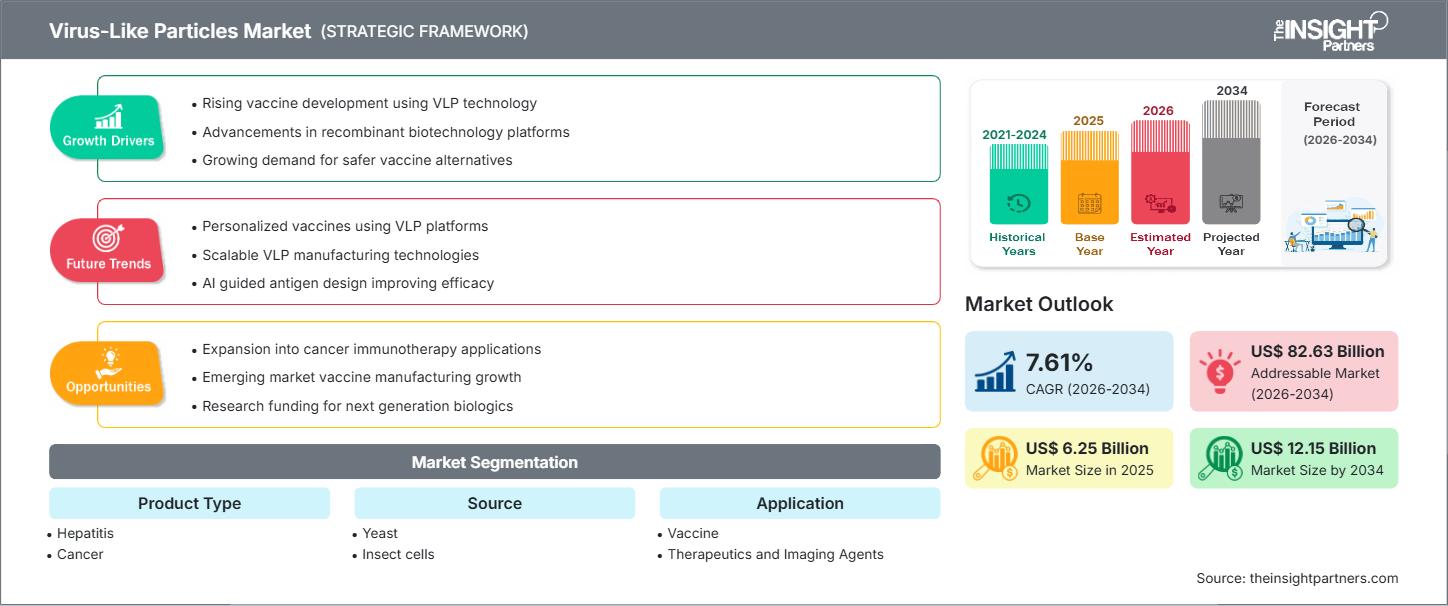
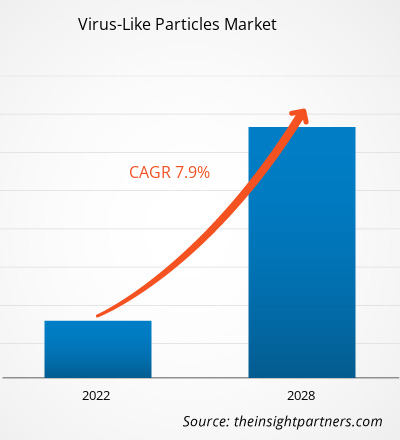
Le marché mondial des particules pseudo-virales devrait atteindre 12,15 milliards de dollars américains d'ici 2034, contre 6,25 milliards de dollars américains en 2025. Ce marché devrait enregistrer un taux de croissance annuel composé (TCAC) de 7,61 % au cours de la période de prévision 2026-2034.
