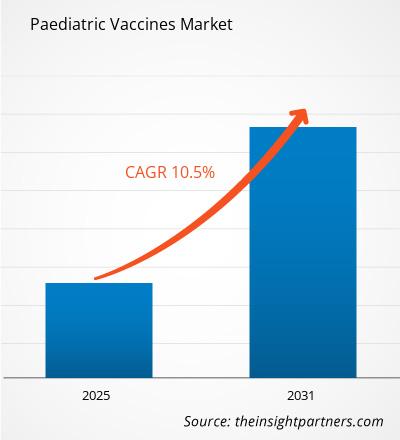
The Paediatric Vaccines Market is expected to register a CAGR of 10.5% from 2025 to 2031, with a market size expanding from US$ XX million in 2024 to US$ XX Million by 2031.
The report is segmented based on Type, Vaccine Form, Application and End User. The global analysis is further broken down at the regional level and major countries. The global analysis is broken down at the regional level and major countries. The market evaluation is presented in US$ for the above segmental analysis.
Purpose of the Report
The report Paediatric Vaccines Market by The Insight Partners aims to describe the present landscape and future growth, top driving factors, challenges, and opportunities. This will provide insights to various business stakeholders, such as:
- Technology Providers/Manufacturers: To understand the evolving market dynamics and know the potential growth opportunities, enabling them to make informed strategic decisions.
- Investors: To conduct a comprehensive trend analysis regarding the market growth rate, market financial projections, and opportunities that exist across the value chain.
- Regulatory bodies: To regulate policies and police activities in the market with the aim of minimizing abuse, preserving investor trust and confidence, and upholding the integrity and stability of the market.
Paediatric Vaccines Market Segmentation
Type
- Monovalent
- Multivalent
Vaccine Form
- Attenuated Live
- Viruses
- Inactivated Viruses
- Subunits
- Toxoids
- Recombinant
- Polysaccharides
- Conjugates
Application
- Infectious disease
- Allergy
End User
- Hospitals
- Pediatric Clinics
- Academic and Research
- Organizations
Customize This Report To Suit Your Requirement
You will get customization on any report - free of charge - including parts of this report, or country-level analysis, Excel Data pack, as well as avail great offers and discounts for start-ups & universities
Paediatric Vaccines Market: Strategic Insights

- Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
Paediatric Vaccines Market Growth Drivers
- Increasing Parental Awareness: Rising awareness about the importance of immunization and its role in preventing infectious diseases is driving demand for pediatric vaccines.
- Government Initiatives: Government initiatives and vaccination programs are boosting the adoption of pediatric vaccines, especially in developing countries.
- Emerging Diseases and Outbreaks: The threat of new diseases and outbreaks is fueling demand for preventive measures.
Paediatric Vaccines Market Future Trends
- Combination Vaccines: The development of combination vaccines that offer protection against multiple diseases in a single dose is a significant trend in the pediatric vaccine market.
- Advancements in Vaccine Technology: Continuous advancements in vaccine technology, such as mRNA vaccines and nanoparticle-based vaccines, are leading to the development of more effective and safer pediatric vaccines.
- Personalized Vaccine Strategies: Tailored vaccination regimens based on individual risk factors and genetic profiles.
Paediatric Vaccines Market Opportunities
- Expansion into Emerging Markets: Emerging markets with large populations and increasing healthcare expenditure offer significant growth opportunities for pediatric vaccine manufacturers.
- Development of Novel Vaccines: The development of vaccines for emerging infectious diseases and neglected tropical diseases can create new market opportunities.
- Strategic Partnerships with Healthcare Providers: Collaborating with healthcare providers can improve vaccine delivery and compliance.
Paediatric Vaccines Market Regional Insights
The regional trends and factors influencing the Paediatric Vaccines Market throughout the forecast period have been thoroughly explained by the analysts at Insight Partners. This section also discusses Paediatric Vaccines Market segments and geography across North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America.
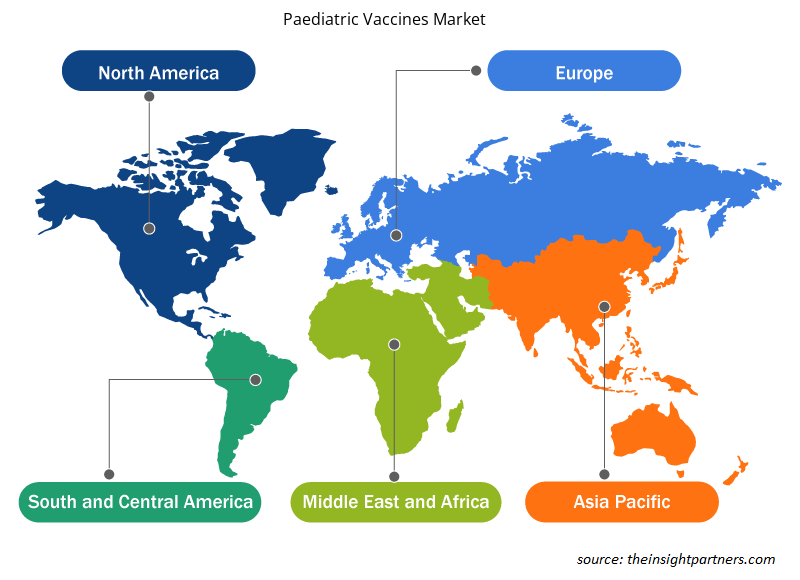
- Get the Regional Specific Data for Paediatric Vaccines Market
Paediatric Vaccines Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2024 | US$ XX million |
| Market Size by 2031 | US$ XX Million |
| Global CAGR (2025 - 2031) | 10.5% |
| Historical Data | 2021-2023 |
| Forecast period | 2025-2031 |
| Segments Covered |
By Type
|
| Regions and Countries Covered | North America
|
| Market leaders and key company profiles |
Paediatric Vaccines Market Players Density: Understanding Its Impact on Business Dynamics
The Paediatric Vaccines Market market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
Market players density refers to the distribution of firms or companies operating within a particular market or industry. It indicates how many competitors (market players) are present in a given market space relative to its size or total market value.
Major Companies operating in the Paediatric Vaccines Market are:
- Pfizer Inc.
- Glaxosmithkline, Plc
- Merck Sharp and Dohme Corp
- Sanofi
- Panacea Biotec Ltd
Disclaimer: The companies listed above are not ranked in any particular order.
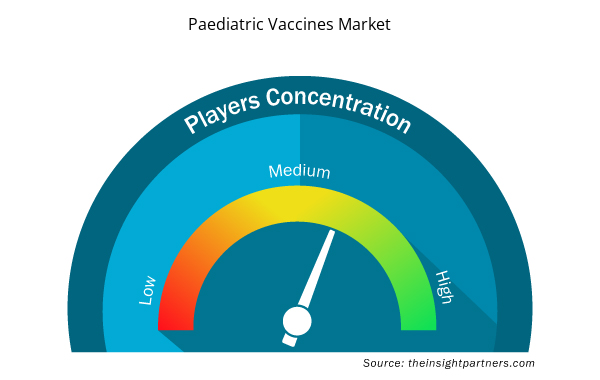
- Get the Paediatric Vaccines Market top key players overview
Key Selling Points
- Comprehensive Coverage: The report comprehensively covers the analysis of products, services, types, and end users of the Paediatric Vaccines Market, providing a holistic landscape.
- Expert Analysis: The report is compiled based on the in-depth understanding of industry experts and analysts.
- Up-to-date Information: The report assures business relevance due to its coverage of recent information and data trends.
- Customization Options: This report can be customized to cater to specific client requirements and suit the business strategies aptly.
The research report on the Paediatric Vaccines Market can, therefore, help spearhead the trail of decoding and understanding the industry scenario and growth prospects. Although there can be a few valid concerns, the overall benefits of this report tend to outweigh the disadvantages.
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Global, Regional, Country
- Industry and Competitive Landscape
- Excel Dataset

Report Coverage
Revenue forecast, Company Analysis, Industry landscape, Growth factors, and Trends
Segment Covered
This text is related
to segments covered.

Regional Scope
North America, Europe, Asia Pacific, Middle East & Africa, South & Central America
Country Scope
This text is related
to country scope.
Frequently Asked Questions
Some of the customization options available based on request are additional 3-5 company profiles and country-specific analysis of 3-5 countries of your choice. Customizations are to be requested/discussed before making final order confirmation, as our team would review the same and check the feasibility.
The report can be delivered in PDF/PPT format; we can also share excel dataset based on the request.
The leading players of the Paediatric Vaccines Market are: Cigna Global, GeoBlue Xplorer, IMG Global Medical Insurance, William Russell, Bupa Global, Now Health International, Integra Global, Allianz International Medical Insurance, AXA Global Healthcare, AIA Group
Paediatric Vaccines Market is expected to grow at a CAGR of 10.5% between 2023-2031
The future trends of the Paediatric Vaccines Market are: Combination Vaccines and Advancements in Vaccine Technology
The driving factors impacting the Paediatric Vaccines Market are: Increasing Parental Awareness and Government Initiatives
Trends and growth analysis reports related to Life Sciences : READ MORE..
- Pfizer Inc.
- Glaxosmithkline, Plc
- Merck Sharp and Dohme Corp
- Sanofi
- Panacea Biotec Ltd
- Sinovac Biotech
- Bio Med Pvt. Ltd.
- CSL Limited
- Serum Institute of India Pvt. Ltd
- Indian Immunologicals Ltd
- Grifols
- Emergent BioSolutions Inc
 Get Free Sample For
Get Free Sample For