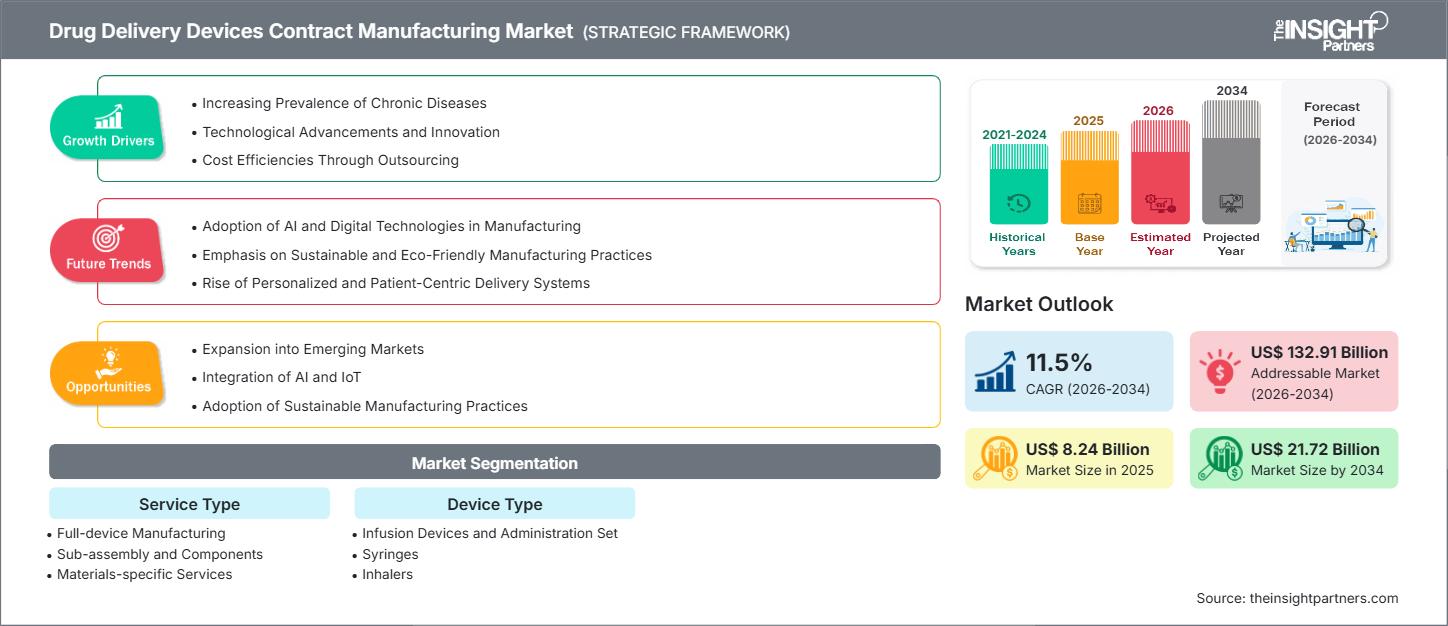
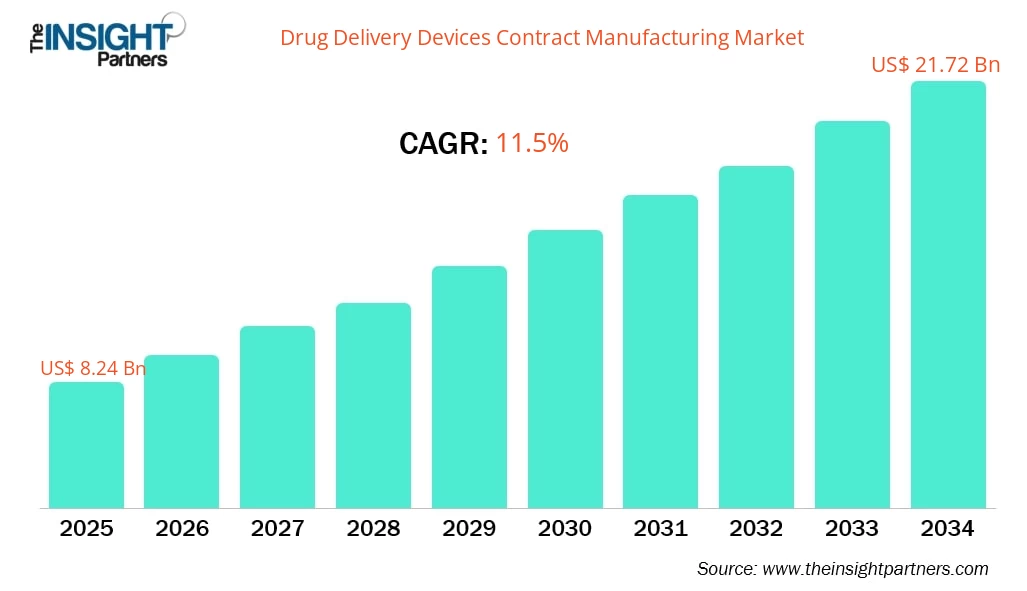
The drug delivery devices contract manufacturing market size is projected to reach US$ 21.72 billion by 2034 from US$ 8.24 billion in 2025. The market is expected to register a CAGR of 11.5% during 2026–2034.
