Sepsis Diagnostics Market Size, Growth & Trends by 2034
Sepsis Diagnostics Market Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Product (Instruments, Reagents and Assays, Blood Culture Media, and Software), Technology [Molecular Diagnostics (Polymerase Chain Reaction, Peptide Nucleic Acid-Fluorescent in Situ Hybridization, Syndromic Panel-Based Testing, and Microarray), Flow Cytometry, Microfluidics, Immunoassay, Biomarker, and Microbiology], Method (Automated Diagnostics and Conventional Diagnostics), Test Type (Point-of-Care Tests and Laboratory Tests), Pathogen (Bacterial Sepsis, Fungal Sepsis, and Others), and End User (Hospitals, Pathology and Reference Laboratories, and Others)
Historic Data: 2021-2024 | Base Year: 2025 | Forecast Period: 2026-2034- Status : Data Released
- Report Code : TIPHE100001391
- Category : Life Sciences
- No. of Pages : 150
- Available Report Formats :


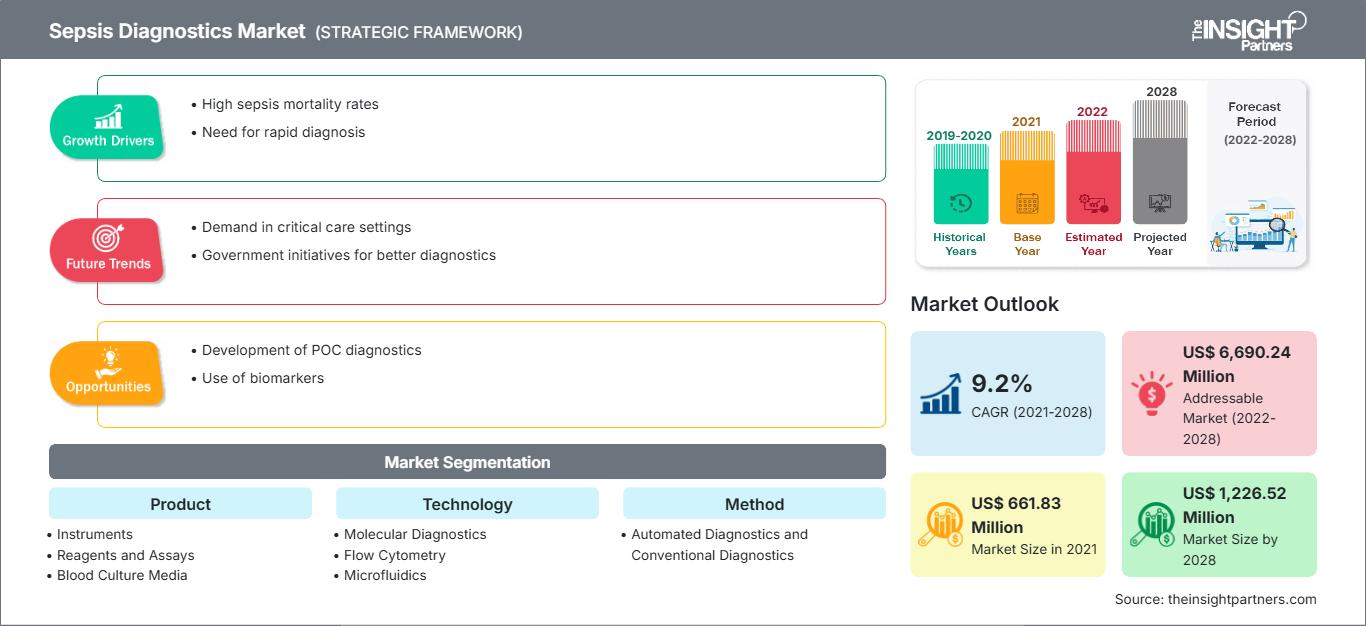
The global Sepsis Diagnostics Market size is projected to reach US$ 1558.98 million by 2034 from US$ 844 Million in 2025. The market is anticipated to register a CAGR of 7.06% during the forecast period 2026–2034
Key market dynamics include the rising global incidence of sepsis particularly hospital-acquired infections (HAIs), advancements in rapid molecular diagnostic tools, and increasing government initiatives to improve early detection protocols. Additionally, the market is expected to benefit from the growing geriatric population, the surge in antimicrobial resistance (AMR), and the rising demand for point-of-care (POC) testing to reduce critical turnaround times in emergency departments.
