The viral vector manufacturing market size is expected to reach US$ 5.76 billion by 2031 from US$ 1.74 billion in 2024. The market is anticipated to register a CAGR of 18.8% during 2025–2031.
Viral Vector Manufacturing Market Analysis
The viral vector manufacturing market is experiencing rapid expansion, driven by increasing demand for gene therapies and vaccines. Key trends include technological advancements, increased investment, and strategic collaborations. Challenges include high production costs, scalability issues, and strict regulatory requirements.
Viral Vector Manufacturing Market Overview
Viral vectors are engineered viruses used to deliver genetic material in gene and cell therapies, vaccines, and research. Manufacturing involves design, cell growth, purification, quality control, and GMP-compliant formulation to ensure safety, potency, and stability.
Customize Research To Suit Your Requirement
We can optimize and tailor the analysis and scope which is unmet through our standard offerings. This flexibility will help you gain the exact information needed for your business planning and decision making.
Viral Vector Manufacturing Market: Strategic Insights
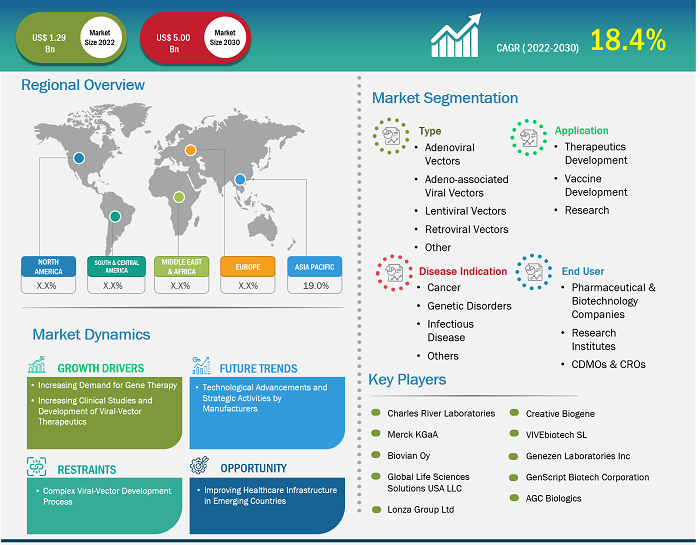
Market Size Value in US$ 1.29 billion in 2022 Market Size Value by US$ 5.00 billion by 2030 Growth rate CAGR of 18.4% from 2022 to 2030 Forecast Period 2022-2030 Base Year 2022

Mrinal
Have a question?
Mrinal will walk you through a 15-minute call to present the report’s content and answer all queries if you have any.
 Speak to Analyst
Viral Vector Manufacturing Market Drivers and Opportunities
Speak to Analyst
Viral Vector Manufacturing Market Drivers and Opportunities
Customize Research To Suit Your Requirement
We can optimize and tailor the analysis and scope which is unmet through our standard offerings. This flexibility will help you gain the exact information needed for your business planning and decision making.
Viral Vector Manufacturing Market: Strategic Insights

| Market Size Value in | US$ 1.29 billion in 2022 |
| Market Size Value by | US$ 5.00 billion by 2030 |
| Growth rate | CAGR of 18.4% from 2022 to 2030 |
| Forecast Period | 2022-2030 |
| Base Year | 2022 |

Mrinal
Have a question?
Mrinal will walk you through a 15-minute call to present the report’s content and answer all queries if you have any.
 Speak to Analyst
Speak to Analyst
Market Drivers:
Rising Pipeline of Gene and Cell Therapies:
The proliferation of gene therapy, cell therapy, and advanced therapeutic programs increases demand for GMP-grade viral vectors (AAV, lentivirus, etc.).Investment & Funding in Biomanufacturing Infrastructure:
Governments, venture capital, and biopharma firms are funding facility expansions, technology upgrades, and CDMO scale‑ups.Technological Innovation:
Use of single‑use bioreactors, closed systems, in‑line analytics, and modular platforms helps reduce contamination risk and improve yield/throughput.Outsourcing to CDMOs / Specialized Service Providers:
Many biotech and pharma players prefer to outsource vector manufacturing rather than build in-house capacity. This trend supports CDMO growth.Regulatory Support & Streamlining for Advanced Therapies:
Some regulatory bodies are introducing clearer guidelines for gene therapy and viral vector products, reducing uncertainty.
Market Opportunities:
Expansion into Gene Therapy and Vaccine Development:
With the increasing number of gene therapies and viral vector vaccine pipelines, the demand for specialized manufacturing is expected to rise, leading to opportunities for developing customized and scalable vector production.Increasing Demand in Rare Genetic Disorders and Cancer:
With an increasing number of approvals and clinical trials for gene therapies targeting rare disorders and cancers, the demand for high-quality viral vectors is rising, expanding manufacturing requirements, and driving market growth across various therapeutic areas.Advancements in Manufacturing Technologies:
Advancements in manufacturing technologies, including single-use bioreactors and continuous processing, maximize yields per batch and minimize contamination risks and manufacturing costs.Customizable and Scalable Manufacturing Options:
Scale and grow, making them appealing to both start-ups and large pharmaceutical companies, resulting in improved global market expansion.
Viral Vector Manufacturing Market Report Segmentation Analysis
The viral vector manufacturing market is categorized into distinct segments to understand its structure, growth prospects, and emerging trends. Below is the standard segmentation approach used in industry reports:
By Type:
Adenoviral Vectors:
Non-integrating vectors delivering large genes; cause strong immune responses; used in gene therapy and vaccines.Adeno-Associated Viral Vectors:
It is nonpathogenic, capable of site-specific integration, providing long-term gene expression with low immunogenicity, ideal for treating nondividing cells in gene therapy.Lentiviral Vectors:
They efficiently integrate into both dividing and nondividing cells’ genomes, allowing stable, long-term gene expression, commonly used in research and clinical gene therapy applications.Retroviral Vectors:
They integrate into the genome of dividing cells only, enabling stable gene transfer, but with limited targeting scope and potential insertional mutagenesis risks.Others:
Other viral vectors, such as herpesvirus and vaccinia virus, offer unique tissue targeting and larger genetic payload capacities, suited for specialized gene therapy and vaccine strategies.
