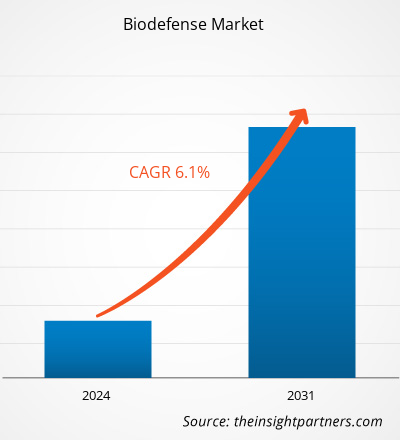
Der Markt für Bioabwehrprodukte hatte 2021 ein Volumen von 12,37 Milliarden US-Dollar und soll bis 2031 auf 21,62 Milliarden US-Dollar anwachsen. Der Markt soll in den Jahren 2023–2031 eine durchschnittliche jährliche Wachstumsrate von 6,1 % verzeichnen. Big Data und künstliche Intelligenz bieten enorme Möglichkeiten für die Durchführung technischer Bewertungen des Risikos und der Möglichkeit verschiedener Biobedrohungen sowie eine beispiellose Möglichkeit zur Kartierung von Krankheitsausbrüchen. Somit kann prädiktive Analytik dazu beitragen, die Bioabwehrbranche neu zu gestalten und als wichtiger Trend auf dem Bioabwehrmarkt zu dienen.
Bioverteidigungsmarktanalyse
Dank der jüngsten wissenschaftlichen Fortschritte in der Gentechnik und Biotechnologie können die tödlichen, natürlich vorkommenden Viren heute leicht verändert werden, um noch mehr Schaden anzurichten . Das wachsende Bewusstsein für die potenziell verheerenden Auswirkungen biologischer Angriffe und Ausbrüche hat die Nachfrage nach Bioabwehrlösungen erhöht. Weltweit investieren Regierungen erheblich in Bioabwehrkapazitäten, was die Expansion des Marktes unterstützt. Technologische Entwicklungen und eine wachsende Betonung von Forschung und Entwicklung haben außerdem zur Einführung hochentwickelter und innovativer Bioabwehrlösungen geführt. Die Weiterentwicklung von Bioabwehrlösungen wird durch technologische Innovationen wie synthetische Biologie, Genomik und Datenanalyse vorangetrieben, die verbesserte Möglichkeiten zur Identifizierung, Charakterisierung und Eindämmung biologischer Bedrohungen bieten. Darüber hinaus haben Partnerschaften zwischen staatlichen, Verteidigungs- und kommerziellen Organisationen die Entwicklung umfassender Bioabwehrstrategien unterstützt und die Marktexpansion gefördert.
Marktübersicht für Bioabwehr
Bioverteidigung bezieht sich auf die Bandbreite von Aktivitäten und Technologien, die darauf abzielen, die Biosicherheit eines Landes wiederherzustellen, das biologischen Bedrohungen oder Infektionskrankheiten ausgesetzt ist oder sein könnte. Sie hilft, den potenziellen Risiken im Zusammenhang mit Pandemien, Bioterrorismus und anderen biologischen Notfällen entgegenzuwirken. Die Bundesregierung der USA koordiniert Programme und richtet Budgets ein, um biologischen Katastrophen vorzubeugen, darauf zu reagieren und Maßnahmen dagegen zu ergreifen. Das US-amerikanische Programm zur biologischen Verteidigung, auch bekannt als Nationale Bioverteidigungsstrategie, umfasst gemeinsame Anstrengungen zahlreicher Regierungsebenen in Verbindung mit privaten Stellen und anderen Interessengruppen zur Durchführung von Bioverteidigungsaktivitäten. Darüber hinaus ist das National Collaborating Centre for Infectious Diseases eine Organisation in Kanada, die politische Entscheidungsträger, Praktiker des öffentlichen Gesundheitswesens, Gesundheitsinspektoren und andere durch Webinare, Podcasts, Online-Ressourcen und Veröffentlichungen mit aktuellen Informationen zu neu auftretenden Infektionskrankheiten versorgen möchte. Daher werden die Bemühungen und Initiativen der Regierung zur Stärkung des Bioverteidigungssystems wahrscheinlich das Marktwachstum im Prognosezeitraum unterstützen.
Passen Sie diesen Bericht Ihren Anforderungen an
Sie erhalten kostenlos individuelle Anpassungen an jedem Bericht, einschließlich Teilen dieses Berichts oder einer Analyse auf Länderebene, eines Excel-Datenpakets sowie tolle Angebote und Rabatte für Start-ups und Universitäten.