Sepsis Diagnostics Market Analysis
The sepsis diagnostics market analysis shows a shift toward host-response-based testing as medical professionals prioritize understanding the patient’s immune status alongside pathogen identification. The market is splitting into high-volume, centralized laboratory microbiology and high-growth, decentralized molecular assays in intensive care units. Strategic opportunities are emerging in the development of syndromic panels that detect co-infections and antibiotic resistance genes in a single run, offering a clear competitive advantage over traditional sequential testing. The analysis also notes that market expansion depends on the seamless integration of diagnostic results into Electronic Health Records (EHR) and the ability of manufacturers to prove cost-savings by reducing the length of hospital stays. Competitive differentiation now stands out depending on the analytical speed and diagnostic accuracy that allows for the de-escalation of broad-spectrum antibiotics, supporting global antimicrobial stewardship goals.
Sepsis Diagnostics Market Overview
Sepsis diagnostics have evolved from niche laboratory procedures to a central pillar of hospital critical care and emergency medicine. Sepsis diagnostics include automated blood culture systems, high-throughput molecular platforms, and bedside point-of-care devices. Both established medical technology giants and specialized biotech startups compete in this market, utilizing technologies such as PCR, NGS (Next-Generation Sequencing), and microfluidics. Growing demand for decentralized testing in North America and Europe has increased the popularity of POC solutions as a life-saving diagnostic tool. North America leads in revenue due to its advanced healthcare infrastructure and high adoption of innovative technologies, while Asia-Pacific is advancing rapidly due to improving medical facilities and a large patient pool. The US market is the most developed, driven by rigorous sepsis awareness programs and favorable reimbursement policies for advanced diagnostic tests.
Customize This Report To Suit Your Requirement
Get FREE CUSTOMIZATIONSepsis Diagnostics Market: Strategic Insights
-
Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
Sepsis Diagnostics Market Drivers and Opportunities
Market Drivers:
- High Incidence of Hospital-Acquired Infections (HAIs): Sepsis is a leading cause of death in hospitals worldwide. The rising frequency of infections following invasive surgeries and ICU admissions is a primary driver for robust diagnostic adoption.
- Technological Shift toward Rapid Molecular Diagnostics: Traditional culture methods can take 24–72 hours. New molecular tools that provide results in under 6 hours are revolutionizing the market by enabling timely clinical decisions.
- Growing Geriatric and Immunocompromised Populations: An aging global demographic and an increase in chronic diseases have created a larger at-risk population vulnerable to septic shock.
Market Opportunities:
- Development of Novel Biomarkers and Multiplex Panels: There is a significant opportunity for diagnostic tests that can differentiate between bacterial, viral, and fungal sepsis at an early stage.
- Expansion in Emerging APAC and LAMEA Markets: Increasing healthcare expenditure and the formalization of clinical protocols in China, India, and Brazil offer untapped growth potential for global diagnostic providers.
- AI-Driven Predictive Analytics: Integrating diagnostic hardware with software that provides real-time risk scores for patients enables a proactive rather than reactive approach to sepsis management.
Sepsis Diagnostics Market Report Segmentation Analysis
The Sepsis Diagnostics Market share is analyzed across various segments to provide a clearer understanding of its structure, growth potential, and emerging trends. Below is the standard segmentation approach used in most industry reports:
By Product:
- Instruments: Includes automated blood culture systems, PCR machines, and POC devices that serve as the hardware foundation for sepsis detection.
- Reagents and Assays: The recurring revenue driver, consisting of the chemical components used to perform molecular, immunoassay, and biochemical tests.
- Blood Culture Media: Specialized nutrient solutions required to grow and identify pathogens from patient blood samples; remains a core component of microbiology workflows.
- Software: The fastest-growing segment, focusing on AI-based clinical decision support and lab data management platforms.