By Technology:
Cancer:
Viral vectors induce targeted therapies and immunotherapies in the treatment of different cancers. This approach can potentially yield more specific treatments and better patient outcomes.Genetic Disorders:
Viral vectors are used to deliver gene therapy approaches that correct defective genes and effectively treat inherited genetic disorders.Infectious Diseases:
Viral vectors are leveraged in vaccine development and the delivery of therapeutics to address infectious diseases such as HIV, COVID-19, and hepatitis.Others:
Includes indications, including neurological, cardiovascular, and rare diseases, where viral vectors will contribute to gene delivery and therapeutic interventions.
By Application:
- Therapeutics Development
- Vaccine Development
- Research
By End User:
- Pharmaceutical and Biotechnology Companies
- CDMOs & CROs
- Research Institutes
By Geography:
- North America
- Europe
- Asia Pacific
- South & Central America
- Middle East & Africa
Market Report ScopeViral Vector Manufacturing Market Share Analysis by Geography
The viral vector manufacturing market in Asia Pacific is experiencing rapid growth driven by rising healthcare investments, technological advancements, and increasing demand for gene therapies and personalized medicine. Emerging markets in South & Central America, the Middle East, and Africa present untapped opportunities for viral vector manufacturing, enabling healthcare providers to expand access to advanced treatments and improve patient outcomes in these regions.
The viral vector manufacturing market growth differs in each region due to differences in healthcare infrastructure, regulatory environments, funding availability, technological adoption, and disease prevalence. Below is a summary of market share and trends by region:
1. North America
Market Share:
Holds a significant portion of the global marketKey Drivers:
- Advanced healthcare infrastructure: Advanced healthcare systems in North America support viral vector manufacturing by facilitating clinical trials, biomanufacturing, and regulatory compliance for gene therapies.
- Strong biotechnology and pharmaceutical ecosystem
- Robust funding and regulatory support for gene therapy development.
Trends:
Expansion of in-house manufacturing by biotech firms.
2. Europe
Market Share:
Substantial share owing to early, stringent EU regulationsKey Drivers:
- Focus on quality and safety in gene therapy production: Europe’s strict regulatory frameworks ensure high standards for viral vector production, accelerating trust and adoption in therapeutic applications.
- Government-backed initiatives for rare disease treatments
- Growing investments in research and development in academic and research institutions.
Trends:
Development of GMP-compliant viral vector facilities.
3. Asia Pacific
Market Share:
Fastest-growing region with dominant market shareKey Drivers:
- Expansion of biotech and pharma manufacturing hubs: Growing life sciences sector is driving demand for localized viral vector production to support clinical trials and commercialization.
- Government investments in precision and regenerative medicine
- Rising demand for affordable gene therapies in populous countries
Trends:
Surge in contract manufacturing organizations (CMOs).
4. Middle East and Africa
Market Share:
Although small, it is growing quicklyKey Drivers:
- National healthcare transformation
- Strategic national health plans promote biotech investments, including facilities for viral vectors to support future accessibility of gene therapy.
- Increasing collaborations with global biotech firms
Trends:
Early-stage infrastructure development and targeted investments in research hubs.
5. South & Central America
Market Share:
Growing market with steady progressKey Drivers:
- Investments in healthcare infrastructure and innovation
- Ongoing modernization of healthcare systems fuels demand for advanced, growing clinical trial activity
- Increased interest from private healthcare providers.
Trends:
Establishment of small-scale viral vector production units and pilot projects.
Viral Vector Manufacturing Market Players Density: Understanding Its Impact on Business Dynamics
High Market Density and Competition
Competition is intense due to the presence of major global players such as Lonza Group AG; Merck KGaA; Thermo Fisher Scientific Inc., Catalent Inc., Charles River Laboratories International Inc., WuXi AppTec Co Ltd; FUJIFILM Holdings Corp; GenScript Biotech Corporation; Takara Bio Inc., and Novartis AG
This high level of competition urges companies to stand out by offering:
- GMP-compliant facilities
- Advanced technologies such as single-use systems and automation
- Integrated end-to-end services
- Scalable and flexible manufacturing capabilities
- Scalable and customizable platforms to meet diverse needs.
Opportunities and Strategic Moves
- Leveraging AI and data analytics to optimize viral vector production processes, predict batch outcomes, and enhance quality control and yield efficiency.
- Enhanced Patient Safety: Implementing real-time monitoring systems to ensure viral vector purity, reduce contamination risks, and comply with stringent global regulatory standards.
Disclaimer: The companies listed above are not ranked in any particular order.
Other companies analyzed during the course of research:
- Addgene, Inc.
- Advanced BioScience Laboratories, Inc.
- Aldevron, L.L.C.
- AstraZeneca
- Audentes Therapeutics
- Batavia Biosciences B.V.
- BioMarin Pharmaceutical
- BioNTech IMFS GmbH
- Biovion Oy
- CEVEC Pharmaceuticals GmbH
- CGT Catapult
- Danaher
Viral Vector Manufacturing Market News and Recent Developments
Lonza acquires the Genentech large-scale biologics manufacturing site, March 2024:
Lonza, a global manufacturing partner to the pharmaceutical, biotech, and nutraceutical markets, announced the signing of an agreement to acquire the Genentech large-scale biologics manufacturing site in Vacaville, California (US) from Roche for US$1.2 billion.Siren Biotechnology entered a strategic partnership with Catalent Inc., May 2024:
Siren Biotechnology, pioneers of Universal AAV Immuno-Gene Therapy for Cancer, and Catalent Inc., the leader in enabling the development and supply of better treatments for patients worldwide, entered a strategic partnership to support the development and manufacturing of Siren Biotechnology’s AAV immuno-gene therapies.
Viral Vector Manufacturing Market Report Coverage and Deliverables
The "Viral Vector Manufacturing Market Size and Forecast (2021–2031)" report provides a detailed analysis of the market covering below areas:
- Viral Vector Manufacturing Market size and forecast at global, regional, and country levels for key market segments covered under the scope
- Viral Vector Manufacturing Market trends, as well as market dynamics such as drivers, restraints, and key opportunities
- Detailed PEST and SWOT analysis
- Real-time Location Systems Market For Healthcare analysis covering key market trends, global and regional framework, major players, regulations, and recent market developments
- Industry landscape and competition analysis covering market concentration, heat map analysis, prominent players, and recent developments for the viral vector manufacturing market
- Detailed company profiles

Report Coverage
Revenue forecast, Company Analysis, Industry landscape, Growth factors, and Trends
Segment Covered
Type, Disease Indication, Application, End User, and Geography

Regional Scope
North America, Europe, Asia Pacific, Middle East & Africa, South & Central America
Country Scope
This text is related
to country scope.
