Key market dynamics include the rising global incidence of sepsis particularly hospital-acquired infections (HAIs), advancements in rapid molecular diagnostic tools, and increasing government initiatives to improve early detection protocols. Additionally, the market is expected to benefit from the growing geriatric population, the surge in antimicrobial resistance (AMR), and the rising demand for point-of-care (POC) testing to reduce critical turnaround times in emergency departments.
Sepsis Diagnostics Market AnalysisThe sepsis diagnostics market analysis shows a shift toward host-response-based testing as medical professionals prioritize understanding the patient’s immune status alongside pathogen identification. The market is splitting into high-volume, centralized laboratory microbiology and high-growth, decentralized molecular assays in intensive care units. Strategic opportunities are emerging in the development of syndromic panels that detect co-infections and antibiotic resistance genes in a single run, offering a clear competitive advantage over traditional sequential testing. The analysis also notes that market expansion depends on the seamless integration of diagnostic results into Electronic Health Records (EHR) and the ability of manufacturers to prove cost-savings by reducing the length of hospital stays. Competitive differentiation now stands out depending on the analytical speed and diagnostic accuracy that allows for the de-escalation of broad-spectrum antibiotics, supporting global antimicrobial stewardship goals.
Sepsis Diagnostics Market OverviewSepsis diagnostics have evolved from niche laboratory procedures to a central pillar of hospital critical care and emergency medicine. Sepsis diagnostics include automated blood culture systems, high-throughput molecular platforms, and bedside point-of-care devices. Both established medical technology giants and specialized biotech startups compete in this market, utilizing technologies such as PCR, NGS (Next-Generation Sequencing), and microfluidics. Growing demand for decentralized testing in North America and Europe has increased the popularity of POC solutions as a life-saving diagnostic tool. North America leads in revenue due to its advanced healthcare infrastructure and high adoption of innovative technologies, while Asia-Pacific is advancing rapidly due to improving medical facilities and a large patient pool. The US market is the most developed, driven by rigorous sepsis awareness programs and favorable reimbursement policies for advanced diagnostic tests.
Customize Research To Suit Your Requirement
We can optimize and tailor the analysis and scope which is unmet through our standard offerings. This flexibility will help you gain the exact information needed for your business planning and decision making.
Sepsis Diagnostics Market: Strategic Insights
| Market Size Value in | US$ 661.83 million in 2021 |
| Market Size Value by | US$ 1,226.52 million by 2028 |
| Growth rate | CAGR of 9.2% from 2021 to 2028. |
| Forecast Period | 2021- 2028 |
| Base Year | 2021 |

Mrinal
Have a question?
Mrinal will walk you through a 15-minute call to present the report’s content and answer all queries if you have any.
 Speak to Analyst
Speak to Analyst
Market Drivers:
- High Incidence of Hospital-Acquired Infections (HAIs): Sepsis is a leading cause of death in hospitals worldwide. The rising frequency of infections following invasive surgeries and ICU admissions is a primary driver for robust diagnostic adoption.
- Technological Shift toward Rapid Molecular Diagnostics: Traditional culture methods can take 24–72 hours. New molecular tools that provide results in under 6 hours are revolutionizing the market by enabling timely clinical decisions.
- Growing Geriatric and Immunocompromised Populations: An aging global demographic and an increase in chronic diseases have created a larger at-risk population vulnerable to septic shock.
Market Opportunities:
- Development of Novel Biomarkers and Multiplex Panels: There is a significant opportunity for diagnostic tests that can differentiate between bacterial, viral, and fungal sepsis at an early stage.
- Expansion in Emerging APAC and LAMEA Markets: Increasing healthcare expenditure and the formalization of clinical protocols in China, India, and Brazil offer untapped growth potential for global diagnostic providers.
- AI-Driven Predictive Analytics: Integrating diagnostic hardware with software that provides real-time risk scores for patients enables a proactive rather than reactive approach to sepsis management.