By Technology:
- Molecular Diagnostics: High-growth segment utilizing PCR and DNA microarrays for rapid pathogen identification and antibiotic resistance detection.
- Flow Cytometry: Used to analyze the physical and chemical characteristics of cells, often helping in immune response monitoring.
- Microfluidics: An emerging technology enabling the miniaturization of diagnostic processes for point-of-care applications.
- Immunoassay: Focuses on measuring host-response markers like Procalcitonin (PCT) and Interleukins.
- Biomarker: Involves the identification of specific biological indicators that signal the onset or severity of sepsis.
- Microbiology: The traditional gold standard technology, involving the cultivation of microorganisms from blood samples.
By Method:
- Automated Diagnostics: Modern systems that reduce human error and turnaround time through robotic sample processing and digital reading.
- Conventional Diagnostics: Manual laboratory techniques that rely on the expertise of technicians and traditional culturing timelines.
By Test Type:
- Laboratory Tests: Comprehensive, high-accuracy tests performed in a centralized hospital or reference labs.
- Point-of-Care Tests: Rapid tests performed at the patient's bedside, primarily in emergency rooms or ICUs, to facilitate immediate treatment.
By Pathogen:
- Bacterial Sepsis: The most common form of sepsis, often triggered by Gram-positive or Gram-negative bacteria.
- Fungal Sepsis: A rising concern among immunocompromised patients, typically caused by Candida species.
- Others: Includes sepsis triggered by viral or parasitic infections.
By End User:
- Hospitals: The primary setting for sepsis diagnosis and management due to high patient inflow and critical care requirements.
- Pathology and Reference Laboratories: Specialized facilities that handle high volumes of diagnostic samples and provide outsourced testing services.
- Others: Includes clinics, ambulatory surgical centers, and research institutes.
By Geography:
- North America
- Europe
- Asia Pacific
- South & Central America
- Middle East & Africa
Sepsis Diagnostics Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2025 | US$ 844 Million |
| Market Size by 2034 | US$ 1558.98 Million |
| Global CAGR (2026 - 2034) | 7.06% |
| Historical Data | 2021-2024 |
| Forecast period | 2026-2034 |
| Segments Covered |
By Product
|
| Regions and Countries Covered |
North America
|
| Market leaders and key company profiles |
|
Sepsis Diagnostics Market Players Density: Understanding Its Impact on Business Dynamics
The Sepsis Diagnostics Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
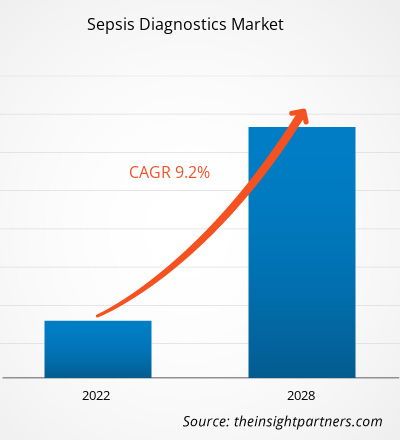
Sepsis Diagnostics Market Share Analysis by Geography
Asia-Pacific is expected to grow fastest in the coming years. Emerging markets in South & Central America, the Middle East, and Africa also have many untapped opportunities for diagnostic technology providers and medical device manufacturers to expand.
The sepsis diagnostics market is undergoing a significant transformation, moving from traditional culture-based methods to a global, high-speed molecular testing environment. Growth is driven by the rising prevalence of antimicrobial resistance (AMR), a surge in hospital-acquired infections (HAIs), and the expansion of the precision medicine sector. Below is a summary of market share and trends by region:
North America
- Market Share: Holds the largest share globally, driven by advanced healthcare infrastructure and high diagnostic spending.
- Key Drivers:
- Rising incidence of sepsis, with over 1.7 million adult cases reported annually in the U.S.
- Rapid clinical adoption of FDA-cleared multiplex PCR panels and automated workstations.
- Stringent CMS (Centers for Medicare & Medicaid Services) protocols that penalize hospitals for poor sepsis management.
