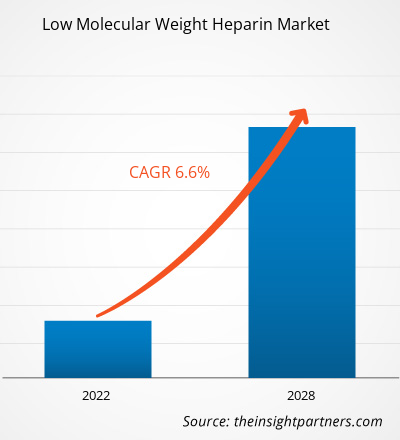
The low molecular weight heparin market is expected to grow from US$ 3,658.27 million in 2021 to US$ 5,733.96 million by 2028; it is estimated to grow at a CAGR of 6.6% from 2021 to 2028.
Low molecular weight heparin (LMWH) is a class of anticoagulants. They are used to prevent blood clots and to treat venous thromboembolism (deep vein thrombosis and pulmonary embolism) and treat myocardial infarction. LMWHs are defined as heparin salts with an average molecular weight of less than 8,000 Da and in which at least 60% of all chains have a molecular weight of less than 8,000 Da. These are obtained by various methods of fractionation or depolymerization of polymeric heparin. Low molecular weight heparin offers advantages over heparin in that it has better bioavailability and longer half-life, simplified dosing, predictable anticoagulant response, lower risk of HIT, and lower risk of osteoporosis. With a longer half-life and a more predictable anticoagulant response, LMWH can be administered subcutaneously once or twice daily without coagulation monitoring, even when the drug is administered in treatment doses. Most patients do not require coagulation monitoring with LMWH. Key factors driving the growth of the market are the advantages of low molecular weight heparin over unfractionated heparin and the introduction of LMWH anticoagulants across the globe. However, the adverse effects of low molecular weight heparin during treatment hinder the market growth.
Customize This Report To Suit Your Requirement
You will get customization on any report - free of charge - including parts of this report, or country-level analysis, Excel Data pack, as well as avail great offers and discounts for start-ups & universities
Low Molecular Weight Heparin Market: Strategic Insights

- Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
You will get customization on any report - free of charge - including parts of this report, or country-level analysis, Excel Data pack, as well as avail great offers and discounts for start-ups & universities
Low Molecular Weight Heparin Market: Strategic Insights

- Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
The low molecular weight heparin market, by region, is segmented into North America, Europe, Asia Pacific (APAC), the Middle East and Africa (MEA), and South and Central America (SAM). North America held the largest market share in 2021. The market in the region is expected to grow at the fastest pace during the forecast period. Major factors such as the growing burden of diseases, coupled with the rising geriatric population, and the rising number of approvals drive the market growth in the US. According to the Population Reference Bureau Factsheet, the number of Americans aged 65 years and above is projected to grow from 52 million in 2018 to 95 million by 2060, and the 65-and-above age group’s share of the total population is likely to rise from 16% to 23%. The geriatric population is more prone to chronic diseases. Therefore, the heparin market would grow significantly during the forecast period.
Market Insights
Advantages of Low Molecular Weight Heparin over Unfractionated Heparin
Low molecular weight heparins (LMWH) can be subcutaneously administered once a day without the need for any supervision. However, the effects of unfractionated heparin (UFH) or standard heparin vary from patient to patient. Therefore, it can only be administered to hospitalized patients under supervision. In addition, the LMWH has more predictable pharmacokinetic properties compared to unfractionated heparin, which allows its administration in fixed doses based on laboratory monitoring, without the need for dose adjustments.
The low molecular weight fractions of heparin have the molecular weight of ~3,500–8,000 Daltons, while unfractionated heparin has a molecular weight of 15,000 Daltons; thus, the former react less with platelets than the latter. Moreover, the LMWH is less likely to induce immune allergic thrombocytopenia, which is a severe side effect of unfractionated heparin (UFH). This condition is often complicated by arterial thrombosis. In addition, properties such as better bioavailability, predictable dose-response, and longer plasma half-life than unfractionated heparin make low molecular weight heparin a preferred choice for anticoagulation therapy. Such advantages of the LMWH over unfractionated heparin are boosting its demand, thereby spurring market growth.