The Sepsis Diagnostics Market share is analyzed across various segments to provide a clearer understanding of its structure, growth potential, and emerging trends. Below is the standard segmentation approach used in most industry reports:
By Product:
- Instruments: Includes automated blood culture systems, PCR machines, and POC devices that serve as the hardware foundation for sepsis detection.
- Reagents and Assays: The recurring revenue driver, consisting of the chemical components used to perform molecular, immunoassay, and biochemical tests.
- Blood Culture Media: Specialized nutrient solutions required to grow and identify pathogens from patient blood samples; remains a core component of microbiology workflows.
- Software: The fastest-growing segment, focusing on AI-based clinical decision support and lab data management platforms.
By Technology:
- Molecular Diagnostics: High-growth segment utilizing PCR and DNA microarrays for rapid pathogen identification and antibiotic resistance detection.
- Flow Cytometry: Used to analyze the physical and chemical characteristics of cells, often helping in immune response monitoring.
- Microfluidics: An emerging technology enabling the miniaturization of diagnostic processes for point-of-care applications.
- Immunoassay: Focuses on measuring host-response markers like Procalcitonin (PCT) and Interleukins.
- Biomarker: Involves the identification of specific biological indicators that signal the onset or severity of sepsis.
- Microbiology: The traditional gold standard technology, involving the cultivation of microorganisms from blood samples.
By Method:
- Automated Diagnostics: Modern systems that reduce human error and turnaround time through robotic sample processing and digital reading.
- Conventional Diagnostics: Manual laboratory techniques that rely on the expertise of technicians and traditional culturing timelines.
By Test Type:
- Laboratory Tests: Comprehensive, high-accuracy tests performed in a centralized hospital or reference labs.
- Point-of-Care Tests: Rapid tests performed at the patient's bedside, primarily in emergency rooms or ICUs, to facilitate immediate treatment.
By Pathogen:
- Bacterial Sepsis: The most common form of sepsis, often triggered by Gram-positive or Gram-negative bacteria.
- Fungal Sepsis: A rising concern among immunocompromised patients, typically caused by Candida species.
- Others: Includes sepsis triggered by viral or parasitic infections.
By End User:
- Hospitals: The primary setting for sepsis diagnosis and management due to high patient inflow and critical care requirements.
- Pathology and Reference Laboratories: Specialized facilities that handle high volumes of diagnostic samples and provide outsourced testing services.
- Others: Includes clinics, ambulatory surgical centers, and research institutes.
By Geography:
- North America
- Europe
- Asia Pacific
- South & Central America
- Middle East & Africa
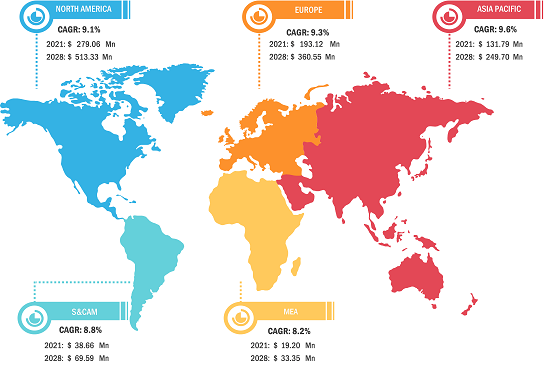
Asia-Pacific is expected to grow fastest in the coming years. Emerging markets in South & Central America, the Middle East, and Africa also have many untapped opportunities for diagnostic technology providers and medical device manufacturers to expand.
The sepsis diagnostics market is undergoing a significant transformation, moving from traditional culture-based methods to a global, high-speed molecular testing environment. Growth is driven by the rising prevalence of antimicrobial resistance (AMR), a surge in hospital-acquired infections (HAIs), and the expansion of the precision medicine sector. Below is a summary of market share and trends by region:
North America
- Market Share: Holds the largest share globally, driven by advanced healthcare infrastructure and high diagnostic spending.
- Key Drivers:
- Rising incidence of sepsis, with over 1.7 million adult cases reported annually in the U.S.