Frequently Asked Questions
Major players include Lonza Group AG, Merck KGaA, Thermo Fisher Scientific Inc., Catalent Inc., and Charles River Laboratories International Inc.
As of 2024, the global viral vector manufacturing market is valued at US$1.74 billion. It is projected to reach US$ 5.76 billion by 2031, growing at a compound annual growth rate (CAGR) of 18.8% during the forecast period from 2025 to 2031.
1. Investment & Funding in Biomanufacturing Infrastructure: Governments, venture capital, and biopharma firms are funding facility expansions, technology upgrades, and CDMO scale‑ups.2. Technological Innovation: Use of single‑use bioreactors, closed systems, in‑line analytics, and modular platforms helps reduce contamination risk and improve yield/throughput.
AI and ML are revolutionizing viral vector manufacturing for healthcare by:1. Process Optimization: AI and ML analyze vast datasets to optimize production parameters, improving yield and consistency.2. Personalized Manufacturing: AI supports customization of viral vectors for individual patients by analyzing genetic and clinical data.
While Asia Pacific and Europe currently dominate, North America, the Middle East & Africa, and South & Central America are expected to expand rapidly due to growing gene therapy demand and infrastructure investments.
The Adeno-Associated Viral Vectorssegment is experiencing significant growth in the viral vector manufacturing market by increasing gene therapy applications, superior safety profile, long-term expression, and strong demand for targeted and efficient delivery.
1. Scalability Issues: Scaling up viral vector production while maintaining quality and consistency is a challenging task.2. Batch Variability: Ensuring batch-to-batch consistency is challenging due to biological variability.
1. Increased adoption of gene and cell therapies2. Advancements in vector engineering3. Scale-up of manufacturing processes4. Automation and continuous manufacturing5. Use of novel viral vectors6. Improved purification technologies
1. Introduction
1.1 Report Guidance
1.2 Market Segmentation
2. Executive Summary
2.1 Analyst Market Outlook
2.2 Market Attractiveness
3. Research Methodology
3.1 Secondary Research
3.2 Primary Research
3.2.1 Hypothesis formulation:
3.2.2 Macro-economic factor analysis:
3.2.3 Developing base number:
3.2.4 Data Triangulation:
3.2.5 Country level data:
3.3 Assumptions and Limitations
4. Viral Vector Manufacturing Market Landscape
4.1 Overview
4.2 PEST Analysis
4.3 Ecosystem Analysis
4.3.1 Raw Material Suppliers:
4.3.2 Vector Design and Development:
4.3.3 Contract Development and Manufacturing Organizations (CDMOs):
4.3.4 End Users:
4.3.5 List of Vendors in the Value Chain
5. Viral Vector Manufacturing Market – Key Market Dynamics
5.1 Viral Vector Manufacturing Market – Key Market Dynamics
5.2 Market Drivers
5.2.1 Rising Demand for Gene and Cell Therapies
5.2.2 Growing Pipeline of Viral Vector-Based Clinical Trials
5.2.3 Rising Government and Private Investment in Advanced Therapies
5.3 Market Restraints
5.3.1 Sensitivity to Raw Material and Supply Chain Fluctuations
5.3.2 Limited Skilled Workforce and Infrastructure
5.4 Market Opportunities
5.4.1 Expansion of Gene Editing Technologies
5.4.2 Emergence of Viral Vector Manufacturing Outsourcing Services
5.4.3 Government Initiatives and Funding
5.5 Future Trends
5.5.1 Technological Advancements in Vector Manufacturing
5.5.2 Government Investments in Biotech Infrastructure
5.5.3 Growth in Personalized Medicine
5.6 Impact of Drivers and Restraints:
6. Viral Vector Manufacturing Market – Global Market Analysis
6.1 Viral Vector Manufacturing Market Revenue (US$ Million), 2021–2031
6.2 Viral Vector Manufacturing Market Forecast Analysis
7. Viral Vector Manufacturing Market Analysis – by Type
7.1 Adenoviral Vectors
7.1.1 Overview
7.1.2 Adenoviral Vectors: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
7.2 Adeno-Associated Viral Vectors
7.2.1 Overview
7.2.2 Adeno-Associated Viral Vectors: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
7.3 Lentiviral Vectors
7.3.1 Overview
7.3.2 Lentiviral Vectors: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
7.4 Retroviral Vectors
7.4.1 Overview
7.4.2 Retroviral Vectors: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
7.5 Others
7.5.1 Overview
7.5.2 Others: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
8. Viral Vector Manufacturing Market Analysis – by Disease Indication
8.1 Cancer
8.1.1 Overview
8.1.2 Cancer: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
8.2 Genetic Disorders
8.2.1 Overview
8.2.2 Genetic Disorders: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
8.3 Infectious Diseases
8.3.1 Overview
8.3.2 Infectious Diseases: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
8.4 Others
8.4.1 Overview
8.4.2 Others: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
9. Viral Vector Manufacturing Market Analysis – by Application
9.1 Therapeutics Development
9.1.1 Overview
9.1.2 Therapeutics Development: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
9.2 Vaccine Development
9.2.1 Overview
9.2.2 Vaccine Development: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
9.3 Research
9.3.1 Overview
9.3.2 Research: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
10. Viral Vector Manufacturing Market Analysis – by End User
10.1 Pharmaceutical and Biotechnology Companies
10.1.1 Overview
10.1.2 Pharmaceutical and Biotechnology Companies: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
10.2 CDMOs and CROs
10.2.1 Overview
10.2.2 CDMOs and CROs: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
10.3 Research Institutes
10.3.1 Overview
10.3.2 Research Institutes: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
11. Viral Vector Manufacturing Market – Geographical Analysis
11.1 Overview
11.2 North America
11.2.1 North America Viral Vector Manufacturing Market Overview
11.2.2 North America: Viral Vector Manufacturing Market – Revenue, 2021–2031 (US$ Million)
11.2.3 North America: Viral Vector Manufacturing Market Breakdown, by Type
11.2.3.1 North America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Type
11.2.4 North America: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.2.4.1 North America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Disease Indication