Analyse du marché des particules pseudo-virales
Le marché des particules pseudo-virales (PPV) exige des acteurs du secteur qu'ils passent du développement traditionnel des vaccins à des stratégies d'administration ciblées, axées sur une forte marge, afin de tirer pleinement parti de la croissance prévue. L'utilisation des PPV comme vecteurs ciblés pour les thérapies à base d'ARN et les composants CRISPR représente une opportunité majeure. Cette approche pourrait constituer une alternative intéressante aux nanoparticules lipidiques (NPL) pour franchir la barrière hémato-encéphalique. Il est également important que les décideurs envisagent les systèmes d'expression sur cellules d'insectes et levures, car ces méthodes peuvent être adaptées à la demande mondiale en matière de vaccins contre les pandémies et les maladies respiratoires saisonnières. Les entreprises qui utilisent la modélisation par intelligence artificielle pour concevoir des échafaudages synthétiques peuvent développer plus rapidement des vaccins multivalents, ce qui leur confère un avantage concurrentiel certain. En concentrant la recherche et le développement sur les immunothérapies en oncologie et en créant des pôles de production locaux dans la région Asie-Pacifique, le profil de sécurité favorable de la technologie PPV peut se traduire par une source de revenus durable et évolutive, permettant de lutter contre les maladies infectieuses et non transmissibles.
Aperçu du marché des particules pseudo-virales
Le marché des particules pseudo-virales (PPV) est un secteur spécialisé de la biotechnologie axé sur la production et l'application de nanostructures auto-assemblées qui imitent l'architecture externe des virus. Ces particules, dépourvues de génome viral, ne peuvent provoquer d'infection, mais induisent une forte réponse immunitaire. La majeure partie du marché se concentre sur les vaccins à base de PPV, comme ceux contre l'hépatite B et le papillomavirus humain (VPH), qui utilisent ces particules pour présenter de nombreux antigènes au système immunitaire. Outre les vaccins, les PPV sont également utilisées pour administrer des médicaments anticancéreux et des agents d'imagerie directement dans des zones ciblées. Cet aperçu offre une vision globale de ce secteur technologique de pointe qui fait le lien entre la vaccination traditionnelle et la médecine de précision de nouvelle génération.
Personnalisez ce rapport selon vos besoins.
Bénéficiez d'une PERSONNALISATION GRATUITEMarché des particules pseudo-virales : perspectives stratégiques
-
Découvrez les principales tendances du marché présentées dans ce rapport.Cet échantillon GRATUIT comprendra une analyse de données, allant des tendances du marché aux estimations et prévisions.
Facteurs de croissance et opportunités du marché des particules pseudo-virales
Facteurs de marché :
- Demande croissante de vaccins et d'immunothérapies à haute efficacité : les organismes de santé du monde entier constatent une augmentation des cas de maladies infectieuses et de maladies chroniques telles que le cancer. De ce fait, le besoin en particules pseudo-virales (VLP), qui offrent une meilleure réponse immunitaire et une sécurité accrue par rapport aux vaccins atténués traditionnels, se fait de plus en plus sentir.
- Progrès technologiques dans les systèmes d'expression : Le passage à la technologie recombinante dans les lignées cellulaires de levure et d'insectes a considérablement élargi le champ d'application de la production de particules pseudo-virales (VLP). Ces systèmes permettent une fabrication à grande échelle et rentable, répondant à la demande mondiale de déploiement rapide de vaccins lors de crises sanitaires.
- Renforcement des initiatives gouvernementales et des financements pour la prévention : les particules pseudo-virales (PPV) font partie des solutions biopharmaceutiques dont la généralisation est soutenue par les stratégies nationales de santé. Des initiatives mondiales de vaccination aux plans régionaux de préparation aux pandémies, les professionnels de santé sont incités à adopter des plateformes à base de PPV pour une protection à large spectre.
Opportunités de marché :
- Expansion en oncologie et médecine personnalisée : les particules pseudo-virales (VLP) sont de plus en plus utilisées pour le développement de vaccins contre le cancer dans les marchés émergents et les centres biopharmaceutiques établis. Ces systèmes d’administration permettent de transporter de manière flexible les antigènes associés aux tumeurs, contribuant ainsi à répondre au besoin croissant en oncologie de précision.