Bioverteidigungsmarkt: Strategische Einblicke

-
Holen Sie sich die wichtigsten Markttrends aus diesem Bericht.Dieses KOSTENLOSE Beispiel umfasst eine Datenanalyse von Markttrends bis hin zu Schätzungen und Prognosen.
Treiber und Chancen auf dem Bioverteidigungsmarkt
Vorhandensein günstiger Regierungsinitiativen zur Förderung des Marktwachstums
Die Regierungen der Industrieländer ergreifen eine Reihe von Maßnahmen und ergreifen vorbeugende Maßnahmen, um Bioterrorismus-Angriffen standzuhalten. Die Regierung hält ständig Vorräte an Impfstoffen und Geräten zur Erkennung biologischer Bedrohungen bereit, um die Bevölkerung zu schützen. Dies ist auf steigende Bundesfinanzierung, technische Durchbrüche und ein wachsendes öffentliches Bewusstsein zurückzuführen. Auch die Präsenz wichtiger Akteure ist für den Markt ermutigend. Im Jahr 2018 veröffentlichte die US-Regierung die Nationale Strategie und den Implementierungsplan zur Bioverteidigung, die Nationale Sicherheitsstrategie, die Nationale Verteidigungsstrategie und die Nationale Strategie zur Terrorismusbekämpfung sowie die Nationale Strategie zur Bekämpfung von Massenvernichtungswaffen, die alle Biosicherheit und Bioverteidigung beinhalten.
Darüber hinaus entwickeln in Schwellenmärkten wie Indien mehrere Einrichtungen, wie etwa die Defense Research and Development Organization, Abwehrmaßnahmen gegen biologische Angriffe. Es werden erhebliche Anstrengungen unternommen, um die Streitkräfte auf einen biologischen Angriff vorzubereiten. In den letzten zwei Jahrzehnten hat sich der Fokus auf die Sensibilisierung für Bioterrorismus in allen Ländern kontinuierlich verstärkt, zusammen mit verschiedenen Vorschlägen und Treffen zur Bekämpfung solcher Bedrohungen, was einer der Hauptgründe für die Nachfrage nach Bioabwehr ist. Die Initiativen von Regierungen weltweit eröffnen Unternehmen, die im Bioabwehrsektor tätig sind, ein Fenster der Chancen und treiben so das Marktwachstum voran.
Verstärkter Einsatz von Technologien zur Förderung des Marktwachstums
Die zunehmende Nutzung von Technologien wie Genchips zur Erkennung von Krankheitserregern in der Forensik, Datenbankverwaltungssystemen, fortschrittlicher Detektorhardware in der Entwicklung von Arzneimitteln für die Bioverteidigung und Nanotechnologie wird erfolgreich zur Verbesserung biologischer Verteidigungstechniken eingesetzt. Die meisten dieser Technologien werden entweder unabhängig oder gemeinsam eingesetzt, um verschiedene Bioverteidigungswerkzeuge zu entwerfen und zu entwickeln. Die Technologie, die derzeit für die Bioverteidigung am wichtigsten ist, ist eng mit der Gentechnik verwandt, einer Technologie, die auf der künstlichen Manipulation und Übertragung von genetischem Material basiert. Gleichzeitig könnte die Gentherapie wirksam zur Heilung verschiedener Krankheiten eingesetzt werden. Viele sagen voraus, dass zukünftige Biowaffen Designer-Biowaffen sein könnten, und diese Technologie könnte eine sehr wichtige Rolle bei der Suche nach Heilmitteln für viele derzeit unbekannte Krankheiten spielen. Jüngste Fortschritte in der Molekularbiologie und Gentechnik haben zu neuen Strategien zur Impfstoffentwicklung geführt und werden wahrscheinlich in Zukunft erhebliche Wachstumschancen für den Bioverteidigungsmarkt schaffen.
Segmentierungsanalyse des Bioverteidigungsmarktberichts
Die wichtigsten Segmente, die zur Ableitung der Bioabwehrmarktanalyse beigetragen haben, sind nach Produkten gegliedert.
- Basierend auf dem Produkt ist der Bioverteidigungsmarkt unterteilt inAnthrax, Pocken, Botulismus, Strahlung/Atom, andere. Das Anthrax-Segment hatte 2023 den größten Marktanteil und wird im Prognosezeitraum voraussichtlich die höchste durchschnittliche jährliche Wachstumsrate verzeichnen.
Bioverteidigung Marktanteilsanalyse nach Geografie
Der geografische Umfang des Bioabwehrmarktberichts ist hauptsächlich in fünf Regionen unterteilt: Nordamerika, Asien-Pazifik, Europa, Naher Osten und Afrika sowie Süd- und Mittelamerika.