- Trends: Scaling of AI-integrated diagnostic software and the successful adoption of rapid point-of-care (POC) tests in emergency departments to reduce patient boarding times.
Europe
- Market Share: Anchored by a strong presence of global leaders and standardized clinical guidelines in Germany, France, and the UK.
- Key Drivers:
- High domestic consumption of biomarker assays for early triage.
- Established public healthcare networks and centralized laboratory systems that facilitate high-throughput testing.
- Robust government funding for research into rapid pathogen identification and antibiotic stewardship.
- Trends: A strategic shift toward syndromic testing, where diagnostic panels are used to screen for multiple respiratory or bloodstream pathogens simultaneously to optimize bed management.
Asia-Pacific
- Market Share: The fastest-growing region, with China and India acting as primary growth for market expansion.
- Key Drivers:
- Massive consumer base and a high burden of infectious diseases in emerging economies.
- Increasing government-supported initiatives focused on improving critical care and laboratory facilities.
- Rapid urbanization and rising healthcare expenditure are leading to a preference for automated diagnostic systems.
- Trends: Heavy reliance on B2B contracts for high-volume microbiology instruments and the rising popularity of portable microfluidic devices for decentralized rural clinics.
South and Central America
- Market Share: Emerging market with a growing specialized laboratory sector in countries like Brazil and Chile.
- Key Drivers:
- Increasing awareness of the clinical superiority of molecular tests over traditional manual culturing.
- Modernization of private hospital networks to supply high-quality diagnostic services to urban centers.
- Rising interest in value-based care to mitigate the high cost of prolonged ICU stays.
- Trends: Growth of reference laboratory partnerships and the introduction of imported automated blood culture systems to differentiate from local manual labs.
Middle East and Africa
- Market Share: Developing market transitioning toward formalized, commercial-grade diagnostic production.
- Key Drivers:
- High demand for rapid diagnostic kits in regions with a high prevalence of neonatal sepsis.
- Strategic investments in smart health infrastructure to reduce reliance on outsourced diagnostic services.
- Implementation of modern cold-chain logistics to support the distribution of temperature-sensitive reagents.
- Trends: Deployment of mobile diagnostic units equipped with rapid POC molecular tools to reach remote populations and improve outbreak response times.
High Market Density and Competition
Competition is intensifying due to the presence of established leaders such as bioMérieux, Becton, Dickinson and Company (BD), and Danaher Corporation.
This competitive environment pushes vendors to differentiate through:
- Premiumization and functional branding: Positioning rapid molecular tests as superior clinical tools by emphasizing their ability to reduce mortality rates and hospital costs.
- Syndromic Panels: Expanding product portfolios to include panels that identify dozens of pathogens and antibiotic resistance genes in a single test.
- Supply Chain Control: Managing the entire diagnostic workflow from sample collection to digital data reporting to ensure quality and meet ethical medical standards.
- Technological Integration: Using new processing technologies like microfluidics and AI to create high-quality bedside diagnostics.
Opportunities and Strategic Moves
- Partner with high-end hospital networks and e-commerce-driven laboratory supply channels to tap into the surging demand for rapid diagnostics in Asia-Pacific and North American markets.
- Incorporate sustainable manufacturing practices to appeal to environmentally conscious health systems seeking ethical medical device alternatives.
Major Companies operating in the Sepsis Diagnostics Market are:
- Abbott
- F. Hoffmann-La Roche Ltd.
- Immunexpress Inc.
- BD
- Danaher
- Luminex Corporation
- Thermo Fisher Scientific Inc.
- bioMerieux SA.
- T2 Biosystems, Inc.
- Axis-Shield Diagnostics Ltd.
Disclaimer: The companies listed above are not ranked in any particular order.
Sepsis Diagnostics Market News and Recent Developments
- In January 2026, Immunexpress announced that its SeptiCyte RAPID test received regulatory clearance from the Australian Therapeutic Goods Administration (TGA) for use in paediatric patients suspected of sepsis. This inclusion in the Australian Register of Therapeutic Goods (ARTG) makes it the first and only host-response test available for children in the region. The test differentiates sepsis from noninfectious systemic inflammation in just one hour, addressing a critical need in Australia where pediatric sepsis is a major cause of mortality and remains significantly underestimated.
