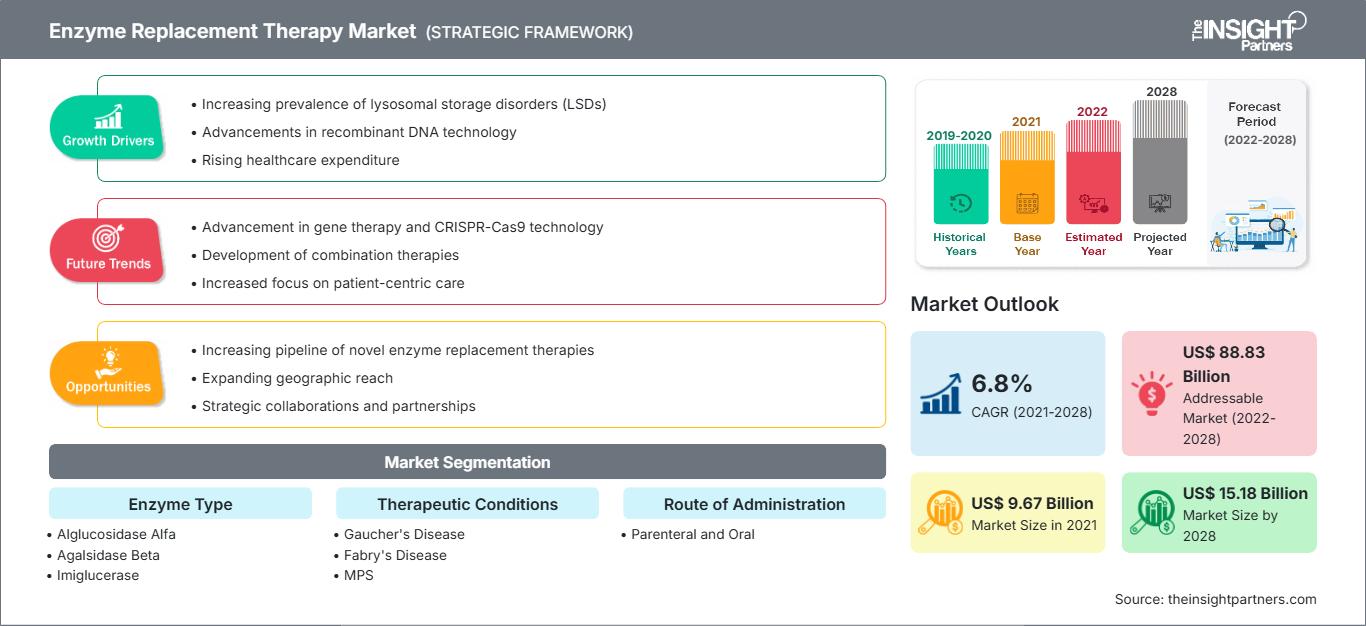
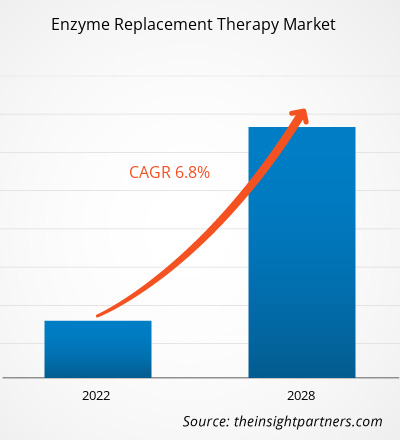
효소 대체 요법 시장은 2021년 96억 7,339만 달러에서 2028년 151억 8,470만 달러로 성장할 것으로 예상되며, 2022년부터 2028년까지 연평균 성장률 6.8%로 성장할 것으로 추산됩니다.
효소 대체 요법(ERT)은 효소 활성을 가진 효소 또는 단백질을 이용하여 선천성 효소 결핍증을 치료하는 요법입니다. 동물, 인간, 그리고 재조합 효소는 치료에 필요한 효소 공급원 중 일부입니다. 이 요법은 일반적으로 폼페병, 파브리병, 고셰병, 테이-삭스병, 헐러 증후군을 포함한 희귀 질환 및 리소좀 축적 질환에 적용됩니다. 가장 일반적인 ERT 방법은 정맥 주입으로, 조절된 수액을 통해 대체 효소를 혈류로 직접 투여하는 방식입니다. 글로벌 효소 대체 요법 시장의 성장은 리소좀 축적 질환(LSD)의 유병률 증가와 희귀의약품 지정 약물에 대한 신속한 규제 승인 및 기타 마케팅 이점 덕분입니다. 그러나 시장 참여자들 간의 치열한 경쟁은 시장 성장을 저해하고 있습니다.
