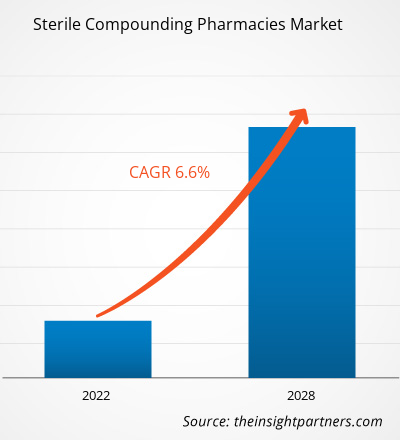
Le marché mondial des pharmacies de préparation stérile devrait atteindre 8,99 milliards de dollars américains d'ici 2034, contre 5,10 milliards de dollars américains en 2025. Ce marché devrait enregistrer un TCAC de 6,5 % entre 2026 et 2034.
Analyse du marché des pharmacies de préparation stérile
Le marché des pharmacies de préparation stérile connaît une transformation majeure, impulsée par la demande croissante de médecine personnalisée de haute précision et par l'externalisation des préparations stériles complexes par les réseaux hospitaliers. L'analyse de marché révèle que la croissance stratégique se concentre en oncologie et en nutrition clinique, où les posologies standardisées ne répondent souvent pas aux besoins des patients pédiatriques ou gériatriques. Le marché s'oriente vers des établissements d'externalisation agréés « 503B » aux États-Unis et des organismes similaires dans d'autres pays. Les hôpitaux privilégient ces solutions pour réduire les risques et les coûts liés à l'exploitation de leurs propres salles blanches, qui doivent se conformer aux normes USP <797> et <800> en constante évolution. L'intégration de systèmes robotisés de préparation automatisés offre des opportunités stratégiques, qui non seulement renforcent l'asepsie, mais fournissent également le débit basé sur les données nécessaire pour gérer l'augmentation des pénuries de médicaments injectables spécialisés.
Aperçu du marché des pharmacies de préparation stérile
Le marché des pharmacies de préparation stérile est essentiel au système de santé, car il permet de préparer des médicaments personnalisés exempts de micro-organismes et de pyrogènes. Ce secteur respecte des normes d'asepsie rigoureuses pour des produits tels que les solutions intraveineuses, les injections intramusculaires et les traitements sous-cutanés. De plus en plus d'entreprises délaissent la préparation manuelle au profit de services basés sur des technologies de pointe afin d'améliorer la sécurité des patients sous chimiothérapie, nutrition parentérale totale et prise en charge spécialisée de la douleur. Face à la prévalence croissante des maladies chroniques nécessitant des traitements injectables, les pharmacies de préparation stérile contribuent au bon fonctionnement des services de soins aigus en permettant aux hôpitaux de proposer des soins pharmaceutiques personnalisés, tout en garantissant leur stabilité financière et leur conformité réglementaire.
Personnalisez ce rapport selon vos besoins.
Bénéficiez d'une PERSONNALISATION GRATUITEMarché des pharmacies de préparation stérile : perspectives stratégiques

-
Découvrez les principales tendances du marché présentées dans ce rapport.Cet échantillon GRATUIT comprendra une analyse de données, allant des tendances du marché aux estimations et prévisions.
Facteurs et opportunités du marché des pharmacies de préparation stérile
Facteurs de marché :
- Demande croissante d'efficacité opérationnelle dans les soins de santé : les établissements de santé constatent une augmentation du nombre de patients nécessitant des traitements injectables complexes, ce qui complexifie le travail des pharmacies internes. Par conséquent, de plus en plus de prestataires externalisent les tâches de préparation à haut risque auprès de services de préparation stérile afin d'assurer la continuité de leurs activités.
- Demande croissante de médicaments personnalisés et de thérapies injectables : Avec la généralisation de la médecine de précision, le besoin en préparations magistrales stériles s’est accru. Les dosages adaptés en oncologie et en soins intensifs font désormais partie intégrante des soins de santé, et non plus un service spécialisé.
- Renforcement des initiatives gouvernementales et des obligations réglementaires : les préparations magistrales stériles sont soumises à une application généralisée des normes de sécurité telles que la loi sur la qualité et la sécurité des médicaments (DQSA). Les établissements de santé sont incités à recourir à des services de préparation magistrale professionnels afin d’abandonner les processus internes à haut risque et non conformes.
