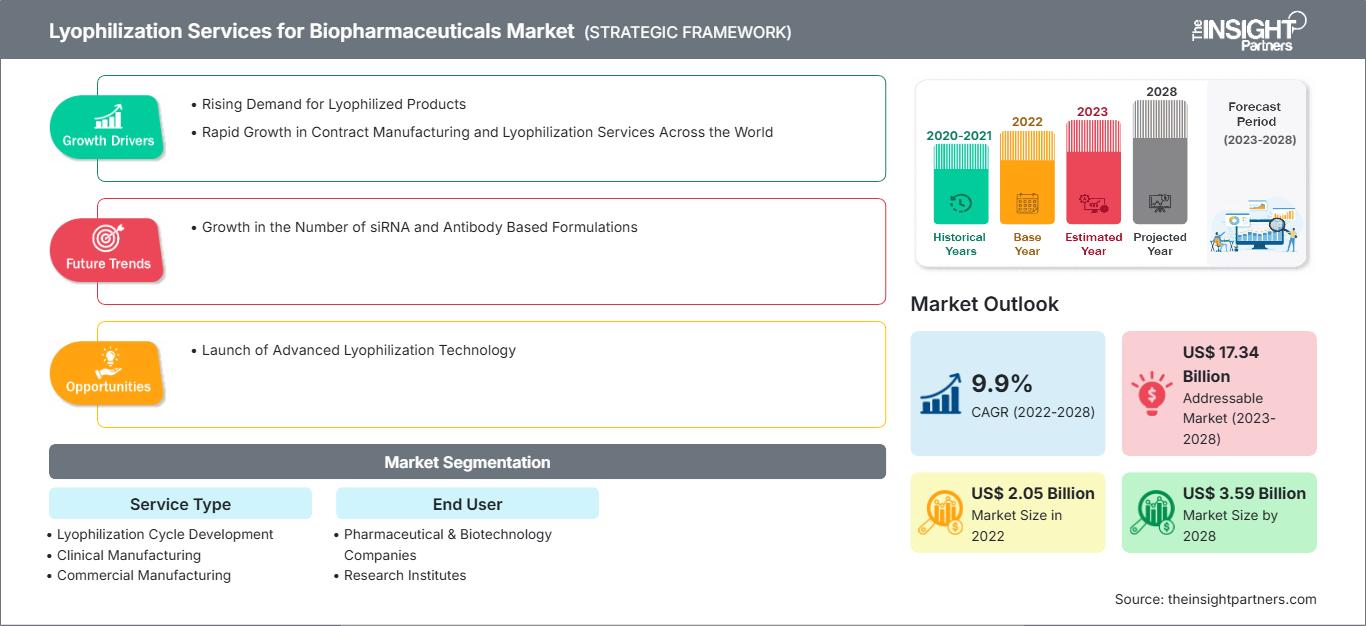
[Rapport de recherche] Le marché des services de lyophilisation pour les produits biopharmaceutiques devrait passer de 2 051,41 millions de dollars américains en 2022 à 3 586,55 millions de dollars américains en 2028 ; il devrait enregistrer un TCAC de 9,9 % entre 2023 et 2028.
La croissance du marché des services de lyophilisation pour les produits biopharmaceutiques est attribuée à la demande croissante de produits lyophilisés et à la croissance rapide des services de fabrication sous contrat et de lyophilisation à travers le monde.
Le marché des services de lyophilisation pour les produits biopharmaceutiques est segmenté en fonction du type de service, de l'utilisateur final et de la région. Le rapport offre des informations et une analyse approfondie du marché, en mettant l'accent sur des paramètres tels que les tendances du marché des services de lyophilisation pour les produits biopharmaceutiques, les avancées technologiques, la dynamique du marché et l'analyse du paysage concurrentiel des principaux acteurs du marché.
Services de lyophilisation pour le marché des produits biopharmaceutiques – Perspectives du marché
L'augmentation rapide de la fabrication sous contrat et des services de lyophilisation stimule la croissance du marché des services de lyophilisation pour les produits biopharmaceutiques
Le marché des produits biopharmaceutiques continue de se développer à l'échelle mondiale, ce qui peut constituer le principal moteur de croissance de l'industrie pharmaceutique. La fabrication parentérale de produits biopharmaceutiques s'est récemment développée avec l'approbation de nouveaux produits biologiques thérapeutiques. Les entreprises de biotechnologie sous-traitent leurs activités à des organisations de fabrication sous contrat (CMO) pour répondre à leurs besoins de remplissage et de finition et réduire le risque de contamination microbienne. Les entreprises biopharmaceutiques dépendent de ces organisations pour fournir les capacités et les compétences nécessaires ; dans certains cas, les CMO assurent une grande partie de la production d'une entreprise.