이 보고서는 시장 동향, 기술 발전, 시장 역학, 그리고 세계 주요 시장 참여자들의 경쟁 환경 분석을 포함한 다양한 요소를 강조하며 글로벌 효소 대체 요법 시장에 대한 통찰력과 심층 분석을 제공합니다. 또한 COVID-19 팬데믹이 모든 지역의 시장에 미친 영향도 다룹니다. COVID-19 팬데믹으로 인해 많은 보건 당국이 팬데믹 관련 치료에 집중했습니다. 더욱이, 2020년 3월에는 COVID-19 환자들의 부담을 감당할 병원 서비스의 부족으로 효소 대체 요법이 필요한 많은 환자들이 수혈을 받지 못했습니다. 효소 대체 요법(ERT)이 필요한 환자들은 입원 중이나 약물 공급과 관련하여 불안감을 호소했습니다. 또한, 일부 환자들은 우울증과 불안감 증가를 경험한 것으로 나타났습니다. 따라서 COVID-19 팬데믹 동안 예정된 효소 대체 요법(ERT) 세션이 중단되어 시장에 부정적인 영향을 미쳤습니다.
요구 사항에 맞게 이 보고서를 사용자 정의하십시오.
이 보고서의 일부, 국가 수준 분석, Excel 데이터 팩을 포함하여 모든 보고서에 대한 사용자 정의를 무료로 받을 수 있을 뿐만 아니라 스타트업 및 대학을 위한 훌륭한 제안 및 할인을 이용할 수 있습니다
효소 대체 요법 시장: 전략적 통찰력
-
이 보고서의 주요 주요 시장 동향을 확인하세요.이 무료 샘플에는 시장 동향부터 추정 및 예측에 이르기까지 데이터 분석이 포함됩니다.
지리 기반 통찰력
지리적으로 볼 때, 글로벌 효소 대체 요법 시장은 북미(미국, 캐나다, 멕시코), 유럽(프랑스, 독일, 영국, 스페인, 이탈리아, 기타 유럽), 아시아 태평양(중국, 인도, 일본, 호주, 한국, 기타 APAC), 중동 및 아프리카(사우디 아라비아, UAE, 남아프리카, 기타 MEA), 남미 및 중미(브라질, 아르헨티나, 기타 남미 및 중미)로 구분됩니다.
시장 통찰력
리소좀 저장 질환(LSD) 유병률 증가
리소좀은 소화 효소를 함유한 막으로 둘러싸인 세포 소기관입니다. 리소좀은 거대분자 분해, 재활용, 신호전달의 핵심 세포 허브입니다. 이러한 기능에 결함이 발생하면 거대분자가 리소좀에 축적되거나 저장되어 세포 손상을 초래합니다. 리소좀 축적 질환(LSD)의 주요 원인은 리소좀 효소를 암호화하는 유전자의 돌연변이입니다. 또한, LSD는 효소의 결핍이나 결핍을 초래하는 선천적 대사 이상으로 정의됩니다. 유아와 어린이는 부모 중 한 명 또는 양쪽으로부터 결함 유전자를 물려받을 수 있기 때문에 성인보다 더 심각한 고통을 겪습니다. 그러나 지난 10년 동안 이러한 양상은 변화해 왔으며, LSD는 성인에게 더 흔하게 발생합니다. 또한, LSD는 고셔병, 니만-픽병, 파브리병, 테이-삭스병, 점액다당증(MPS), 폼페병을 포함하여 약 70가지의 질병을 일컫습니다. 이러한 질환들은 개별적으로는 드물지만, 전체적으로는 상당히 흔하며 출생아 5,000명 중 1명에게 영향을 미칩니다. 많은 국가에서 유병률 패턴을 파악하기 위해 설문조사를 실시했으며, 전체 유병률은 증가하고 있었습니다. 예를 들어, 랜싯 지역 보건(The Lancet Regional Health)에 발표된 "2009년부터 2020년까지 호주의 리소좀 저장 장애 유병률"이라는 제목의 연구가 있습니다. 연구에서는 다음과 같은 결과가 밝혀졌습니다.
- 호주 인구의 유병률은 1.6배(4,800명의 출생아당 1명)로, 1996년에 보고된 유병률(7,700명의 출생아당 1명)보다 높습니다.
- 파브리병이 가장 흔했으며, 모든 진단의 34%를 차지했습니다(2020년까지).
- LSD는 소아보다 성인에게 더 흔합니다.
따라서 LSD의 유병률 증가와 질병을 치료하는 제한된 치료법이 글로벌 효소 대체 요법 시장의 성장을 촉진하고 있습니다.
효소 유형 기반 통찰력
효소 유형을 기준으로 글로벌 효소 대체 요법 시장은 알글루코시다제 알파, 아갈시다제 베타, 이미글루세라제, 이두르설파제, 갈설파제, 벨라글루세라제 알파 및 기타 효소로 세분화됩니다. 2021년에는 다른 효소 부문이 가장 큰 시장 점유율을 차지했습니다. 이 부문은 2022년부터 2028년까지 시장에서 가장 높은 CAGR을 기록할 것으로 예상됩니다.