Opportunités de marché :
- Expansion sur les marchés émergents avec des secteurs de soins de santé privés en croissance : les marchés émergents tels que l’Inde, la Chine et le Brésil connaissent une adoption accrue de la préparation stérile à mesure que les réseaux d’hôpitaux privés se développent et que les investissements dans les cliniques de soins spécialisés augmentent.
- Intégration aux plateformes d'automatisation et de préparation robotisée : L'intégration de la préparation stérile à la robotique avancée permet une préparation des médicaments fluide et sans erreur. L'automatisation pilotée par l'IA améliore les décisions cliniques et l'efficacité du système grâce à une analyse intelligente des volumes.
- Demande de soins spécialisés et de perfusion à domicile : L’essor des infrastructures de soins à domicile est un facteur clé de la demande de perfusions stériles externalisées. Ces services optimisent les flux de travail et permettent une utilisation efficace des ressources pour la prise en charge à long terme des patients.
Analyse de segmentation du rapport sur le marché des pharmacies de préparation stérile
Le marché des pharmacies de préparation stérile est analysé selon différents segments afin de mieux comprendre sa structure, son potentiel de croissance et les tendances émergentes. Voici l'approche de segmentation standard utilisée dans la plupart des rapports sectoriels :
Par produit :
- Médicaments injectables
- perfusions
Par voie d'administration :
- Intraveineux (IV)
- Intramusculaire
- Sous-cutané
Par géographie :
- Amérique du Nord
- Europe
- Asie-Pacifique
- Amérique du Sud et centrale
- Moyen-Orient et Afrique
Aperçu régional du marché des pharmacies de préparation stérile
Les tendances régionales et les facteurs influençant le marché des pharmacies de préparations stériles tout au long de la période prévisionnelle ont été analysés en détail par les analystes de The Insight Partners. Cette section aborde également les segments de marché et la répartition géographique du marché des pharmacies de préparations stériles en Amérique du Nord, en Europe, en Asie-Pacifique, au Moyen-Orient et en Afrique, ainsi qu'en Amérique du Sud et centrale.
Portée du rapport sur le marché des pharmacies de préparation stérile
| Attribut du rapport | Détails |
|---|---|
| Taille du marché en 2025 | 5,10 milliards de dollars américains |
| Taille du marché d'ici 2034 | 8,99 milliards de dollars américains |
| TCAC mondial (2026 - 2034) | 6,5% |
| Données historiques | 2021-2024 |
| Période de prévision | 2026-2034 |
| Segments couverts |
Sous-produit
|
| Régions et pays couverts |
Amérique du Nord
|
| Leaders du marché et profils d'entreprises clés |
|
Densité des acteurs du marché des pharmacies de préparation stérile : comprendre son impact sur la dynamique commerciale
Le marché des pharmacies de préparations stériles connaît une croissance rapide, portée par une demande croissante des utilisateurs finaux. Cette demande est alimentée par l'évolution des préférences des consommateurs, les progrès technologiques et une meilleure connaissance des avantages du produit. Face à cette demande grandissante, les entreprises diversifient leur offre, innovent pour répondre aux besoins des consommateurs et tirent parti des tendances émergentes, ce qui contribue à stimuler la croissance du marché.
- Obtenez un aperçu des principaux acteurs du marché des pharmacies de préparation stérile.
Analyse des parts de marché des pharmacies de préparation stérile par zone géographique
La région Asie-Pacifique devrait connaître la croissance la plus rapide dans les années à venir. Les marchés émergents d'Amérique du Sud et centrale, du Moyen-Orient et d'Afrique offrent également de nombreuses opportunités inexploitées pour le développement des centres d'externalisation et des prestataires de services de formulation spécialisés.
Le marché des pharmacies de préparation stérile présente une évolution différente selon les régions, en raison de facteurs tels que la rigueur réglementaire (par exemple, la conformité à la norme USP <797>), les tendances à l'externalisation hospitalière et la demande croissante de traitements personnalisés en nutrition parentérale et en oncologie. Vous trouverez ci-dessous un résumé des parts de marché et des tendances par région :
Amérique du Nord
- Part de marché : Détient la plus grande part de marché grâce à une infrastructure de soins de santé avancée et à l'adoption précoce des modèles d'externalisation 503B.
-
Facteurs clés :
- De nombreuses organisations suivent les normes USP <797> et <800> pour répondre aux exigences réglementaires.
- Il existe une forte demande pour des perfusions intraveineuses spécialisées, prêtes à l'emploi, en oncologie et en gestion de la douleur.