11.2.5 North America: Viral Vector Manufacturing Market Breakdown, by Application
11.2.5.1 North America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Application
11.2.6 North America: Viral Vector Manufacturing Market Breakdown, by End User
11.2.6.1 North America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by End User
11.2.7 North America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Country
11.2.7.1 North America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Country
11.2.7.2 United States: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.2.7.2.1 United States: Viral Vector Manufacturing Market Breakdown, by Type
11.2.7.2.2 United States: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.2.7.2.3 United States: Viral Vector Manufacturing Market Breakdown, by Application
11.2.7.2.4 United States: Viral Vector Manufacturing Market Breakdown, by End User
11.2.7.3 Canada: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.2.7.3.1 Canada: Viral Vector Manufacturing Market Breakdown, by Type
11.2.7.3.2 Canada: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.2.7.3.3 Canada: Viral Vector Manufacturing Market Breakdown, by Application
11.2.7.3.4 Canada: Viral Vector Manufacturing Market Breakdown, by End User
11.2.7.4 Mexico: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.2.7.4.1 Mexico: Viral Vector Manufacturing Market Breakdown, by Type
11.2.7.4.2 Mexico: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.2.7.4.3 Mexico: Viral Vector Manufacturing Market Breakdown, by Application
11.2.7.4.4 Mexico: Viral Vector Manufacturing Market Breakdown, by End User
11.3 Europe
11.3.1 Europe Viral Vector Manufacturing Market Overview
11.3.2 Europe: Viral Vector Manufacturing Market – Revenue, 2021–2031 (US$ Million)
11.3.3 Europe: Viral Vector Manufacturing Market Breakdown, by Type
11.3.3.1 Europe: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Type
11.3.4 Europe: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.3.4.1 Europe: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Disease Indication
11.3.5 Europe: Viral Vector Manufacturing Market Breakdown, by Application
11.3.5.1 Europe: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Application
11.3.6 Europe: Viral Vector Manufacturing Market Breakdown, by End User
11.3.6.1 Europe: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by End User
11.3.7 Europe: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Country
11.3.7.1 Europe: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Country
11.3.7.2 Germany: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.3.7.2.1 Germany: Viral Vector Manufacturing Market Breakdown, by Type
11.3.7.2.2 Germany: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.3.7.2.3 Germany: Viral Vector Manufacturing Market Breakdown, by Application
11.3.7.2.4 Germany: Viral Vector Manufacturing Market Breakdown, by End User
11.3.7.3 United Kingdom: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.3.7.3.1 United Kingdom: Viral Vector Manufacturing Market Breakdown, by Type
11.3.7.3.2 United Kingdom: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.3.7.3.3 United Kingdom: Viral Vector Manufacturing Market Breakdown, by Application
11.3.7.3.4 United Kingdom: Viral Vector Manufacturing Market Breakdown, by End User
11.3.7.4 France: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.3.7.4.1 France: Viral Vector Manufacturing Market Breakdown, by Type
11.3.7.4.2 France: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.3.7.4.3 France: Viral Vector Manufacturing Market Breakdown, by Application
11.3.7.4.4 France: Viral Vector Manufacturing Market Breakdown, by End User
11.3.7.5 Italy: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.3.7.5.1 Italy: Viral Vector Manufacturing Market Breakdown, by Type
11.3.7.5.2 Italy: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.3.7.5.3 Italy: Viral Vector Manufacturing Market Breakdown, by Application
11.3.7.5.4 Italy: Viral Vector Manufacturing Market Breakdown, by End User
11.3.7.6 Spain: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.3.7.6.1 Spain: Viral Vector Manufacturing Market Breakdown, by Type
11.3.7.6.2 Spain: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.3.7.6.3 Spain: Viral Vector Manufacturing Market Breakdown, by Application
11.3.7.6.4 Spain: Viral Vector Manufacturing Market Breakdown, by End User
11.3.7.7 Rest of Europe: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.3.7.7.1 Rest of Europe: Viral Vector Manufacturing Market Breakdown, by Type
11.3.7.7.2 Rest of Europe: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.3.7.7.3 Rest of Europe: Viral Vector Manufacturing Market Breakdown, by Application
11.3.7.7.4 Rest of Europe: Viral Vector Manufacturing Market Breakdown, by End User
11.4 Asia Pacific
11.4.1 Asia Pacific Viral Vector Manufacturing Market Overview
11.4.2 Asia Pacific: Viral Vector Manufacturing Market – Revenue, 2021–2031 (US$ Million)
11.4.3 Asia Pacific: Viral Vector Manufacturing Market Breakdown, by Type
11.4.3.1 Asia Pacific: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Type
11.4.4 Asia Pacific: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.4.4.1 Asia Pacific: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Disease Indication
11.4.5 Asia Pacific: Viral Vector Manufacturing Market Breakdown, by Application
11.4.5.1 Asia Pacific: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Application
11.4.6 Asia Pacific: Viral Vector Manufacturing Market Breakdown, by End User
11.4.6.1 Asia Pacific: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by End User
11.4.7 Asia Pacific: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Country
11.4.7.1 Asia Pacific: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Country
11.4.7.2 China: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.4.7.2.1 China: Viral Vector Manufacturing Market Breakdown, by Type
11.4.7.2.2 China: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.4.7.2.3 China: Viral Vector Manufacturing Market Breakdown, by Application
11.4.7.2.4 China: Viral Vector Manufacturing Market Breakdown, by End User
11.4.7.3 Japan: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.4.7.3.1 Japan: Viral Vector Manufacturing Market Breakdown, by Type