- Intégration à l'imagerie avancée et aux thérapies ciblées : l'association de particules pseudo-virales (VLPs) à des marqueurs fluorescents et à des agents thérapeutiques améliore le diagnostic et le traitement. L'utilisation des VLPs comme nanovecteurs favorise une meilleure prise de décision clinique et une administration plus efficace des chimiothérapies aux zones ciblées.
- Demande croissante de plateformes multi-antigéniques et d'optimisation des processus : La tendance aux vaccins polyvalents, capables de protéger simultanément contre plusieurs souches ou différents agents pathogènes, est un moteur essentiel de l'innovation en matière de VLP. Ces systèmes permettent d'alléger la charge administrative des établissements de santé et d'optimiser l'utilisation des ressources pour les stocks nationaux de vaccins.
Analyse de segmentation du rapport sur le marché des particules pseudo-virales
La part de marché des particules pseudo-virales est analysée selon différents segments afin de mieux comprendre sa structure et son potentiel de croissance. Voici l'approche de segmentation standard utilisée dans les rapports sectoriels :
Par type de produit :
- Hépatite
- Cancer
Source :
- Levure
- Cellules d'insectes
Sur demande :
- Vaccins
- Agents thérapeutiques et d'imagerie
Par géographie :
- Amérique du Nord
- Europe
- Asie-Pacifique
- Amérique du Sud et centrale
- Moyen-Orient et Afrique
Aperçu régional du marché des particules pseudo-virales
Les tendances régionales et les facteurs influençant le marché des particules pseudo-virales tout au long de la période prévisionnelle ont été analysés en détail par les analystes de The Insight Partners. Cette section aborde également les segments de marché et la répartition géographique du marché des particules pseudo-virales en Amérique du Nord, en Europe, en Asie-Pacifique, au Moyen-Orient et en Afrique, ainsi qu'en Amérique du Sud et centrale.
Portée du rapport sur le marché des particules pseudo-virales
| Attribut du rapport | Détails |
|---|---|
| Taille du marché en 2025 | 6,25 milliards de dollars américains |
| Taille du marché d'ici 2034 | 12,15 milliards de dollars américains |
| TCAC mondial (2026 - 2034) | 7,61% |
| Données historiques | 2021-2024 |
| Période de prévision | 2026-2034 |
| Segments couverts |
Par type de produit
|
| Régions et pays couverts |
Amérique du Nord
|
| Leaders du marché et profils d'entreprises clés |
|
Densité des acteurs du marché des particules pseudo-virales : comprendre son impact sur la dynamique commerciale
Le marché des particules pseudo-virales connaît une croissance rapide, portée par une demande croissante des utilisateurs finaux. Cette demande est alimentée par l'évolution des préférences des consommateurs, les progrès technologiques et une meilleure connaissance des avantages du produit. Face à cette demande grandissante, les entreprises diversifient leur offre, innovent pour répondre aux besoins des consommateurs et tirent parti des tendances émergentes, ce qui contribue à stimuler la croissance du marché.
- Obtenez un aperçu des principaux acteurs du marché des particules pseudo-virales
Analyse des parts de marché des particules pseudo-virales par zone géographique
La région Asie-Pacifique devrait connaître la croissance la plus rapide dans les années à venir. Les marchés émergents d'Amérique du Sud et centrale, du Moyen-Orient et d'Afrique offrent également de nombreuses opportunités inexploitées aux fournisseurs de VLP pour se développer.
Le marché des particules pseudo-virales présente une trajectoire de croissance différente selon les régions, en raison de facteurs tels que l'infrastructure biopharmaceutique, les investissements en R&D, le contexte réglementaire et la prévalence des maladies infectieuses. Vous trouverez ci-dessous un résumé des parts de marché et des tendances par région :
Amérique du Nord
- Part de marché : Détient la plus grande part de marché grâce à un secteur biopharmaceutique mature et à des investissements élevés en R&D.