- Les grandes entreprises d'externalisation, telles que QuVa Pharma et Nephron, jouent un rôle majeur sur le marché.
- Tendances : Une forte évolution vers la préparation robotisée et la coordination automatisée des flux de travail afin d'éliminer les erreurs humaines et de réduire les risques de contamination dans les installations à haut volume.
2. Europe
- Part de marché : Déterminée par les systèmes de santé publique et une gouvernance stricte en matière de sécurité, avec une forte emphase sur la préparation magistrale en milieu hospitalier.
-
Facteurs clés :
- Projets nationaux de numérisation visant à unifier les dossiers médicaux électroniques et les ordonnances pharmaceutiques
- Besoin croissant d'efficacité dans les services hospitaliers multiservices pour gérer les pénuries de médicaments
- Adoption croissante de « centres de préparation magistrale centralisés » pour desservir plusieurs cliniques
- Tendances : Utilisation croissante de plateformes interopérables pour soutenir les initiatives sanitaires transfrontalières et la normalisation des protocoles de préparation aseptique dans toute l'UE.
3. Asie-Pacifique
- Part de marché : Région à la croissance la plus rapide grâce à la modernisation rapide du système de santé et à l’expansion du tourisme médical en Chine, en Inde et au Japon.
-
Facteurs clés :
- Des programmes gouvernementaux comme « Digital India » et « Healthy China 2030 » sont des moteurs de changement dans ce secteur.
- De plus en plus de prestataires de soins de santé privés investissent désormais dans leurs propres salles stériles.
- Les maladies chroniques nécessitant un traitement parentéral à long terme sont de plus en plus fréquentes.
- Tendances : Adoption rapide des systèmes automatisés de préparation magistrale pour la distribution de médicaments spécialisés sur rendez-vous, notamment dans les grands centres urbains de pédiatrie et d'oncologie.
4. Amérique du Sud et centrale
- Part de marché : Marché émergent avec une transition ciblée vers la production locale de médicaments stériles personnalisés.
-
Facteurs clés :
- Modernisation des réseaux hospitaliers privés au Brésil, au Mexique et en Colombie
- L’évolution de la réglementation (par exemple, INVIMA et ANVISA) pousse vers des normes d’asepsie plus élevées.
- Forte demande pour les traitements hormonaux substitutifs et les thérapies anti-âge personnalisés
- Tendances : Dépendance croissante à l'égard des transferts de technologie mondiaux (par exemple, de fournisseurs comme Fagron) pour établir des laboratoires stériles conformes aux normes de sécurité élevées des hôpitaux locaux.
5. Moyen-Orient et Afrique
- Part de marché : Marché en développement présentant un potentiel de croissance important, notamment dans les villes médicales de pointe du CCG.
-
Facteurs clés :
- Stratégies nationales de santé numérique et essor des « hôpitaux intelligents » en Arabie saoudite et aux Émirats arabes unis
- Investissement dans les infrastructures biopharmaceutiques et de préparation magistrale locales pour sécuriser la chaîne d'approvisionnement
- Demande croissante de formulations stériles spécialisées pour les enfants et les nouveau-nés
- Tendances : Mise en place de salles robotisées spécialisées dans les nouvelles « villes de la santé » afin de soutenir la médecine de précision et de réduire la dépendance aux produits injectables spécialisés importés.
Densité des acteurs du marché des pharmacies de préparation stérile : comprendre son impact sur la dynamique commerciale
Forte densité de marché et concurrence
La concurrence s'intensifie du fait de la présence de leaders mondiaux de l'industrie pharmaceutique et de la préparation magistrale tels que Fagron, Inc., B. Braun Melsungen AG et Fresenius Kabi AG. Le paysage est également façonné par d'importants centres d'externalisation agréés 503B et des prestataires régionaux spécialisés comme Avella Specialty Pharmacy et Pace Pharmacy (Canada), créant ainsi un marché très concurrentiel axé sur le respect des protocoles cliniques.
Ce contexte concurrentiel pousse les fournisseurs à se différencier par :
- Automatisation robotique avancée : tirer parti de la robotique de préparation des solutions intraveineuses et des systèmes de distribution automatisés pour éliminer les erreurs humaines, améliorer le débit et garantir la plus haute précision dans les préparations stériles.
- Alignement réglementaire rigoureux : Investissement important dans une infrastructure conforme aux normes USP <797> et USP <800>, y compris des salles blanches modulaires et une surveillance environnementale continue pour garantir l'intégrité des données et la sécurité des patients.