- In December 2024, T2 Biosystems, Inc. announced plans to license its proprietary technology to expand its leadership in direct-from-whole-blood detection of sepsis-causing pathogens. T2 Biosystems’ proprietary technology is a key component of its FDA-cleared products, including the first and only products able to detect sepsis-causing pathogens directly from whole blood.
Sepsis Diagnostics Market Report Coverage and Deliverables
The Sepsis Diagnostics Market Size and Forecast (2021–2034) report provides a detailed analysis of the market covering below areas:
- Sepsis Diagnostics Market size and forecast at global, regional, and country levels for all the key market segments covered under the scope
- Sepsis Diagnostics Market trends, as well as market dynamics such as drivers, restraints, and key opportunities
- Detailed PEST and SWOT analysis
- Sepsis Diagnostics Market analysis covering key market trends, global and regional framework, major players, regulations, and recent market developments
- Industry landscape and competition analysis covering market concentration, heat map analysis, prominent players, and recent developments in the Sepsis Diagnostics Market.
- Detailed company profiles
Frequently Asked Questions
Mrinal is a seasoned research analyst with over 8 years of experience in Life Sciences Market Intelligence and Consulting. With a strategic mindset and unwavering commitment to excellence, she has built deep expertise in pharmaceutical forecasting, market opportunity assessment, and developing industry benchmarks. Her work is anchored in delivering actionable insights that empower clients to make informed strategic decisions.
Mrinal’s core strength lies in translating complex quantitative datasets into meaningful business intelligence. Her analytical acumen is instrumental in shaping go-to-market (GTM) strategies and uncovering growth opportunities across the pharmaceutical and medical device sectors. As a trusted consultant, she consistently focuses on streamlining workflow processes and establishing best practices, thereby driving innovation and operational efficiency for her clients.
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Global, Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Testimonials
The Insight Partners' SCADA System Market report is comprehensive, with valuable insights on current trends and future forecasts. The team was highly professional, responsive, and supportive throughout. We are very satisfied and highly recommend their services.
RAN KEDEM Partner, Reali Technologies LTDsI requested a report on a very specific software market and the team produced the report in a few days. The information was very relevant and well presented. I then requested some changes and additions to the report. The team was again very responsive and I got the final report in less than a week.
JEAN-HERVE JENN Chairman, Future AnalyticaWe worked with The Insight Partners for an important market study and forecast. They gave us clear insights into opportunities and risks, which helped shape our plans. Their research was easy to use and based on solid data. It helped us make smart, confident decisions. We highly recommend them.
PIYUSH NAGPAL Sr. Vice President, High Beam GlobalThe Insight Partners delivered insightful, well-structured market research with strong domain expertise. Their team was professional and responsive throughout. The user-friendly website made accessing industry reports seamless. We highly recommend them for reliable, high-quality research services
YUKIHIKO ADACHI CEO, Deep Blue, LLC.This is the first time I have purchased a market report from The Insight Partners.While I was unsure at first, I visited their web site and felt more comfortable to take the risk and purchase a market report.I am completely satisfied with the quality of the report and customer service. I had several questions and comments with the initial report, but after a couple of dialogs over email with their analyst I believe I have a report that I can use as input to our strategic planning process.Thank you so much for taking the extra time and making this a positive experience.I will definitely recommend your service to others and you will be my first call when we need further market data.
JOHN SUZUKI President and Chief Executive Officer, Board Director, BK TechnologiesI wish to appreciate your support and the professionalism you displayed in the course of attending to my request for information regarding to infectious disease IVD market in Nigeria. I appreciate your patience, your guidance, and the fact that you were willing to offer a discount, which eventually made it possible for us to close a deal. I look forward to engaging The Insight Partners in the future, all thanks to the impression you have created in me as a result of this first encounter.
DR CHIJIOKE ONYIA MANAGING DIRECTOR, PineCrest Healthcare Ltd.Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends





 Get Free Sample For
Get Free Sample For