-
Facteurs clés :
- Forte présence des principaux acteurs biopharmaceutiques
- infrastructure avancée pour les essais cliniques
- Forte sensibilisation du public à la vaccination préventive
- Tendances : Évolution vers la technologie VLP auto-assemblée et les unités de bioprocédés intégrées afin de rationaliser la fabrication et de réduire les délais de mise sur le marché.
Europe
- Part de marché : Déterminée par les systèmes de santé publique et le soutien gouvernemental important à la recherche sur le cancer.
-
Facteurs clés :
- Des réglementations de sécurité strictes et des protocoles alignés sur l'EMA
- Des programmes nationaux de vaccination robustes
- Investissement dans l'immunothérapie anticancéreuse basée sur les VLP
- Tendances : Utilisation croissante des plateformes VLP pour le développement de vaccins contre les virus respiratoires et zoonotiques émergents.
Asie-Pacifique
- Part de marché : Région à la croissance la plus rapide grâce à l’industrialisation rapide du secteur biopharmaceutique en Chine, en Inde et en Corée du Sud.
-
Facteurs clés :
- Initiatives « Fabriquer en Inde » et « Fabriqué en Chine 2025 » soutenues par le gouvernement
- Augmentation du nombre de patients et des dépenses de santé
- Expansion des organisations locales de fabrication sous contrat (CMO)
- Tendances : Production locale de vaccins abordables à base de VLP destinés à la consommation intérieure et à l'exportation vers d'autres économies émergentes.
Amérique du Sud et centrale
- Part de marché : Marché émergent axé sur la production locale pour réduire la dépendance aux importations.
-
Facteurs clés :
- Prévalence croissante des maladies à transmission vectorielle comme Zika et la dengue
- Partenariats public-privé pour l'autosuffisance vaccinale régionale
- Modernisation des installations de production biologique au Brésil et au Mexique
- Tendances : Partenariats entre entreprises mondiales de biotechnologie et instituts régionaux (comme Fiocruz ou Butantan) pour faciliter le transfert de la technologie VLP en vue de la lutte contre les maladies endémiques.
Moyen-Orient et Afrique
- Part de marché : Marché en développement avec un potentiel de croissance stimulé par les objectifs nationaux de sécurité sanitaire.
-
Facteurs clés :
- Stratégies nationales de cybersanté et localisation de la production biopharmaceutique
- Demande croissante de vaccins contre l'hépatite et le VPH à un coût abordable
- Investissements des fonds souverains dans les sciences de la vie
- Tendances : Priorité à la mise en place de plateformes de production modulaires de particules pseudo-virales (VLP) afin de répondre rapidement aux épidémies virales régionales et aux urgences de santé publique.
Densité des acteurs du marché des particules pseudo-virales : comprendre son impact sur la dynamique commerciale
Forte densité de marché et concurrence
La concurrence s'intensifie en raison de la présence de leaders pharmaceutiques mondiaux tels que Merck & Co., Inc., GlaxoSmithKline plc. et Pfizer Inc. Les entreprises de biotechnologie spécialisées et les fabricants à grande échelle comme le Serum Institute of India et LG Chem contribuent également à un marché de plus en plus encombré.
Ce contexte concurrentiel pousse les fournisseurs à se différencier par :
- Plateformes de production évolutives : Utilisation de bioréacteurs à usage unique avancés pour les lignées cellulaires de levure, de cellules d'insectes (baculovirus) et de mammifères afin de garantir une production à haut rendement.
- Polyvalence de la plateforme : Développement de VLPs « prêtes à l’emploi » pouvant être facilement conjuguées à divers antigènes pour une réponse rapide aux épidémies de maladies infectieuses émergentes.
- Partenariats stratégiques de recherche : Collaborer avec des instituts de recherche internationaux pour étendre les applications des VLP au-delà des vaccins, notamment en oncologie et en gestion des maladies chroniques.
- Intégration avancée d'adjuvants : Amélioration de l'immunogénicité des candidats VLP grâce à des technologies adjuvantes exclusives afin d'améliorer l'efficacité clinique dans différents groupes d'âge.