- Rapid clinical adoption of FDA-cleared multiplex PCR panels and automated workstations.
- Stringent CMS (Centers for Medicare & Medicaid Services) protocols that penalize hospitals for poor sepsis management.
- Trends: Scaling of AI-integrated diagnostic software and the successful adoption of rapid point-of-care (POC) tests in emergency departments to reduce patient boarding times.
Europe
- Market Share: Anchored by a strong presence of global leaders and standardized clinical guidelines in Germany, France, and the UK.
- Key Drivers:
- High domestic consumption of biomarker assays for early triage.
- Established public healthcare networks and centralized laboratory systems that facilitate high-throughput testing.
- Robust government funding for research into rapid pathogen identification and antibiotic stewardship.
- Trends: A strategic shift toward syndromic testing, where diagnostic panels are used to screen for multiple respiratory or bloodstream pathogens simultaneously to optimize bed management.
Asia-Pacific
- Market Share: The fastest-growing region, with China and India acting as primary growth for market expansion.
- Key Drivers:
- Massive consumer base and a high burden of infectious diseases in emerging economies.
- Increasing government-supported initiatives focused on improving critical care and laboratory facilities.
- Rapid urbanization and rising healthcare expenditure are leading to a preference for automated diagnostic systems.
- Trends: Heavy reliance on B2B contracts for high-volume microbiology instruments and the rising popularity of portable microfluidic devices for decentralized rural clinics.
South and Central America
- Market Share: Emerging market with a growing specialized laboratory sector in countries like Brazil and Chile.
- Key Drivers:
- Increasing awareness of the clinical superiority of molecular tests over traditional manual culturing.
- Modernization of private hospital networks to supply high-quality diagnostic services to urban centers.
- Rising interest in value-based care to mitigate the high cost of prolonged ICU stays.
- Trends: Growth of reference laboratory partnerships and the introduction of imported automated blood culture systems to differentiate from local manual labs.
Middle East and Africa
- Market Share: Developing market transitioning toward formalized, commercial-grade diagnostic production.
- Key Drivers:
- High demand for rapid diagnostic kits in regions with a high prevalence of neonatal sepsis.
- Strategic investments in smart health infrastructure to reduce reliance on outsourced diagnostic services.
- Implementation of modern cold-chain logistics to support the distribution of temperature-sensitive reagents.
- Trends: Deployment of mobile diagnostic units equipped with rapid POC molecular tools to reach remote populations and improve outbreak response times.
High Market Density and Competition
Competition is intensifying due to the presence of established leaders such as bioMérieux, Becton, Dickinson and Company (BD), and Danaher Corporation.
This competitive environment pushes vendors to differentiate through:
- Premiumization and functional branding: Positioning rapid molecular tests as superior clinical tools by emphasizing their ability to reduce mortality rates and hospital costs.
- Syndromic Panels: Expanding product portfolios to include panels that identify dozens of pathogens and antibiotic resistance genes in a single test.
- Supply Chain Control: Managing the entire diagnostic workflow from sample collection to digital data reporting to ensure quality and meet ethical medical standards.
- Technological Integration: Using new processing technologies like microfluidics and AI to create high-quality bedside diagnostics.
Opportunities and Strategic Moves
- Partner with high-end hospital networks and e-commerce-driven laboratory supply channels to tap into the surging demand for rapid diagnostics in Asia-Pacific and North American markets.
- Incorporate sustainable manufacturing practices to appeal to environmentally conscious health systems seeking ethical medical device alternatives.
- Abbott
- F. Hoffmann-La Roche Ltd.
- Immunexpress Inc.
- BD
- Danaher
- Luminex Corporation
- Thermo Fisher Scientific Inc.
- bioMerieux SA.
- T2 Biosystems, Inc.
- Axis-Shield Diagnostics Ltd.
Disclaimer: The companies listed above are not ranked in any particular order.