- Produits biologiques et injectables personnalisés : développement de formulations spécifiques aux patients pour l’oncologie, la nutrition parentérale et les maladies rares que les fabricants commerciaux ne peuvent pas fournir aux doses standard.
- Solutions de traçabilité numérique : mise en œuvre d’un suivi basé sur la blockchain et de dossiers de lots électroniques pour assurer une visibilité en temps réel tout au long du cycle de production, de l’approvisionnement en ingrédients à la livraison finale.
Opportunités et initiatives stratégiques
- Collaborer avec de grands réseaux hospitaliers et centres chirurgicaux pour passer du mélange interne à l'externalisation spécialisée 503B, réduisant ainsi les frais généraux et contribuant à remédier aux pénuries locales de médicaments.
- Utilisez des outils de contrôle qualité basés sur l'IA et de gestion prédictive des stocks pour améliorer l'approvisionnement en mélanges stériles importants, notamment dans les domaines à forte demande tels que l'anesthésie et les soins intensifs.
Les principales entreprises opérant sur le marché des pharmacies de préparation stérile sont :
- Fagron, Inc. – Pays-Bas
- B. Braun Melsungen AG – Allemagne
- Fresenius Kabi AG – Allemagne
- PharMedium Healthcare Holdings, Inc. – États-Unis
- Pharmacie spécialisée Avella – États-Unis
- Pharmacie de préparation magistrale Triangle – États-Unis
- Pharmacie de préparation magistrale Pencol – États-Unis
- Pavilion Compounding Pharmacy, LLC – États-Unis
- Pharmacie Pace – Canada
Avertissement : Les entreprises mentionnées ci-dessus ne sont classées dans aucun ordre particulier.
Actualités et développements récents du marché des pharmacies de préparation stérile
- En septembre 2025, Medisca s'est associée au groupe Stevanato pour distribuer les flacons stériles prêts à l'emploi EZ-fill®, élargissant ainsi l'accès à des solutions de confinement de haute qualité pour l'industrie pharmaceutique, améliorant l'efficacité opérationnelle et la sécurité des patients.
- En décembre 2025, Wolters Kluwer a lancé son module de préparation avancée pour Simplifi 797, offrant aux pharmacies de préparation stérile une solution complète pour répondre aux normes actualisées du chapitre 797 de l'USP. Ce module prend en charge les procédés stériles à haut risque, le suivi des lots, la gestion des dates de péremption et une documentation améliorée, tout en fournissant des ressources intégrées de formation et de conformité pour le personnel officinal.
Rapport sur le marché des pharmacies de préparation stérile : contenu et livrables
Le rapport « Taille et prévisions du marché des pharmacies de préparation stérile (2021-2034) » fournit une analyse détaillée du marché couvrant les domaines suivants :
- Taille et prévisions du marché des pharmacies de préparation stérile aux niveaux mondial, régional et national pour tous les segments de marché clés couverts par le périmètre de l'étude
- Tendances du marché des pharmacies de préparation stérile, ainsi que sa dynamique, notamment les facteurs de croissance, les contraintes et les principales opportunités.
- Analyse PEST et SWOT détaillée
- Analyse du marché des pharmacies de préparation stérile couvrant les principales tendances du marché, le cadre mondial et régional, les principaux acteurs, la réglementation et les développements récents du marché
- Analyse du paysage industriel et de la concurrence, incluant la concentration du marché, une analyse par carte thermique, les principaux acteurs et les développements récents sur le marché des pharmacies de préparation stérile.
- Profils d'entreprise détaillés
- Analyse historique (2 ans), année de base, prévision (7 ans) avec TCAC
- Analyse PEST et SWOT
- Taille du marché Valeur / Volume - Mondial, Régional, Pays
- Industrie et paysage concurrentiel
- Ensemble de données Excel
Rapports récents
Témoignages
Raison d'acheter
- Prise de décision éclairée
- Compréhension de la dynamique du marché
- Analyse concurrentielle
- Connaissances clients
- Prévisions de marché
- Atténuation des risques
- Planification stratégique
- Justification des investissements
- Identification des marchés émergents
- Amélioration des stratégies marketing
- Amélioration de l'efficacité opérationnelle
- Alignement sur les tendances réglementaires






















 Obtenez un échantillon gratuit pour - Marché des pharmacies de préparation stérile
Obtenez un échantillon gratuit pour - Marché des pharmacies de préparation stérile