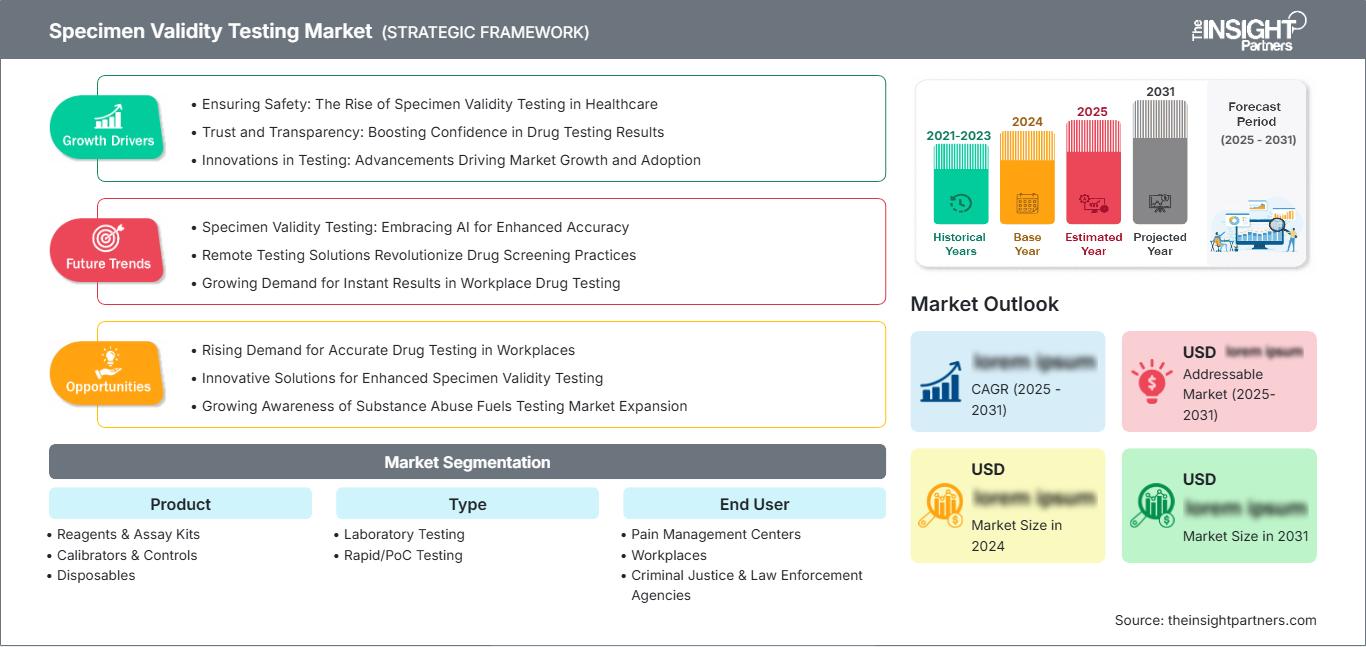
Si prevede che il mercato dei test di validità dei campioni raggiungerà i 5,03 miliardi di dollari entro il 2031. Si prevede che il mercato registrerà un CAGR del 6,8% nel periodo 2025-2031.
Il rapporto è suddiviso in base al prodotto (reagenti e kit di analisi, calibratori e controlli, materiali monouso) e analizza ulteriormente il mercato in base alla tipologia (test di laboratorio, test rapidi/PoC). Esamina inoltre il mercato per utente finale (centri di terapia del dolore, luoghi di lavoro, agenzie di giustizia penale e forze dell'ordine, centri di riabilitazione dalla droga, laboratori di screening antidroga). Per ciascuno di questi segmenti chiave viene fornita una ripartizione completa a livello globale, regionale e nazionale.
Il rapporto include dimensioni e previsioni di mercato per tutti i segmenti, presentando i valori in dollari statunitensi. Fornisce inoltre statistiche chiave sullo stato attuale del mercato dei principali attori, insieme ad approfondimenti sulle tendenze di mercato prevalenti e sulle opportunità emergenti.
Scopo del rapporto
Il rapporto Specimen Validity Testing Market di The Insight Partners mira a descrivere il panorama attuale e la crescita futura, i principali fattori trainanti, le sfide e le opportunità. Ciò fornirà spunti di riflessione a diversi stakeholder aziendali, tra cui:
- Fornitori/produttori di tecnologia: per comprendere le dinamiche di mercato in evoluzione e conoscere le potenziali opportunità di crescita, consentendo loro di prendere decisioni strategiche informate.
- Investitori: per condurre un'analisi completa delle tendenze in merito al tasso di crescita del mercato, alle proiezioni finanziarie del mercato e alle opportunità esistenti lungo la catena del valore.
- Enti di regolamentazione: regolamentano le politiche e le attività di controllo sul mercato con l'obiettivo di ridurre al minimo gli abusi, preservare la fiducia degli investitori e sostenere l'integrità e la stabilità del mercato.