치료 질환 기반 통찰력
치료 질환을 기준으로 전 세계 효소 대체 요법 시장은 고셔병, 파브리병, 폼페병, 다발성 경화증(SCID), MPS 및 기타 치료 질환으로 구분됩니다. 고셔병 부문은 2021년에 가장 큰 시장 점유율을 기록했으며, 예측 기간 동안 가장 높은 CAGR을 기록할 것으로 예상됩니다.
투여 경로 기반 통찰력
투여 경로를 기준으로 전 세계 효소 대체 요법 시장은 비경구 투여와 경구 투여로 구분됩니다. 비경구 부문은 2021년에 더 큰 시장 점유율을 차지했으며 예측 기간 동안 더 높은 CAGR을 기록할 것으로 예상됩니다.
효소 대체 요법 시장The Insight Partners의 분석가들은 예측 기간 동안 효소 대체 요법 시장에 영향을 미치는 지역별 동향과 요인을 면밀히 분석했습니다. 이 섹션에서는 북미, 유럽, 아시아 태평양, 중동 및 아프리카, 그리고 중남미 지역의 효소 대체 요법 시장 부문 및 지역별 현황도 살펴봅니다.
효소 대체 요법 시장 보고서 범위
| 보고서 속성 | 세부 |
|---|---|
| 시장 규모 2021 | US$ 9.67 Billion |
| 시장규모별 2028 | US$ 15.18 Billion |
| 글로벌 CAGR (2021 - 2028) | 6.8% |
| 이전 데이터 | 2019-2020 |
| 예측 기간 | 2022-2028 |
| 다루는 세그먼트 |
By 효소 유형
|
| 포함된 지역 및 국가 |
북미
|
| 시장 선도 기업 및 주요 회사 프로필 |
|
효소 대체 요법 시장 참여자 밀도: 비즈니스 역학에 미치는 영향 이해
효소 대체 요법 시장은 소비자 선호도 변화, 기술 발전, 그리고 제품 효능에 대한 인식 제고 등의 요인으로 인한 최종 사용자 수요 증가에 힘입어 빠르게 성장하고 있습니다. 수요가 증가함에 따라 기업들은 제품 라인업을 확장하고, 소비자 니즈를 충족하기 위한 혁신을 추진하며, 새로운 트렌드를 적극 활용하고 있으며, 이는 시장 성장을 더욱 가속화하고 있습니다.
- 을 얻으세요 효소 대체 요법 시장 주요 주요 플레이어 개요
최종 사용자 기반 인사이트
최종 사용자를 기준으로 전 세계 효소 대체 요법 시장은 병원, 주입 센터 등으로 구분됩니다. 병원 부문은 2021년 가장 큰 시장 점유율을 기록했습니다. 그러나 주입 센터 부문은 예측 기간 동안 가장 높은 CAGR을 기록할 것으로 예상됩니다.
전 세계 효소 대체 요법 시장 참여자들은 제품 출시 및 확장을 포함한 유기적 전략을 통해 전 세계적으로 사업 영역과 제품 포트폴리오를 확장하고 증가하는 수요를 충족합니다. 이 시장에 기여하는 주요 기업으로는 다케다 제약(Takeda Pharmaceutical Company Limited), 사노피(Sanofi SA), 애브비(AbbVie Inc.), 바이오마린 제약(BioMarin Pharmaceutical Inc.), 아미쿠스 테라퓨틱스(Amicus Therapeutics), 알렉시온 제약(Alexion Pharmaceuticals, Inc., AstraZeneca), 얀센 제약(Janssen Pharmaceuticals, Inc., Johnson & Johnson Services, Inc.), 레코르다티(Recordati SpA), 화이자(Pfizer Inc.), 키에시 파마슈티치(CHIESI Farmaceutici SpA) 등이 있습니다.
- 과거 분석(2년), 기준 연도, CAGR을 포함한 예측(7년)
- PEST 및 SWOT 분석
- 시장 규모 가치/거래량 - 글로벌, 지역, 국가
- 산업 및 경쟁 환경
- Excel 데이터세트
사용 후기
구매 이유
- 정보에 기반한 의사 결정
- 시장 역학 이해
- 경쟁 분석
- 고객 인사이트
- 시장 예측
- 위험 완화
- 전략 기획
- 투자 타당성 분석
- 신흥 시장 파악
- 마케팅 전략 강화
- 운영 효율성 향상
- 규제 동향에 발맞춰 대응






















 무료 샘플 받기 - 효소 대체 요법 시장
무료 샘플 받기 - 효소 대체 요법 시장