Product Type-Based Insights
The global low molecular weight heparin market, based on product type, is segmented into enoxaparin, dalteparin, tinzaparin, fraxiparine, nadroparin, and others. The enoxaparin segment held the largest share of the market in 2021. However, the dalteparin segment is anticipated to register the highest CAGR in the market during the forecast period.
Packaging-Based Insights
The global low molecular weight heparin market, based on packaging, is bifurcated into multi-vials and prefilled syringes. In 2021, the multi-vials segment held a larger share of the market. However, the prefilled syringes segment is expected to register a higher CAGR during 2021–2028.
Application-Based Insights
The global low molecular weight heparin market, based on application, is segmented into deep vein thrombosis, acute coronary syndrome (ACS), pulmonary embolism, and others. The deep vein thrombosis segment held the largest share of the market in 2021. However, the acute coronary syndrome (ACS) segment is anticipated to register the highest CAGR in the market during the forecast period.
End Use-Based Insights
The global low molecular weight heparin market, based on end user, has been segmented into hospitals, clinics, ambulatory surgical centers, and others. The hospitals segment held the largest share of the market in 2021. However, the ambulatory surgical centers segment is anticipated to register the highest CAGR in the market during the forecast period.
The low molecular weight heparin market players adopt organic strategies, such as product launch and expansion, to expand their footprint and product portfolio worldwide and to meet the growing demand.
Market by Geography
Geographically, the low molecular weight heparin market is segmented into North America (the US, Canada, and Mexico), Europe (France, Germany, the UK, Spain, Italy, and the Rest of Europe), Asia Pacific (China, India, Japan, Australia, South Korea, and the Rest of APAC), Middle East & Africa (Saudi Arabia, the UAE, South Africa, and the Rest of MEA), and South and Central America (Brazil, Argentina, and the Rest of SCAM).
Low Molecular Weight Heparin Market Regional Insights
The regional trends and factors influencing the Low Molecular Weight Heparin Market throughout the forecast period have been thoroughly explained by the analysts at Insight Partners. This section also discusses Low Molecular Weight Heparin Market segments and geography across North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America.
- Get the Regional Specific Data for Low Molecular Weight Heparin Market
Low Molecular Weight Heparin Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2021 | US$ 3.66 Billion |
| Market Size by 2028 | US$ 5.73 Billion |
| Global CAGR (2021 - 2028) | 6.6% |
| Historical Data | 2019-2020 |
| Forecast period | 2022-2028 |
| Segments Covered |
By Product Type
|
| Regions and Countries Covered | North America
|
| Market leaders and key company profiles |
Low Molecular Weight Heparin Market Players Density: Understanding Its Impact on Business Dynamics
The Low Molecular Weight Heparin Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
Market players density refers to the distribution of firms or companies operating within a particular market or industry. It indicates how many competitors (market players) are present in a given market space relative to its size or total market value.
Major Companies operating in the Low Molecular Weight Heparin Market are:
- Aspen Holdings
- Changzhou Qianhong Bio-pharma
- LEOPHARMA A/S
- Teva Pharmaceutical Industries Ltd.
- B. Braun Medical Inc.
Disclaimer: The companies listed above are not ranked in any particular order.
- Get the Low Molecular Weight Heparin Market top key players overview
COVID Impacts
The spread of the COVID-19 pandemic has emphasized health data sharing and interoperability under the microscope. The infection of novel coronavirus in children is mild compared to adults, and children are assigned a better prognosis. The interruptions in the supply chain and the massive demand for efficient treatments for COVID-19 have put the health research industry in Asia-Pacific in a crucial position.
Company Profiles
- Aspen Holdings
- Changzhou Qianhong Bio-Pharma
- Leopharma A/S
- Teva Pharmaceutical Industries Ltd.
- B. Braun Medical, Inc.
- Pfizer, Inc.