11.4.7.3.2 Japan: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.4.7.3.3 Japan: Viral Vector Manufacturing Market Breakdown, by Application
11.4.7.3.4 Japan: Viral Vector Manufacturing Market Breakdown, by End User
11.4.7.4 India: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.4.7.4.1 India: Viral Vector Manufacturing Market Breakdown, by Type
11.4.7.4.2 India: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.4.7.4.3 India: Viral Vector Manufacturing Market Breakdown, by Application
11.4.7.4.4 India: Viral Vector Manufacturing Market Breakdown, by End User
11.4.7.5 Australia: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.4.7.5.1 Australia: Viral Vector Manufacturing Market Breakdown, by Type
11.4.7.5.2 Australia: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.4.7.5.3 Australia: Viral Vector Manufacturing Market Breakdown, by Application
11.4.7.5.4 Australia: Viral Vector Manufacturing Market Breakdown, by End User
11.4.7.6 South Korea: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.4.7.6.1 South Korea: Viral Vector Manufacturing Market Breakdown, by Type
11.4.7.6.2 South Korea: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.4.7.6.3 South Korea: Viral Vector Manufacturing Market Breakdown, by Application
11.4.7.6.4 South Korea: Viral Vector Manufacturing Market Breakdown, by End User
11.4.7.7 Rest of APAC: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.4.7.7.1 Rest of APAC: Viral Vector Manufacturing Market Breakdown, by Type
11.4.7.7.2 Rest of APAC: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.4.7.7.3 Rest of APAC: Viral Vector Manufacturing Market Breakdown, by Application
11.4.7.7.4 Rest of APAC: Viral Vector Manufacturing Market Breakdown, by End User
11.5 Middle East and Africa
11.5.1 Middle East and Africa Viral Vector Manufacturing Market Overview
11.5.2 Middle East and Africa: Viral Vector Manufacturing Market – Revenue, 2021–2031 (US$ Million)
11.5.3 Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by Type
11.5.3.1 Middle East and Africa: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Type
11.5.4 Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.5.4.1 Middle East and Africa: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Disease Indication
11.5.5 Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by Application
11.5.5.1 Middle East and Africa: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Application
11.5.6 Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by End User
11.5.6.1 Middle East and Africa: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by End User
11.5.7 Middle East and Africa: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Country
11.5.7.1 Middle East and Africa: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Country
11.5.7.2 Saudi Arabia: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.5.7.2.1 Saudi Arabia: Viral Vector Manufacturing Market Breakdown, by Type
11.5.7.2.2 Saudi Arabia: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.5.7.2.3 Saudi Arabia: Viral Vector Manufacturing Market Breakdown, by Application
11.5.7.2.4 Saudi Arabia: Viral Vector Manufacturing Market Breakdown, by End User
11.5.7.3 South Africa: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.5.7.3.1 South Africa: Viral Vector Manufacturing Market Breakdown, by Type
11.5.7.3.2 South Africa: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.5.7.3.3 South Africa: Viral Vector Manufacturing Market Breakdown, by Application
11.5.7.3.4 South Africa: Viral Vector Manufacturing Market Breakdown, by End User
11.5.7.4 United Arab Emirates: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.5.7.4.1 United Arab Emirates: Viral Vector Manufacturing Market Breakdown, by Type
11.5.7.4.2 United Arab Emirates: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.5.7.4.3 United Arab Emirates: Viral Vector Manufacturing Market Breakdown, by Application
11.5.7.4.4 United Arab Emirates: Viral Vector Manufacturing Market Breakdown, by End User
11.5.7.5 Rest of Middle East and Africa: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.5.7.5.1 Rest of Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by Type
11.5.7.5.2 Rest of Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.5.7.5.3 Rest of Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by Application
11.5.7.5.4 Rest of Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by End User
11.6 South and Central America
11.6.1 South and Central America Viral Vector Manufacturing Market Overview
11.6.2 South and Central America: Viral Vector Manufacturing Market – Revenue, 2021–2031 (US$ Million)
11.6.3 South and Central America: Viral Vector Manufacturing Market Breakdown, by Type
11.6.3.1 South and Central America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Type
11.6.4 South and Central America: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.6.4.1 South and Central America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Disease Indication
11.6.5 South and Central America: Viral Vector Manufacturing Market Breakdown, by Application
11.6.5.1 South and Central America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Application
11.6.6 South and Central America: Viral Vector Manufacturing Market Breakdown, by End User
11.6.6.1 South and Central America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by End User
11.6.7 South and Central America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Country
11.6.7.1 South and Central America: Viral Vector Manufacturing Market – Revenue and Forecast Analysis – by Country
11.6.7.2 Brazil: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.6.7.2.1 Brazil: Viral Vector Manufacturing Market Breakdown, by Type
11.6.7.2.2 Brazil: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.6.7.2.3 Brazil: Viral Vector Manufacturing Market Breakdown, by Application
11.6.7.2.4 Brazil: Viral Vector Manufacturing Market Breakdown, by End User
11.6.7.3 Argentina: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.6.7.3.1 Argentina: Viral Vector Manufacturing Market Breakdown, by Type
11.6.7.3.2 Argentina: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.6.7.3.3 Argentina: Viral Vector Manufacturing Market Breakdown, by Application