Sepsis Diagnostics Market News and Recent Developments- In January 2026, Immunexpress announced that its SeptiCyte RAPID test received regulatory clearance from the Australian Therapeutic Goods Administration (TGA) for use in paediatric patients suspected of sepsis. This inclusion in the Australian Register of Therapeutic Goods (ARTG) makes it the first and only host-response test available for children in the region. The test differentiates sepsis from noninfectious systemic inflammation in just one hour, addressing a critical need in Australia where pediatric sepsis is a major cause of mortality and remains significantly underestimated.
- In December 2024, T2 Biosystems, Inc. announced plans to license its proprietary technology to expand its leadership in direct-from-whole-blood detection of sepsis-causing pathogens. T2 Biosystems’ proprietary technology is a key component of its FDA-cleared products, including the first and only products able to detect sepsis-causing pathogens directly from whole blood.
The Sepsis Diagnostics Market Size and Forecast (2021–2034) report provides a detailed analysis of the market covering below areas:
- Sepsis Diagnostics Market size and forecast at global, regional, and country levels for all the key market segments covered under the scope
- Sepsis Diagnostics Market trends, as well as market dynamics such as drivers, restraints, and key opportunities
- Detailed PEST and SWOT analysis
- Sepsis Diagnostics Market analysis covering key market trends, global and regional framework, major players, regulations, and recent market developments
- Industry landscape and competition analysis covering market concentration, heat map analysis, prominent players, and recent developments in the Sepsis Diagnostics Market.
- Detailed company profiles

Report Coverage
Revenue forecast, Company Analysis, Industry landscape, Growth factors, and Trends
Segment Covered
Product, Technology, Method, Test Type, Pathogen, and End User

Regional Scope
North America, Europe, Asia Pacific, Middle East & Africa, South & Central America
Country Scope
Argentina, Australia, Brazil, Canada, China, France, Germany, India, Italy, Japan, Mexico, Saudi Arabia, South Africa, South Korea, Spain, United Arab Emirates, United Kingdom, United States
Frequently Asked Questions
Major players operating in the sepsis diagnostics market are Abbott, F. Hoffman-La Roche Ltd, Immunexpress Inc., BD, Danaher (Beckman Coulter), Luminex Corporation, Thermo Fischer Scientific Inc., bioMerieux SA, T2 Biosystems, Inc., and Axis-Shield Diagnostics Ltd, among others.
Main challenges include the high cost of automated diagnostic devices and the specialized training required for advanced molecular testing.
Asia-Pacific is the fastest-growing region, with a strong focus on improved healthcare infrastructure and rising awareness of infectious disease management.
The market is expected to reach approximately US$ 1558.98 million, growing from a valuation of US$ 844 Million in 2025.
Trends include the shift toward syndromic panel testing in Europe, the rise of mobile diagnostics in Africa, and the massive scaling of automated laboratory infrastructure in Asia-Pacific.
Yes! We provide a free sample of the report, which includes Report Scope (Table of Contents), report structure, and selected insights to help you assess the value of the full report. Please click on the "Download Sample" button or contact us to receive your copy.
Absolutely - analyst assistance is part of the package. You can connect with our analyst post-purchase to clarify report insights, methodology or discuss how the findings apply to your business needs.
Once your order is successfully placed, you will receive a confirmation email along with your invoice.
• For published reports: You'll receive access to the report within 4-6 working hours via a secured email sent to your email.
• For upcoming reports: Your order will be recorded as a pre-booking. Our team will share the estimated release date and keep you informed of any updates. As soon as the report is published, it will be delivered to your registered email.
We offer customization options to align the report with your specific objectives. Whether you need deeper insights into a particular region, industry segment, competitor analysis, or data cut, our research team can tailor the report accordingly. Please share your requirements with us, and we'll be happy to provide a customized proposal or scope.
The report is available in either PDF format or as an Excel dataset, depending on the license you choose.
The PDF version provides the full analysis and visuals in a ready-to-read format. The Excel dataset includes all underlying data tables for easy manipulation and further analysis.