Personnalisez ce rapport en fonction de vos besoins
Vous bénéficierez d’une personnalisation sur n’importe quel rapport - gratuitement - y compris des parties de ce rapport, ou une analyse au niveau du pays, un pack de données Excel, ainsi que de profiter d’offres exceptionnelles et de réductions pour les start-ups et les universités
Services de lyophilisation pour le marché des produits biopharmaceutiques: Perspectives stratégiques
-
Obtenez les principales tendances clés du marché de ce rapport.Cet échantillon GRATUIT comprendra une analyse de données, allant des tendances du marché aux estimations et prévisions.
La mise en place de capacités et d'opérations de lyophilisation en interne nécessite des équipements et une expertise spécialisés, ce qui est coûteux et chronophage. Cependant, l'externalisation est moins coûteuse et améliore l'efficacité des processus de fabrication. De plus, elle permet aux entreprises de biotechnologie de réorienter leurs ressources vers d'autres domaines. Ainsi, les développeurs de médicaments et les sociétés biopharmaceutiques externalisent ces opérations auprès de CMO afin de réduire les coûts globaux de production et de rendement. Il y a quelques années, le secteur des CMO était un marché de services de niche, offrant des capacités de production supplémentaires ou des services spécifiques aux entreprises de biotechnologie. Aujourd'hui, de nombreuses entreprises de biotechnologie externalisent divers services, du développement précoce de médicaments à la production commerciale. Alors que le secteur des biotechnologies évolue d'une production à grande échelle vers des thérapies de niche et ciblées (médecine personnalisée), la demande de capacités opérationnelles flexibles, d'échelles de production et d'opérations multi-produits augmente. En raison de tous ces facteurs, la tendance vers les CMO est croissante. Jubilant HollisterStier Contract Manufacturing & Services est l'une de ces CMO qui fournit des installations spécialisées et des lignes dédiées aux opérations de lyophilisation. Le CMO propose des services de remplissage/finition stériles de phase I grâce à un produit injectable stérile commercial et une gamme complète de services de lyophilisation. Pour répondre à la demande croissante, Jubilant installe un nouveau lyophilisateur de 385 pieds carrés. Ainsi, la capacité et la disponibilité croissantes des CMO biopharmaceutiques alimentent la croissance des services de lyophilisation pour le marché biopharmaceutique.
Services de lyophilisation pour le marché biopharmaceutique – Informations par type de service
Selon le type de service, le marché mondial des services de lyophilisation pour les produits biopharmaceutiques est segmenté en fabrication commerciale, développement de cycles de lyophilisation, fabrication clinique et services d'analyse de lyophilisation. Le segment de la fabrication commerciale détenait la plus grande part de marché en 2022. Le segment du développement de cycles de lyophilisation devrait enregistrer le TCAC le plus élevé au cours de la période de prévision.
Services de lyophilisation pour le marché biopharmaceutique – Informations par type de service Selon les informations basées sur l'utilisateur final, le marché mondial des services de lyophilisation pour les produits biopharmaceutiques est segmenté en fonction de l'utilisateur final : sociétés pharmaceutiques et biotechnologiques, instituts de recherche, etc. Le segment des sociétés pharmaceutiques et biotechnologiques détenait la plus grande part de marché en 2022. Le segment des instituts de recherche devrait enregistrer le TCAC le plus élevé au cours de la période de prévision. Les entreprises du marché des services de lyophilisation pour les produits biopharmaceutiques adoptent des stratégies inorganiques et organiques, telles que les fusions et acquisitions. Voici quelques développements clés récents du marché : En novembre 2022, LTI a annoncé son engagement dans le développement, l'ingénierie des procédés et la préparation du matériel clinique pour le candidat vaccin contre la tuberculose ID93/GLA-SE. Le vaccin contre la tuberculose a atteint avec succès la phase 2 des essais cliniques. Cette étude représente le premier rapport sur la lyophilisation réussie d'un candidat vaccin sous-unitaire thermostable contenant un adjuvant à base d'émulsion.
- En mai 2022, Jubilant HollisterStier LLC a conclu un accord de coopération de 149,6 millions de dollars américains avec l'Army Contracting Command, en coordination avec le Joint Program Executive Office for Chemical, Biological, Radiological, and Nuclear Defense (JPEOCBRND) pour le compte de la Biomedical Advanced Research and Development Authority (BARDA), au sein du ministère américain de la Santé et des Services sociaux.
- En octobre 2021, PCI Pharma Services (PCI) a annoncé la signature d'un accord définitif pour l'acquisition de Lyophilization Servicesof New England, Inc. (LSNE), une organisation de développement et de fabrication sous contrat (CDMO) de premier plan dont le siège social est à Bedford, dans le New Hampshire, auprès de la société mondiale de capital-investissement Permira. Cette acquisition ajoute cinq installations approuvées par la FDA aux États-Unis (New Hampshire, Wisconsin) et en Europe (Espagne), une sixième devant être approuvée dans les prochains mois. De plus, trois installations supplémentaires sont en cours de développement. Ces installations renforceront son réseau mondial de 30 sites.