11.6.7.3.4 Argentina: Viral Vector Manufacturing Market Breakdown, by End User
11.6.7.4 Rest of South and Central America: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
11.6.7.4.1 Rest of South and Central America: Viral Vector Manufacturing Market Breakdown, by Type
11.6.7.4.2 Rest of South and Central America: Viral Vector Manufacturing Market Breakdown, by Disease Indication
11.6.7.4.3 Rest of South and Central America: Viral Vector Manufacturing Market Breakdown, by Application
11.6.7.4.4 Rest of South and Central America: Viral Vector Manufacturing Market Breakdown, by End User
12. Competitive Landscape
12.1 Heat Map Analysis by Key Players
12.2 Company Market Share Analysis, 2024
13. Industry Landscape
13.1 Overview
13.2 Market Initiative
13.3 Merger and Acquisition
13.4 Other Developments
14. Company Profiles
14.1 Lonza Group AG
14.1.1 Key Facts
14.1.2 Business Description
14.1.3 Products and Services
14.1.4 Financial Overview
14.1.5 SWOT Analysis
14.1.6 Key Developments
14.2 Merck KGaA
14.2.1 Key Facts
14.2.2 Business Description
14.2.3 Products and Services
14.2.4 Financial Overview
14.2.5 SWOT Analysis
14.2.6 Key Developments
14.3 Thermo Fisher Scientific Inc.
14.3.1 Key Facts
14.3.2 Business Description
14.3.3 Products and Services
14.3.4 Financial Overview
14.3.5 SWOT Analysis
14.3.6 Key Developments
14.4 Catalent Inc
14.4.1 Key Facts
14.4.2 Business Description
14.4.3 Products and Services
14.4.4 Financial Overview
14.4.5 SWOT Analysis
14.4.6 Key Developments
14.5 Charles River Laboratories InternationalInc
14.5.1 Key Facts
14.5.2 Business Description
14.5.3 Products and Services
14.5.4 Financial Overview
14.5.5 SWOT Analysis
14.5.6 Key Developments
14.6 WuXi AppTec Co Ltd
14.6.1 Key Facts
14.6.2 Business Description
14.6.3 Products and Services
14.6.4 Financial Overview
14.6.5 SWOT Analysis
14.6.6 Key Developments
14.7 FUJIFILM Holdings Corp
14.7.1 Key Facts
14.7.2 Business Description
14.7.3 Products and Services
14.7.4 Financial Overview
14.7.5 SWOT Analysis
14.7.6 Key Developments
14.8 GenScript Biotech Corporation
14.8.1 Key Facts
14.8.2 Business Description
14.8.3 Products and Services
14.8.4 Financial Overview
14.8.5 SWOT Analysis
14.8.6 Key Developments
14.9 Takara Bio Inc
14.9.1 Key Facts
14.9.2 Business Description
14.9.3 Products and Services
14.9.4 Financial Overview
14.9.5 SWOT Analysis
14.9.6 Key Developments
14.10 Novartis AG
14.10.1 Key Facts
14.10.2 Business Description
14.10.3 Products and Services
14.10.4 Financial Overview
14.10.5 SWOT Analysis
14.10.6 Key Developments
15. Appendix
15.1 About The Insight Partners
15.2 List of Abbreviations
List of Tables
Table 1. Viral Vector Manufacturing Market Segmentation
Table 2. List of Vendors
Table 3. Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million)
Table 4. Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million)
Table 5. Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 6. Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 7. Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 8. Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 9. Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 10. Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 11. Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 12. Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 13. North America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 14. North America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 15. North America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 16. North America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 17. North America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 18. North America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 19. North America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 20. North America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 21. North America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Country
Table 22. North America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Country
Table 23. United States: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 24. United States: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 25. United States: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 26. United States: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 27. United States: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 28. United States: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 29. United States: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 30. United States: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 31. Canada: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 32. Canada: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 33. Canada: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 34. Canada: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 35. Canada: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 36. Canada: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 37. Canada: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 38. Canada: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 39. Mexico: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 40. Mexico: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 41. Mexico: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 42. Mexico: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 43. Mexico: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 44. Mexico: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 45. Mexico: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 46. Mexico: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 47. Europe: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 48. Europe: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 49. Europe: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 50. Europe: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 51. Europe: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 52. Europe: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 53. Europe: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 54. Europe: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 55. Europe: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Country
Table 56. Europe: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Country
Table 57. Germany: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 58. Germany: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 59. Germany: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 60. Germany: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 61. Germany: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 62. Germany: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 63. Germany: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 64. Germany: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 65. United Kingdom: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 66. United Kingdom: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 67. United Kingdom: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 68. United Kingdom: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 69. United Kingdom: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 70. United Kingdom: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 71. United Kingdom: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 72. United Kingdom: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 73. France: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 74. France: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 75. France: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 76. France: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 77. France: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 78. France: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 79. France: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 80. France: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 81. Italy: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 82. Italy: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 83. Italy: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 84. Italy: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 85. Italy: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 86. Italy: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 87. Italy: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 88. Italy: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 89. Spain: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 90. Spain: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 91. Spain: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 92. Spain: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 93. Spain: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 94. Spain: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 95. Spain: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 96. Spain: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 97. Rest of Europe: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 98. Rest of Europe: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 99. Rest of Europe: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 100. Rest of Europe: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 101. Rest of Europe: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 102. Rest of Europe: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 103. Rest of Europe: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 104. Rest of Europe: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 105. Asia Pacific: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 106. Asia Pacific: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 107. Asia Pacific: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 108. Asia Pacific: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 109. Asia Pacific: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 110. Asia Pacific: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 111. Asia Pacific: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 112. Asia Pacific: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 113. Asia Pacific: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Country
Table 114. Asia Pacific: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Country
Table 115. China: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 116. China: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 117. China: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 118. China: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 119. China: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 120. China: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 121. China: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 122. China: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 123. Japan: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 124. Japan: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 125. Japan: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 126. Japan: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 127. Japan: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 128. Japan: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 129. Japan: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 130. Japan: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 131. India: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 132. India: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 133. India: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 134. India: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 135. India: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 136. India: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 137. India: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 138. India: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 139. Australia: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 140. Australia: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 141. Australia: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 142. Australia: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 143. Australia: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 144. Australia: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 145. Australia: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 146. Australia: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 147. South Korea: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 148. South Korea: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 149. South Korea: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 150. South Korea: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 151. South Korea: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 152. South Korea: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 153. South Korea: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 154. South Korea: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 155. Rest of APAC: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 156. Rest of APAC: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 157. Rest of APAC: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 158. Rest of APAC: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 159. Rest of APAC: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 160. Rest of APAC: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 161. Rest of APAC: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 162. Rest of APAC: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 163. Middle East and Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 164. Middle East and Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 165. Middle East and Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 166. Middle East and Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 167. Middle East and Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 168. Middle East and Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 169. Middle East and Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 170. Middle East and Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 171. Middle East and Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Country