- Sanofi
- Dr. Reddy's Laboratories Limited
- Abbott
- Novartis AG
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Global, Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Testimonials
I wish to appreciate your support and the professionalism you displayed in the course of attending to my request for information regarding to infectious disease IVD market in Nigeria. I appreciate your patience, your guidance, and the fact that you were willing to offer a discount, which eventually made it possible for us to close a deal. I look forward to engaging The Insight Partners in the future, all thanks to the impression you have created in me as a result of this first encounter.
DR CHIJIOKE ONYIA, MANAGING DIRECTOR, PineCrest Healthcare Ltd.The Insight Partners delivered insightful, well-structured market research with strong domain expertise. Their team was professional and responsive throughout. The user-friendly website made accessing industry reports seamless. We highly recommend them for reliable, high-quality research services
Yukihiko Adachi CEO, Deep Blue, LLC.Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Strategic Planning
- Investment Justification
- Identifying Emerging Markets
- Enhancing Marketing Strategies
- Boosting Operational Efficiency
- Tracking Industry Innovations
- Aligning with Regulatory Trends
Yes! We provide a free sample of the report, which includes Report Scope (Table of Contents), report structure, and selected insights to help you assess the value of the full report. Please click on the "Download Sample" button or contact us to receive your copy.
Absolutely — analyst assistance is part of the package. You can connect with our analyst post-purchase to clarify report insights, methodology or discuss how the findings apply to your business needs.
Once your order is successfully placed, you will receive a confirmation email along with your invoice.
• For published reports: You’ll receive access to the report within 4–6 working hours via a secured email sent to your email.
• For upcoming reports: Your order will be recorded as a pre-booking. Our team will share the estimated release date and keep you informed of any updates. As soon as the report is published, it will be delivered to your registered email.
We offer customization options to align the report with your specific objectives. Whether you need deeper insights into a particular region, industry segment, competitor analysis, or data cut, our research team can tailor the report accordingly. Please share your requirements with us, and we’ll be happy to provide a customized proposal or scope.
The report is available in either PDF format or as an Excel dataset, depending on the license you choose.
The PDF version provides the full analysis and visuals in a ready-to-read format. The Excel dataset includes all underlying data tables for easy manipulation and further analysis.
Please review the license options at checkout or contact us to confirm which formats are included with your purchase.
Our payment process is fully secure and PCI-DSS compliant.
We use trusted and encrypted payment gateways to ensure that all transactions are protected with industry-standard SSL encryption. Your payment details are never stored on our servers and are handled securely by certified third-party processors.
You can make your purchase with confidence, knowing your personal and financial information is safe with us.
Yes, we do offer special pricing for bulk purchases.
If you're interested in purchasing multiple reports, we’re happy to provide a customized bundle offer or volume-based discount tailored to your needs. Please contact our sales team with the list of reports you’re considering, and we’ll share a personalized quote.
Yes, absolutely.
Our team is available to help you make an informed decision. Whether you have questions about the report’s scope, methodology, customization options, or which license suits you best, we’re here to assist. Please reach out to us at sales@theinsightpartners.com, and one of our representatives will get in touch promptly.
Yes, a billing invoice will be automatically generated and sent to your registered email upon successful completion of your purchase.
If you need the invoice in a specific format or require additional details (such as company name, GST, or VAT information), feel free to contact us, and we’ll be happy to assist.
Yes, certainly.
If you encounter any difficulties accessing or receiving your report, our support team is ready to assist you. Simply reach out to us via email or live chat with your order information, and we’ll ensure the issue is resolved quickly so you can access your report without interruption.















The List of Companies - Low Molecular Weight Heparin Market
- Aspen Holdings
- Changzhou Qianhong Bio-pharma
- LEOPHARMA A/S
- Teva Pharmaceutical Industries Ltd.
- B. Braun Medical Inc.
- Pfizer Inc.
- Sanofi-Aventis
- Dr. Reddy's Laboratories Limited
- Abbott
- Novartis AG






 Get Free Sample For
Get Free Sample For