- En juillet 2021, Albany Molecular Research, Inc. (AMRI) a annoncé qu'elle changeait de nom pour devenir Curia, à compter du 12 juillet 2021. Le nouveau nom renforce le positionnement stratégique de l'entreprise en tant que CDMO mondial de bout en bout, appliquant son expertise scientifique et ses vastes capacités de recherche et développement (R&D) à la fabrication commerciale pour permettre à ses clients pharmaceutiques et biotechnologiques de faire progresser de nouveaux produits importants qui améliorent la vie.
- Services de lyophilisation pour le marché biopharmaceutiqueLes tendances et facteurs régionaux influençant le marché des services de lyophilisation pour produits biopharmaceutiques tout au long de la période de prévision ont été analysés en détail par les analystes de The Insight Partners. Cette section aborde également les segments et la géographie du marché des services de lyophilisation pour produits biopharmaceutiques en Amérique du Nord, en Europe, en Asie-Pacifique, au Moyen-Orient et en Afrique, ainsi qu'en Amérique du Sud et en Amérique centrale.Portée du rapport sur le marché des services de lyophilisation pour les produits biopharmaceutiques
| Attribut de rapport | Détails |
|---|---|
| Taille du marché en 2022 | US$ 2.05 Billion |
| Taille du marché par 2028 | US$ 3.59 Billion |
| TCAC mondial (2022 - 2028) | 9.9% |
| Données historiques | 2020-2021 |
| Période de prévision | 2023-2028 |
| Segments couverts |
By Type de service
|
| Régions et pays couverts |
Amérique du Nord
|
| Leaders du marché et profils d'entreprises clés |
|
Densité des acteurs du marché des services de lyophilisation pour les produits biopharmaceutiques : comprendre son impact sur la dynamique commerciale
Le marché des services de lyophilisation pour les produits biopharmaceutiques connaît une croissance rapide, portée par une demande croissante des utilisateurs finaux, due à des facteurs tels que l'évolution des préférences des consommateurs, les avancées technologiques et une meilleure connaissance des avantages du produit. Face à cette demande croissante, les entreprises élargissent leur offre, innovent pour répondre aux besoins des consommateurs et capitalisent sur les tendances émergentes, ce qui alimente la croissance du marché.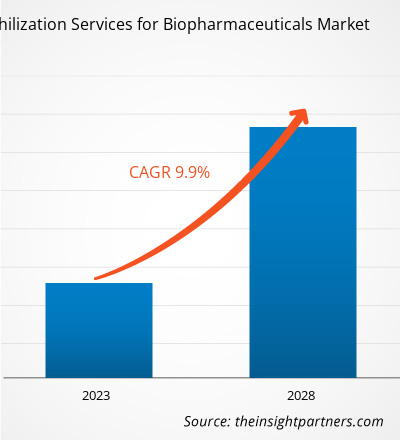
- Obtenez le Services de lyophilisation pour le marché des produits biopharmaceutiques Aperçu des principaux acteurs clés
Profils d'entreprise - Services de lyophilisation pour le marché biopharmaceutique
- ATTWILL Medical Solutions
- Axcellerate Pharma LLC
- Labyrinth Biopharma LLC
- Berkshire Sterile Manufacturing
- PCI Pharma Services
- Curia Global Inc
- Emergent BioSolutions Inc
- Jubilant HollisterStier LLC
- Biofortuna
- Lyophilization Technology Inc.
- SYNERLAB GROUP
- Analyse historique (2 ans), année de base, prévision (7 ans) avec TCAC
- Analyse PEST et SWOT
- Taille du marché Valeur / Volume - Mondial, Régional, Pays
- Industrie et paysage concurrentiel
- Ensemble de données Excel
Témoignages
Raison d'acheter
- Prise de décision éclairée
- Compréhension de la dynamique du marché
- Analyse concurrentielle
- Connaissances clients
- Prévisions de marché
- Atténuation des risques
- Planification stratégique
- Justification des investissements
- Identification des marchés émergents
- Amélioration des stratégies marketing
- Amélioration de l'efficacité opérationnelle
- Alignement sur les tendances réglementaires















Débloquez des remises exclusives sur les rapports
Demander maintenant






 Obtenez un échantillon gratuit pour - Services de lyophilisation pour le marché des produits biopharmaceutiques
Obtenez un échantillon gratuit pour - Services de lyophilisation pour le marché des produits biopharmaceutiques