Table 172. Middle East and Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Country
Table 173. Saudi Arabia: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 174. Saudi Arabia: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 175. Saudi Arabia: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 176. Saudi Arabia: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 177. Saudi Arabia: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 178. Saudi Arabia: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 179. Saudi Arabia: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 180. Saudi Arabia: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 181. South Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 182. South Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 183. South Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 184. South Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 185. South Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 186. South Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 187. South Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 188. South Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 189. United Arab Emirates: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 190. United Arab Emirates: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 191. United Arab Emirates: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 192. United Arab Emirates: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 193. United Arab Emirates: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 194. United Arab Emirates: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 195. United Arab Emirates: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 196. United Arab Emirates: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 197. Rest of Middle East and Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 198. Rest of Middle East and Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 199. Rest of Middle East and Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 200. Rest of Middle East and Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 201. Rest of Middle East and Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 202. Rest of Middle East and Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 203. Rest of Middle East and Africa: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 204. Rest of Middle East and Africa: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 205. South and Central America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 206. South and Central America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 207. South and Central America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 208. South and Central America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 209. South and Central America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 210. South and Central America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 211. South and Central America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 212. South and Central America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 213. South and Central America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Country
Table 214. South and Central America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Country
Table 215. Brazil: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 216. Brazil: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 217. Brazil: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 218. Brazil: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 219. Brazil: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 220. Brazil: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 221. Brazil: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 222. Brazil: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 223. Argentina: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 224. Argentina: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 225. Argentina: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 226. Argentina: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 227. Argentina: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 228. Argentina: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 229. Argentina: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 230. Argentina: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 231. Rest of South and Central America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Type
Table 232. Rest of South and Central America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Type
Table 233. Rest of South and Central America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Disease Indication
Table 234. Rest of South and Central America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Disease Indication
Table 235. Rest of South and Central America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by Application
Table 236. Rest of South and Central America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by Application
Table 237. Rest of South and Central America: Viral Vector Manufacturing Market – Revenue, 2021–2024 (US$ Million) – by End User
Table 238. Rest of South and Central America: Viral Vector Manufacturing Market – Revenue Forecast, 2025–2031 (US$ Million) – by End User
Table 239. Heat Map Analysis by Key Players
Table 240. List of Abbreviations
List of Figures
Figure 1. Viral Vector Manufacturing Market Segmentation, by Geography
Figure 2. PEST Analysis
Figure 3. Impact Analysis of Drivers and Restraints
Figure 4. Viral Vector Manufacturing Market Revenue (US$ Million), 2021–2031
Figure 5. Viral Vector Manufacturing Market Share (%) – by Type (2024 and 2031)
Figure 6. Adenoviral Vectors: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 7. Adeno-Associated Viral Vectors: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 8. Lentiviral Vectors: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 9. Retroviral Vectors: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 10. Others: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 11. Viral Vector Manufacturing Market Share (%) – by Disease Indication (2024 and 2031)
Figure 12. Cancer: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 13. Genetic Disorders: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 14. Infectious Diseases: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 15. Others: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 16. Viral Vector Manufacturing Market Share (%) – by Application (2024 and 2031)
Figure 17. Therapeutics Development: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 18. Vaccine Development: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 19. Research: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 20. Viral Vector Manufacturing Market Share (%) – by End User (2024 and 2031)
Figure 21. Pharmaceutical and Biotechnology Companies: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 22. CDMOs and CROs: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 23. Research Institutes: Viral Vector Manufacturing Market – Revenue and Forecast to 2031 (US$ Million)
Figure 24. Viral Vector Manufacturing Market Breakdown by Region, 2024 and 2031 (%)
Figure 25. North America: Viral Vector Manufacturing Market – Revenue, 2021–2031 (US$ Million)
Figure 26. North America: Viral Vector Manufacturing Market Breakdown, by Type (2024 and 2031)
Figure 27. North America: Viral Vector Manufacturing Market Breakdown, by Disease Indication (2024 and 2031)
Figure 28. North America: Viral Vector Manufacturing Market Breakdown, by Application (2024 and 2031)
Figure 29. North America: Viral Vector Manufacturing Market Breakdown, by End User (2024 and 2031)
Figure 30. North America: Viral Vector Manufacturing Market Breakdown, by Key Countries, 2024 and 2031 (%)
Figure 31. United States: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 32. Canada: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 33. Mexico: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 34. Europe: Viral Vector Manufacturing Market – Revenue, 2021–2031 (US$ Million)
Figure 35. Europe: Viral Vector Manufacturing Market Breakdown, by Type (2024 and 2031)
Figure 36. Europe: Viral Vector Manufacturing Market Breakdown, by Disease Indication (2024 and 2031)
Figure 37. Europe: Viral Vector Manufacturing Market Breakdown, by Application (2024 and 2031)
Figure 38. Europe: Viral Vector Manufacturing Market Breakdown, by End User (2024 and 2031)
Figure 39. Europe: Viral Vector Manufacturing Market Breakdown, by Key Countries, 2024 and 2031 (%)
Figure 40. Germany: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 41. United Kingdom: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 42. France: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 43. Italy: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 44. Spain: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 45. Rest of Europe: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 46. Asia Pacific: Viral Vector Manufacturing Market – Revenue, 2021–2031 (US$ Million)
Figure 47. Asia Pacific: Viral Vector Manufacturing Market Breakdown, by Type (2024 and 2031)
Figure 48. Asia Pacific: Viral Vector Manufacturing Market Breakdown, by Disease Indication (2024 and 2031)
Figure 49. Asia Pacific: Viral Vector Manufacturing Market Breakdown, by Application (2024 and 2031)
Figure 50. Asia Pacific: Viral Vector Manufacturing Market Breakdown, by End User (2024 and 2031)
Figure 51. Asia Pacific: Viral Vector Manufacturing Market Breakdown, by Key Countries, 2024 and 2031 (%)
Figure 52. China: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 53. Japan: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 54. India: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 55. Australia: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 56. South Korea: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 57. Rest of APAC: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 58. Middle East and Africa: Viral Vector Manufacturing Market – Revenue, 2021–2031 (US$ Million)
Figure 59. Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by Type (2024 and 2031)
Figure 60. Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by Disease Indication (2024 and 2031)
Figure 61. Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by Application (2024 and 2031)
Figure 62. Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by End User (2024 and 2031)
Figure 63. Middle East and Africa: Viral Vector Manufacturing Market Breakdown, by Key Countries, 2024 and 2031 (%)
Figure 64. Saudi Arabia: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 65. South Africa: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 66. United Arab Emirates: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 67. Rest of Middle East and Africa: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 68. South and Central America: Viral Vector Manufacturing Market – Revenue, 2021–2031 (US$ Million)
Figure 69. South and Central America: Viral Vector Manufacturing Market Breakdown, by Type (2024 and 2031)
Figure 70. South and Central America: Viral Vector Manufacturing Market Breakdown, by Disease Indication (2024 and 2031)
Figure 71. South and Central America: Viral Vector Manufacturing Market Breakdown, by Application (2024 and 2031)
Figure 72. South and Central America: Viral Vector Manufacturing Market Breakdown, by End User (2024 and 2031)
Figure 73. South and Central America: Viral Vector Manufacturing Market Breakdown, by Key Countries, 2024 and 2031 (%)
Figure 74. Brazil: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 75. Argentina: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 76. Rest of South and Central America: Viral Vector Manufacturing Market – Revenue and Forecast, 2021–2031 (US$ Million)
Figure 77. Company Market Share Analysis, 2024
Yes! We provide a free sample of the report, which includes Report Scope (Table of Contents), report structure, and selected insights to help you assess the value of the full report. Please click on the "Download Sample" button or contact us to receive your copy.
Absolutely - analyst assistance is part of the package. You can connect with our analyst post-purchase to clarify report insights, methodology or discuss how the findings apply to your business needs.
Once your order is successfully placed, you will receive a confirmation email along with your invoice.
• For published reports: You'll receive access to the report within 4-6 working hours via a secured email sent to your email.
• For upcoming reports: Your order will be recorded as a pre-booking. Our team will share the estimated release date and keep you informed of any updates. As soon as the report is published, it will be delivered to your registered email.
We offer customization options to align the report with your specific objectives. Whether you need deeper insights into a particular region, industry segment, competitor analysis, or data cut, our research team can tailor the report accordingly. Please share your requirements with us, and we'll be happy to provide a customized proposal or scope.
The report is available in either PDF format or as an Excel dataset, depending on the license you choose.
The PDF version provides the full analysis and visuals in a ready-to-read format. The Excel dataset includes all underlying data tables for easy manipulation and further analysis.
Please review the license options at checkout or contact us to confirm which formats are included with your purchase.