Nordamerika dominiert den Markt für Bioabwehr. Biologische Bedrohungen für Menschen, Tiere und die Umwelt gehören zu den schwerwiegendsten Problemen, mit denen die USA und ihre nationale Gemeinschaft konfrontiert sind. Da die biologische Bedrohung weiter zunimmt, konzentrieren sich die USA auf die Stärkung ihrer Fähigkeiten und betrachten die Vorbereitung auf Biobedrohungen und Bioterrorismus als einen der kritischen Aspekte der nationalen Sicherheit. Ein starker Fokus auf die nationale Sicherheit, ein gut etabliertes Gesundheitssystem und robuste Forschungs- und Entwicklungskapazitäten begünstigen das Marktwachstum in der Region. Aufgrund starker und proaktiver Regierungsaktivitäten, Partnerschaften mit Akteuren aus der Industrie und eines hohen Maßes an Vorbereitung auf Bioterrorismusbedrohungen hat die Region erhebliche Investitionen in Bioabwehrtechnologie, einschließlich Therapeutika, Impfstoffe und Erkennungssysteme, verzeichnet.
Bioverteidigungsstrategien von Organisationen wie BARDA, NIAID, CDC, ASPR und der FDA tragen zum Wachstum der Region bei. Die beträchtliche Finanzierung der Bioverteidigung durch das US-Militär und zivile Behörden ist auch einer der Hauptfaktoren, die für den höchsten Umsatzanteil der Region verantwortlich sind.
Der asiatisch-pazifische Raum wird in den kommenden Jahren voraussichtlich die höchste durchschnittliche jährliche Wachstumsrate aufweisen. Aufgrund steigender Investitionen in Forschung und Entwicklung unterstützen die Bedeutung der Bioabwehr und die wachsende Bedrohung durch gefährliche biologische Stoffe, die Notfälle verursachen, das Marktwachstum in der Region. Die hohe Anfälligkeit der Region für Infektionskrankheiten und die große Bevölkerung haben zusammen mit der Präsenz einer hochentwickelten Gesundheitsinfrastruktur auch die Nachfrage nach Bioabwehrprodukten und -technologien erhöht.
Regionale Einblicke in den Bioverteidigungsmarkt
Die regionalen Trends und Faktoren, die den Bioverteidigungsmarkt während des Prognosezeitraums beeinflussen, wurden von den Analysten von Insight Partners ausführlich erläutert. In diesem Abschnitt werden auch die Marktsegmente und die Geografie des Bioverteidigungsmarkts in Nordamerika, Europa, im asiatisch-pazifischen Raum, im Nahen Osten und Afrika sowie in Süd- und Mittelamerika erörtert.
- Erhalten Sie regionale Daten zum Bioverteidigungsmarkt
Umfang des Marktberichts zur Bioabwehr
| Berichtsattribut | Details |
|---|---|
| Marktgröße im Jahr 2021 | 12,37 Milliarden US-Dollar |
| Marktgröße bis 2031 | 21,62 Milliarden US-Dollar |
| Globale CAGR (2023 - 2031) | 6,1 % |
| Historische Daten | 2021-2023 |
| Prognosezeitraum | 2024–2031 |
| Abgedeckte Segmente |
Nach Produkt
|
| Abgedeckte Regionen und Länder |
Nordamerika
|
| Marktführer und wichtige Unternehmensprofile |
|
Dichte der Akteure auf dem Bioverteidigungsmarkt: Die Auswirkungen auf die Geschäftsdynamik verstehen
Der Markt für Bioabwehrprodukte wächst rasant, angetrieben durch die steigende Nachfrage der Endverbraucher aufgrund von Faktoren wie sich entwickelnden Verbraucherpräferenzen, technologischen Fortschritten und einem größeren Bewusstsein für die Vorteile des Produkts. Mit steigender Nachfrage erweitern Unternehmen ihr Angebot, entwickeln Innovationen, um die Bedürfnisse der Verbraucher zu erfüllen, und nutzen neue Trends, was das Marktwachstum weiter ankurbelt.
Die Marktteilnehmerdichte bezieht sich auf die Verteilung der Firmen oder Unternehmen, die in einem bestimmten Markt oder einer bestimmten Branche tätig sind. Sie gibt an, wie viele Wettbewerber (Marktteilnehmer) in einem bestimmten Marktraum im Verhältnis zu seiner Größe oder seinem gesamten Marktwert präsent sind.
Die wichtigsten auf dem Bioabwehrmarkt tätigen Unternehmen sind:
- Bayerisch Nordisch
- Alnylam Pharmaceuticals, Inc.