Segmentazione del mercato dei test di validità dei campioni Prodotto
- Reagenti e kit di analisi
- Calibratori e controlli
- Monouso
Tipo
- Test di laboratorio
- Test rapidi/PoC
Utente finale
- Centri di gestione del dolore
- Luoghi di lavoro
- Agenzie di giustizia penale e forze dell'ordine
- Centri di riabilitazione dalla droga
- Laboratori di screening antidroga
Personalizza questo report in base alle tue esigenze
Ottieni la PERSONALIZZAZIONE GRATUITAMercato dei test di validità dei campioni: approfondimenti strategici
-
Scopri le principali tendenze di mercato di questo rapporto.Questo campione GRATUITO includerà analisi dei dati, che spaziano dalle tendenze di mercato alle stime e alle previsioni.
Fattori trainanti della crescita del mercato dei test di validità dei campioni
- Garantire la sicurezza: l'ascesa dei test di validità dei campioni in ambito sanitario
- Fiducia e trasparenza: aumentare la fiducia nei risultati dei test antidroga
- Innovazioni nei test: progressi che guidano la crescita e l'adozione del mercato
Tendenze future del mercato dei test di validità dei campioni
- Test di validità dei campioni: adottare l'intelligenza artificiale per una maggiore accuratezza
- Le soluzioni per i test a distanza rivoluzionano le pratiche di screening antidroga
- Crescente domanda di risultati immediati nei test antidroga sul posto di lavoro
Opportunità di mercato per i test di validità dei campioni
- Crescente richiesta di test antidroga accurati nei luoghi di lavoro
- Soluzioni innovative per test di validità dei campioni migliorati
- La crescente consapevolezza dell'abuso di sostanze stimola l'espansione del mercato dei test
Approfondimenti regionali sul mercato dei test di validità dei campioni
Le tendenze regionali e i fattori che influenzano il mercato dei test di validità dei campioni durante il periodo di previsione sono stati ampiamente spiegati dagli analisti di The Insight Partners. Questa sezione illustra anche i segmenti e la geografia del mercato dei test di validità dei campioni in Nord America, Europa, Asia-Pacifico, Medio Oriente e Africa, America Meridionale e Centrale.
Ambito del rapporto di mercato sui test di validità dei campioni
| Attributo del report | Dettagli |
|---|---|
| Dimensioni del mercato nel 2024 | XX miliardi di dollari USA |
| Dimensioni del mercato entro il 2031 | 5,03 miliardi di dollari USA |
| CAGR globale (2025 - 2031) | 6,8% |
| Dati storici | 2021-2023 |
| Periodo di previsione | 2025-2031 |
| Segmenti coperti |
Per prodotto
|
| Regioni e paesi coperti |
America del Nord
|
| Leader di mercato e profili aziendali chiave |
|
Densità degli attori del mercato dei test di validità dei campioni: comprendere il suo impatto sulle dinamiche aziendali
Il mercato dei test di validità dei campioni è in rapida crescita, trainato dalla crescente domanda degli utenti finali, dovuta a fattori quali l'evoluzione delle preferenze dei consumatori, i progressi tecnologici e una maggiore consapevolezza dei benefici del prodotto. Con l'aumento della domanda, le aziende stanno ampliando la propria offerta, innovando per soddisfare le esigenze dei consumatori e sfruttando le tendenze emergenti, alimentando ulteriormente la crescita del mercato.
- Ottieni una panoramica dei principali attori del mercato dei test di validità dei campioni
Punti di forza chiave
- Copertura completa: il rapporto copre in modo completo l'analisi di prodotti, servizi, tipologie e utenti finali del mercato dei test di validità dei campioni, fornendo un panorama olistico.
- Analisi degli esperti: il rapporto è redatto sulla base della conoscenza approfondita di esperti e analisti del settore.
- Informazioni aggiornate: il rapporto garantisce la pertinenza aziendale grazie alla copertura delle informazioni più recenti e delle tendenze dei dati.
- Opzioni di personalizzazione: questo report può essere personalizzato per soddisfare le esigenze specifiche del cliente e adattarsi in modo appropriato alle strategie aziendali.
Il rapporto di ricerca sul mercato dei test di validità dei campioni può quindi contribuire a tracciare un percorso di decodificazione e comprensione dello scenario del settore e delle prospettive di crescita. Sebbene possano esserci alcune valide preoccupazioni, i vantaggi complessivi di questo rapporto tendono a superare gli svantaggi.
- Analisi storica (2 anni), anno base, previsione (7 anni) con CAGR
- Analisi PEST e SWOT
- Valore/volume delle dimensioni del mercato - Globale, Regionale, Nazionale
- Industria e panorama competitivo
- Set di dati Excel
Report recenti
Rapporti correlati
Testimonianze
Motivo dell'acquisto
- Processo decisionale informato
- Comprensione delle dinamiche di mercato
- Analisi competitiva
- Analisi dei clienti
- Previsioni di mercato
- Mitigazione del rischio
- Pianificazione strategica
- Giustificazione degli investimenti
- Identificazione dei mercati emergenti
- Miglioramento delle strategie di marketing
- Aumento dell'efficienza operativa
- Allineamento alle tendenze normative






















 Ottieni un campione gratuito per - Mercato dei test di validità dei campioni
Ottieni un campione gratuito per - Mercato dei test di validità dei campioni