Opportunités et initiatives stratégiques
- La collaboration avec les organismes de santé publique et les organisations non gouvernementales (ONG) peut étendre la distribution des vaccins dans les économies émergentes, faisant ainsi progresser la vaccination mondiale et renforçant la préparation aux pandémies.
- L'utilisation de la modélisation des protéines pilotée par l'IA et de la conception informatique peut améliorer la stabilité des particules pseudo-virales (VLP) et permettre un ciblage précis des voies immunitaires pour les thérapies de nouvelle génération.
Les principales entreprises opérant sur le marché des particules pseudo-virales sont :
- Merck & Co., Inc. – États-Unis
- GlaxoSmithKline plc. – Royaume-Uni
- Pfizer Inc. – États-Unis
- Dynavax Technologies – États-Unis
- Sanofi – France
- Wantai BioPharm – Chine
- Institut du sérum de l'Inde Pvt. Ltd. – Inde
- BHARAT BIOTECH – Inde
- LG Chem – Corée du Sud
Avertissement : Les entreprises mentionnées ci-dessus ne sont classées dans aucun ordre particulier.
Actualités et développements récents du marché des particules pseudo-virales
- En mars 2024, Merck & Co. a annoncé son intention de lancer le développement clinique d'un vaccin HPV multivalent de nouvelle génération, s'appuyant sur sa technologie exclusive de particules pseudo-virales, ainsi que des essais évaluant un schéma vaccinal à dose unique de GARDASIL 9. Ce développement témoigne de l'innovation continue, de l'expansion du portefeuille de produits et du potentiel de croissance à long terme du marché des vaccins à particules pseudo-virales.
- En février 2024, AstraZeneca a annoncé la finalisation de l'acquisition d'Icosavax, Inc., société biopharmaceutique américaine en phase clinique spécialisée dans le développement de vaccins différenciés à fort potentiel grâce à une plateforme innovante de particules pseudo-virales (VLP) protéiques. Suite à cette acquisition, Icosavax est devenue une filiale d'AstraZeneca et est basée à Seattle, aux États-Unis. Cette acquisition témoigne de l'investissement croissant du secteur pharmaceutique et de la confiance accordée aux technologies vaccinales basées sur les VLP.
Couverture et livrables du rapport sur le marché des particules pseudo-virales
Le rapport « Taille et prévisions du marché des particules pseudo-virales (2021-2034) » fournit une analyse détaillée du marché couvrant les domaines suivants :
- Taille et prévisions du marché des particules pseudo-virales aux niveaux mondial, régional et national pour tous les segments de marché clés couverts par le présent document.
- Tendances du marché des particules pseudo-virales, ainsi que sa dynamique, notamment les facteurs moteurs, les contraintes et les principales opportunités
- Analyse PEST et SWOT détaillée
- Analyse du marché des particules pseudo-virales couvrant les principales tendances du marché, le cadre mondial et régional, les principaux acteurs, la réglementation et les développements récents du marché
- Analyse du paysage industriel et de la concurrence, incluant la concentration du marché, l'analyse par carte thermique, les principaux acteurs et les développements récents sur le marché des particules pseudo-virales
- Profils d'entreprise détaillés
- Analyse historique (2 ans), année de base, prévision (7 ans) avec TCAC
- Analyse PEST et SWOT
- Taille du marché Valeur / Volume - Mondial, Régional, Pays
- Industrie et paysage concurrentiel
- Ensemble de données Excel
Rapports récents
Témoignages
Raison d'acheter
- Prise de décision éclairée
- Compréhension de la dynamique du marché
- Analyse concurrentielle
- Connaissances clients
- Prévisions de marché
- Atténuation des risques
- Planification stratégique
- Justification des investissements
- Identification des marchés émergents
- Amélioration des stratégies marketing
- Amélioration de l'efficacité opérationnelle
- Alignement sur les tendances réglementaires






















 Obtenez un échantillon gratuit pour - Marché des particules pseudo-virales
Obtenez un échantillon gratuit pour - Marché des particules pseudo-virales