Drug Delivery Devices Contract Manufacturing Market Analysis
The market growth is driven by the increasing prevalence of chronic diseases, technological advancements and innovation, and cost efficiencies through outsourcing. Expansion into emerging markets and integration of advanced technologies like AI and IoT are expected to create ample opportunities.
Drug Delivery Devices Contract Manufacturing Market Overview
The pharmaceutical and medical device companies are procuring the manufacture of inhalers, autoinjectors, infusion pumps, and wearable drug delivery devices through contract manufacturers. The complexity of modern drug delivery technology, as well as the increasing regulatory responsibilities and cost efficiencies associated with scale production, are the primary reasons leading to outsourcing. Additionally, the growing prevalence of chronic illnesses, the increase in personalized medicines, and the growth of biologic therapies are increasing the demand for tailored solutions designed for the specific needs of patients and the environment in which they reside. Drug delivery devices are one of the fastest-growing segments of the medical device contract manufacturing market as a result of a continued embrace by the industry of collaboration and specialization.
Market Assessment and Insights
- North America dominated the market with 35.5% share in 2025.
- Asia Pacific is poised to grow at a CAGR of 12% over the forecast period.
- United States market is projected to grow at a CAGR of 11.8% over the forecast period.
- By Service Type, the Full-device Manufacturing segment accounted for the largest market share of 62.5% in 2025.
- By Device Type, the Syringes segment is anticipated to witness the fastest growth, registering a CAGR of 12.1% over the forecast period
- The report profiles key industry players such as Freudenberg Group, TE Connectivity Ltd, Jabil Inc, Integer Holdings Corp, Heraeus Group, Flex Ltd, Cirtec Medical Corp, Tegra Medical, SMC Ltd, MICRO, while also analyzing key developments in novel ideas, disruptive products, and innovative services that could reshape the future market and reveal emerging themes across the industry.
Customize This Report To Suit Your Requirement
Get FREE CUSTOMIZATIONDrug Delivery Devices Contract Manufacturing Market: Strategic Insights
-
Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
Drug Delivery Devices Contract Manufacturing Market Drivers and Opportunities
Market Drivers:
- Increasing Prevalence of Chronic Diseases: Chronic diseases (cardiovascular, respiratory, and diabetes) are becoming common globally. As a result, there is an increase in the demand for scalable drug delivery devices, prompting pharmaceutical firms to seek third-party specialized manufacturing solutions to enable patients to remain well for the long term.
- Technological Advancements and Innovation: New technologies such as smart devices, nanotechnology, wearable devices, and microneedling have introduced complexity to the manufacturing of drug delivery devices to the point where firms are choosing to work with contract manufacturers who specialize in precision engineering, regulatory compliance, and high-volume custom manufacturing of these drug delivery devices.
- Cost Efficiencies Through Outsourcing: By outsourcing the manufacturing of drug delivery devices, pharmaceutical manufacturers reduce the amount of capital invested, reduce labor and operating expenses, and realize economies of scale, allowing them to mitigate risk, expedite time to market, and focus on research and innovation.
Market Opportunities:
- Expansion into Emerging Markets: Emerging technologies provide contract manufacturers with lower labor costs, burgeoning labor pools with substantial experience, and a variety of favorable governmental policies, making them an ideal candidate for the manufacture and distribution of cost-effective drug delivery devices while creating robust supply chain networks in high burden areas.
- Integration of Advanced Technologies like AI and IoT: Integrating AI and IoT into drug delivery devices allows them to become smarter. It improves the way manufacturers can get products to market at scale, optimizing processes through improved manufacturing methods and better quality control. Leveraging predictive maintenance to manufacture drug delivery devices provides patients with greater adherence and supports the use of data in health care.