- SIGA Technologies
- Emergent BioSolutions Inc.
- Cleveland Bio Labs
- Dynavax-Technologien
Haftungsausschluss : Die oben aufgeführten Unternehmen sind nicht in einer bestimmten Reihenfolge aufgeführt.
- Überblick über die wichtigsten Akteure auf dem Bioverteidigungsmarkt
Neuigkeiten und aktuelle Entwicklungen zum Bioverteidigungsmarkt
Der Markt für Bioabwehr wird durch die Erhebung qualitativer und quantitativer Daten aus Primär- und Sekundärforschung bewertet, die wichtige Unternehmensveröffentlichungen, Verbandsdaten und Datenbanken umfasst. Im Folgenden finden Sie eine Liste der Entwicklungen auf dem Markt für Bioabwehr und -strategien:
- Im Februar 2022 gab Appili Therapeutics Inc. bekannt, dass das US-Verteidigungsministerium über das Joint Science and Technology Office der Defense Threat Reduction Agency eine Finanzierung in Höhe von über 10 Millionen US-Dollar bereitgestellt hat, um den Bioabwehrimpfstoffkandidaten ATI-1701 des Unternehmens voranzutreiben , einen potenziellen First-in-Class-Impfstoffkandidaten zur Vorbeugung einer Infektion mit Francisella tularensis. Die Finanzierung soll die Präsenz des Unternehmens stärken und das Wachstum auf dem Bioabwehrmarkt steigern. (Quelle: Appili Therapeutics, Pressemitteilung, 2022)
- Im September 2022 schloss Emergent BioSolutions Inc. seine endgültige Vereinbarung mit Chimerix, Inc. ab, um die exklusiven weltweiten Rechte von Chimerix an TEMBEXA (Brincidofovir) zu erwerben, dem ersten oralen antiviralen Mittel, das von der US-amerikanischen Food and Drug Administration (FDA) zur Behandlung von Pocken in allen Altersgruppen zugelassen wurde. (Quelle: Emergent BioSolutions Inc., Pressemitteilung, 2022)
- Im Juli 2023Emergent BioSolutions Inc. erhielt von der US-amerikanischen FDA die Zulassung für CYFENDUS (Anthrax Vaccine Adsorbed, Adjuvanted), zuvor bekannt als AV7909, zur Postexpositionsprophylaxe der Krankheit nach vermuteter oder bestätigter Exposition gegenüber Bacillus anthracis bei Personen im Alter von 18 bis 65 Jahren, wenn es zusammen mit empfohlenen antibakteriellen Medikamenten verabreicht wird. (Quelle: Emergent BioSolutions Inc., Pressemitteilung, 2023)
Marktbericht zur Bioverteidigung – Umfang und Ergebnisse
Der Bericht „Marktgröße und Prognose für Bioverteidigung (2021–2031)“ bietet eine detaillierte Analyse des Marktes, die die folgenden Bereiche abdeckt:
- Marktgröße und Prognose auf globaler, regionaler und Länderebene für alle wichtigen Marktsegmente, die im Rahmen des Projekts abgedeckt sind
- Marktdynamik wie Treiber, Beschränkungen und wichtige Chancen
- Wichtige Zukunftstrends
- Detaillierte PEST/Porters Five Forces- und SWOT-Analyse
- Globale und regionale Marktanalyse mit wichtigen Markttrends, wichtigen Akteuren, Vorschriften und aktuellen Marktentwicklungen
- Branchenlandschaft und Wettbewerbsanalyse, einschließlich Marktkonzentration, Heatmap-Analyse, prominenten Akteuren und aktuellen Entwicklungen
- Detaillierte Firmenprofile
- Historische Analyse (2 Jahre), Basisjahr, Prognose (7 Jahre) mit CAGR
- PEST- und SWOT-Analyse
- Marktgröße Wert/Volumen – Global, Regional, Land
- Branchen- und Wettbewerbslandschaft
- Excel-Datensatz
Aktuelle Berichte
Verwandte Berichte
Erfahrungsberichte
Grund zum Kauf
- Fundierte Entscheidungsfindung
- Marktdynamik verstehen
- Wettbewerbsanalyse
- Kundeneinblicke
- Marktprognosen
- Risikominimierung
- Strategische Planung
- Investitionsbegründung
- Identifizierung neuer Märkte
- Verbesserung von Marketingstrategien
- Steigerung der Betriebseffizienz
- Anpassung an regulatorische Trends






















 Kostenlose Probe anfordern für - Markt für Bioabwehr
Kostenlose Probe anfordern für - Markt für Bioabwehr