Our payment process is fully secure and PCI-DSS compliant.
We use trusted and encrypted payment gateways to ensure that all transactions are protected with industry-standard SSL encryption. Your payment details are never stored on our servers and are handled securely by certified third-party processors.
You can make your purchase with confidence, knowing your personal and financial information is safe with us.
Yes, we do offer special pricing for bulk purchases.
If you're interested in purchasing multiple reports, we're happy to provide a customized bundle offer or volume-based discount tailored to your needs. Please contact our sales team with the list of reports you're considering, and we'll share a personalized quote.
Yes, absolutely.
Our team is available to help you make an informed decision. Whether you have questions about the report's scope, methodology, customization options, or which license suits you best, we're here to assist. Please reach out to us at sales@theinsightpartners.com, and one of our representatives will get in touch promptly.
Yes, a billing invoice will be automatically generated and sent to your registered email upon successful completion of your purchase.
If you need the invoice in a specific format or require additional details (such as company name, GST, or VAT information), feel free to contact us, and we'll be happy to assist.
Yes, certainly.
If you encounter any difficulties accessing or receiving your report, our support team is ready to assist you. Simply reach out to us via email or live chat with your order information, and we'll ensure the issue is resolved quickly so you can access your report without interruption.
The Insight Partners performs research in 4 major stages: Data Collection & Secondary Research, Primary Research, Data Analysis and Data Triangulation & Final Review.
- Data Collection and Secondary Research:
As a market research and consulting firm operating from a decade, we have published many reports and advised several clients across the globe. First step for any study will start with an assessment of currently available data and insights from existing reports. Further, historical and current market information is collected from Investor Presentations, Annual Reports, SEC Filings, etc., and other information related to company’s performance and market positioning are gathered from Paid Databases (Factiva, Hoovers, and Reuters) and various other publications available in public domain.
Several associations trade associates, technical forums, institutes, societies and organizations are accessed to gain technical as well as market related insights through their publications such as research papers, blogs and press releases related to the studies are referred to get cues about the market. Further, white papers, journals, magazines, and other news articles published in the last 3 years are scrutinized and analyzed to understand the current market trends.
- Primary Research:
The primarily interview analysis comprise of data obtained from industry participants interview and answers to survey questions gathered by in-house primary team.
For primary research, interviews are conducted with industry experts/CEOs/Marketing Managers/Sales Managers/VPs/Subject Matter Experts from both demand and supply side to get a 360-degree view of the market. The primary team conducts several interviews based on the complexity of the markets to understand the various market trends and dynamics which makes research more credible and precise.
A typical research interview fulfils the following functions:
- Provides first-hand information on the market size, market trends, growth trends, competitive landscape, and outlook
- Validates and strengthens in-house secondary research findings
- Develops the analysis team’s expertise and market understanding
Primary research involves email interactions and telephone interviews for each market, category, segment, and sub-segment across geographies. The participants who typically take part in such a process include, but are not limited to:
- Industry participants: VPs, business development managers, market intelligence managers and national sales managers
- Outside experts: Valuation experts, research analysts and key opinion leaders specializing in the electronics and semiconductor industry.
Below is the breakup of our primary respondents by company, designation, and region:
Once we receive the confirmation from primary research sources or primary respondents, we finalize the base year market estimation and forecast the data as per the macroeconomic and microeconomic factors assessed during data collection.
- Data Analysis:
Once data is validated through both secondary as well as primary respondents, we finalize the market estimations by hypothesis formulation and factor analysis at regional and country level.
- 3.1 Macro-Economic Factor Analysis:
We analyse macroeconomic indicators such the gross domestic product (GDP), increase in the demand for goods and services across industries, technological advancement, regional economic growth, governmental policies, the influence of COVID-19, PEST analysis, and other aspects. This analysis aids in setting benchmarks for various nations/regions and approximating market splits. Additionally, the general trend of the aforementioned components aid in determining the market's development possibilities.
- 3.2 Country Level Data:
Various factors that are especially aligned to the country are taken into account to determine the market size for a certain area and country, including the presence of vendors, such as headquarters and offices, the country's GDP, demand patterns, and industry growth. To comprehend the market dynamics for the nation, a number of growth variables, inhibitors, application areas, and current market trends are researched. The aforementioned elements aid in determining the country's overall market's growth potential.
- 3.3 Company Profile:
The “Table of Contents” is formulated by listing and analyzing more than 25 - 30 companies operating in the market ecosystem across geographies. However, we profile only 10 companies as a standard practice in our syndicate reports. These 10 companies comprise leading, emerging, and regional players. Nonetheless, our analysis is not restricted to the 10 listed companies, we also analyze other companies present in the market to develop a holistic view and understand the prevailing trends. The “Company Profiles” section in the report covers key facts, business description, products & services, financial information, SWOT analysis, and key developments. The financial information presented is extracted from the annual reports and official documents of the publicly listed companies. Upon collecting the information for the sections of respective companies, we verify them via various primary sources and then compile the data in respective company profiles. The company level information helps us in deriving the base number as well as in forecasting the market size.
- 3.4 Developing Base Number:
Aggregation of sales statistics (2020-2022) and macro-economic factor, and other secondary and primary research insights are utilized to arrive at base number and related market shares for 2022. The data gaps are identified in this step and relevant market data is analyzed, collected from paid primary interviews or databases. On finalizing the base year market size, forecasts are developed on the basis of macro-economic, industry and market growth factors and company level analysis.
- Data Triangulation and Final Review:
The market findings and base year market size calculations are validated from supply as well as demand side. Demand side validations are based on macro-economic factor analysis and benchmarks for respective regions and countries. In case of supply side validations, revenues of major companies are estimated (in case not available) based on industry benchmark, approximate number of employees, product portfolio, and primary interviews revenues are gathered. Further revenue from target product/service segment is assessed to avoid overshooting of market statistics. In case of heavy deviations between supply and demand side values, all thes steps are repeated to achieve synchronization.
We follow an iterative model, wherein we share our research findings with Subject Matter Experts (SME’s) and Key Opinion Leaders (KOLs) until consensus view of the market is not formulated – this model negates any drastic deviation in the opinions of experts. Only validated and universally acceptable research findings are quoted in our reports.
We have important check points that we use to validate our research findings – which we call – data triangulation, where we validate the information, we generate from secondary sources with primary interviews and then we re-validate with our internal data bases and Subject matter experts. This comprehensive model enables us to deliver high quality, reliable data in shortest possible time.


 Get Free Sample For
Get Free Sample For