- Adoption of Sustainable Manufacturing Practices: Green manufacturing offers the opportunity for manufacturers to use environmentally friendly materials, operate in an energy-efficient manner, and develop circular manufacturing processes, in order for contract manufacturers to comply with environmental regulations and requirements of ESG, reduce their costs, and provide pharma partners with responsible innovation.
Drug Delivery Devices Contract Manufacturing Market Report Segmentation Analysis
The drug delivery devices contract manufacturing market is divided into different segments to give a clearer view of how it works, its growth potential, and the latest trends. Below is the standard segmentation approach used in most industry reports:
By Service Type:
- Full-device Manufacturing: Full-device manufacturing includes outsourced manufacturing from start to finish and gives manufacturers assurance of meeting regulatory compliance standards along with faster commercialization, cost-effectiveness, scalability, and innovative solutions to meet patient-specific needs for complex drug delivery systems and therapies.
- Sub-assembly and Components: Fabrication of sub-assemblies and components is offered by companies that provide manufacturers with precision-engineered modular pieces to reduce costs, improve flexibility, and increase supply chain resilience; moreover, by reducing reliance on multiple suppliers and allowing for increased manufacturing volume.
- Materials-specific Services: Material-specific services are focused on selecting biocompatible and environmentally sustainable materials to manufacture devices that will be stable and comply with regulatory requirements while providing cost-effective performance for advanced drug delivery systems.
By Device Type:
- Infusion Devices and Administration Set
- Syringes
- Inhalers
- Autoinjectors and Pen Injectors
Each end user in the market has distinct handling, safety, and regulatory needs, shaping equipment selection and operational protocols.
By Geography:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
Drug Delivery Devices Contract Manufacturing Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2025 | US$ 8.24 Billion |
| Market Size by 2034 | US$ 21.72 Billion |
| Global CAGR (2026 - 2034) | 11.5% |
| Historical Data | 2021-2024 |
| Forecast period | 2026-2034 |
| Segments Covered |
By Service Type
|
| Regions and Countries Covered |
North America
|
| Market leaders and key company profiles |
|
Drug Delivery Devices Contract Manufacturing Market Players Density: Understanding Its Impact on Business Dynamics
The Drug Delivery Devices Contract Manufacturing Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
Drug Delivery Devices Contract Manufacturing Market Share Analysis by Geography
The drug delivery devices contract manufacturing market in Asia Pacific is witnessing the fastest growth, driven by the increasing prevalence of chronic disorders and rising demand for outsourcing. Emerging markets in Latin America, the Middle East, and Africa have untapped opportunities for drug delivery devices contract manufacturing providers to expand.
The drug delivery devices contract manufacturing market grows differently across regions, as economic growth and rising healthcare expenditure. Below is a summary of market share and trends by region:
1. North America
- Market Share: Holds a significant portion of the global market
- Key Drivers:
- Highly developed healthcare marketplace, with a high level of R&D spending and substantial governmental regulatory support for the development of complex drug delivery devices.
- Trends: Rapid adoption of advanced and connected delivery systems
2. Europe
- Market Share: Substantial share due to early adoption of drug delivery devices contract manufacturing
- Key Drivers:
- Stringent requirements regarding product quality standards and product innovation are driving the demand for outsourcing advanced delivery technologies.
- Trends: Focus on high-quality biologics and compliance-oriented outsourcing in a harmonized regulatory environment
3. Asia Pacific
- Market Share: Fastest-growing region with rising market share every year
- Key Drivers:
- The combination of having a lower cost of performing such manufacturing, as well as government incentives, is leading to the increasing demand for contract Manufacturing for large-volume drug delivery devices.
- Trends: Cost advantages, expanding manufacturing capabilities, and increasing generics/biosimilars production
4. South and Central America
- Market Share: Steadily progressing market share
- Key Drivers:
- Better access to healthcare, an increasing burden of chronic disease, and a rise in local manufacturing of pharmaceutical products.
- Trends: Growing local manufacturing partnerships and healthcare access improvements
5. Middle East and Africa
- Market Share: Small market share, growing at a rapid pace
- Key Drivers:
- The increased investment into healthcare and governmental support, as well as an increasing rate of occurrence of chronic diseases.
- Trends: Rising healthcare investments and development of domestic pharmaceutical hubs
High Market Density and Competition
Competition is strong due to the presence of established players, such as Jabil Inc, Flex Ltd, and TE Connectivity Ltd. Regional and niche players are adding to the competitive landscape.
This high level of competition urges companies to stand out by offering:
- Advanced security features
- Value-added services such as analytics and predictive maintenance, real‑time operational analytics, and installation
- Competitive pricing models
- Strong customer support and easy integration
Opportunities and Strategic Moves
- A significant expansion of the biologics industry has emerged with the emergence of GLP-1 therapies. The vast majority (80%) of these are composed of injectable products and represent a growing need for a new category of drug delivery device. Large-volume subcutaneous (LVSC) drug delivery devices represent approximately 15% of all biologics in clinical development, requiring doses of 2 mL-5 mL and which allows for outpatient and home-based administration and increases the patient's independence and comfort level of treatment. Increased need for innovative methods to deliver cell and gene therapies is expanding into this category of product delivery systems, as new regulatory risks are associated with all types of cell and gene therapies.
- Businesses are focusing on environmentally friendly manufacturing practices, using minimal waste materials, and investing in reusable injector systems, to meet ESG requirements and address the requests of payers. These businesses are incorporating digital technology for precise dosing as well as secure data storage. Other trends, such as reshoring and nearshoring, along with remote inspection processes, support enhanced resiliency.
Other companies analyzed during the course of research:
- Sanmina Corporation
- Plexus Corp.
- West Pharmaceutical Services, Inc.
- Nipro Corporation
- Celestica Inc.
- Benchmark Electronics, Inc.
- Nemera Development S.A.
- Viant Medical Holdings, Inc.
- Tecomet, Inc.
- SMC Ltd.
- Phillips‑Medisize Corporation
- Tessy Plastics Corp.
- MeHow (China)
- Tekni‑Plex
- Peter’s Technology
- Teleflex Incorporated
- Nordson Corporation
- Elos Medtech AB
- AMETEK, Inc.
- Danaher Corporation
- Gerresheimer AG
- Imball Electronics, Inc.
- Nortech Systems, Inc.
- Carclo Plc
- Nolato GW, Inc.
- Angiplast Private Limited
- Forefront Medical Technologies
- Cadence Device (Cadence, Inc.)
- Seasky Medical
- Scapa
- Spectrum Solution
- NAGL MedTech
- Meridian Medical
- MME Group Inc.
- Providence Enterprise USA, Inc.
- HDA Technology
- Advantech Plastics, LLC
- Europlaz Ltd.
- Stevanato Group
- Elcam Drug Delivery Devices (E3D)
- Ypsomed Delivery Systems (YDS)
- SHL Medical
- Haselmeier
- Owen Mumford
- CCBio / Copernicus (Nemera)
- Solteam Medical
Drug Delivery Devices Contract Manufacturing Market News and Recent Developments
- Jubilant Biosys Announces New Facility in India: In January 2026, Jubilant Biosys Limited announced that it is opening a discovery and preclinical facility in Noida, India. The new facility will double the contract research, development, and manufacturing organization’s (CRDMO) chemistry capacity. It will combine discovery and early-phase scale-up capabilities with R7B labs and two pilot plants. This expansion will meet the company’s demand for fee-for-service contracts.
- Stevanato Group Expands Drug Delivery Systems Capacity in Germany: In October 2025, Stevanato Group expanded its drug delivery system manufacturing capacity. The company recently added a production space for drug delivery devices to its facility in Bad Oeynhausen, northern Germany. This initiative provides more than 2,500 square meters of advanced manufacturing capacity.
Drug Delivery Devices Contract Manufacturing Market Report Coverage and Deliverables
The "Drug Delivery Devices Contract Manufacturing Market Size and Forecast (2021–2034)" report provides a detailed analysis of the market covering below areas:
- Drug Delivery Devices Contract Manufacturing Market size and forecast at global, regional, and country levels for all the segments covered under the scope
- Drug Delivery Devices Contract Manufacturing Market trends, as well as dynamics such as drivers, restraints, and key opportunities
- Detailed PEST and SWOT analysis
- Drug Delivery Devices Contract Manufacturing Market analysis covering key trends, global and regional framework, major players, regulations, and recent developments
- Industry landscape and competition analysis covering market concentration, heat map analysis, prominent players, and recent developments for the drug delivery devices contract manufacturing market
- Detailed company profiles
Frequently Asked Questions
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Global, Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Recent Reports
Testimonials
Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends
Unlock Exclusive Report Discounts
Enquire Now






 Get Free Sample For
Get Free Sample For