Please review the license options at checkout or contact us to confirm which formats are included with your purchase.
Our payment process is fully secure and PCI-DSS compliant.
We use trusted and encrypted payment gateways to ensure that all transactions are protected with industry-standard SSL encryption. Your payment details are never stored on our servers and are handled securely by certified third-party processors.
You can make your purchase with confidence, knowing your personal and financial information is safe with us.
Yes, we do offer special pricing for bulk purchases.
If you're interested in purchasing multiple reports, we're happy to provide a customized bundle offer or volume-based discount tailored to your needs. Please contact our sales team with the list of reports you're considering, and we'll share a personalized quote.
Yes, absolutely.
Our team is available to help you make an informed decision. Whether you have questions about the report's scope, methodology, customization options, or which license suits you best, we're here to assist. Please reach out to us at sales@theinsightpartners.com, and one of our representatives will get in touch promptly.
Yes, a billing invoice will be automatically generated and sent to your registered email upon successful completion of your purchase.
If you need the invoice in a specific format or require additional details (such as company name, GST, or VAT information), feel free to contact us, and we'll be happy to assist.
Yes, certainly.
If you encounter any difficulties accessing or receiving your report, our support team is ready to assist you. Simply reach out to us via email or live chat with your order information, and we'll ensure the issue is resolved quickly so you can access your report without interruption.
The Insight Partners performs research in 4 major stages: Data Collection & Secondary Research, Primary Research, Data Analysis and Data Triangulation & Final Review.
- Data Collection and Secondary Research:
As a market research and consulting firm operating from a decade, we have published many reports and advised several clients across the globe. First step for any study will start with an assessment of currently available data and insights from existing reports. Further, historical and current market information is collected from Investor Presentations, Annual Reports, SEC Filings, etc., and other information related to company’s performance and market positioning are gathered from Paid Databases (Factiva, Hoovers, and Reuters) and various other publications available in public domain.
Several associations trade associates, technical forums, institutes, societies and organizations are accessed to gain technical as well as market related insights through their publications such as research papers, blogs and press releases related to the studies are referred to get cues about the market. Further, white papers, journals, magazines, and other news articles published in the last 3 years are scrutinized and analyzed to understand the current market trends.
- Primary Research:
The primarily interview analysis comprise of data obtained from industry participants interview and answers to survey questions gathered by in-house primary team.
For primary research, interviews are conducted with industry experts/CEOs/Marketing Managers/Sales Managers/VPs/Subject Matter Experts from both demand and supply side to get a 360-degree view of the market. The primary team conducts several interviews based on the complexity of the markets to understand the various market trends and dynamics which makes research more credible and precise.
A typical research interview fulfils the following functions:
- Provides first-hand information on the market size, market trends, growth trends, competitive landscape, and outlook
- Validates and strengthens in-house secondary research findings
- Develops the analysis team’s expertise and market understanding
Primary research involves email interactions and telephone interviews for each market, category, segment, and sub-segment across geographies. The participants who typically take part in such a process include, but are not limited to:
- Industry participants: VPs, business development managers, market intelligence managers and national sales managers
- Outside experts: Valuation experts, research analysts and key opinion leaders specializing in the electronics and semiconductor industry.
Below is the breakup of our primary respondents by company, designation, and region:
Once we receive the confirmation from primary research sources or primary respondents, we finalize the base year market estimation and forecast the data as per the macroeconomic and microeconomic factors assessed during data collection.
- Data Analysis:
Once data is validated through both secondary as well as primary respondents, we finalize the market estimations by hypothesis formulation and factor analysis at regional and country level.
- 3.1 Macro-Economic Factor Analysis:
We analyse macroeconomic indicators such the gross domestic product (GDP), increase in the demand for goods and services across industries, technological advancement, regional economic growth, governmental policies, the influence of COVID-19, PEST analysis, and other aspects. This analysis aids in setting benchmarks for various nations/regions and approximating market splits. Additionally, the general trend of the aforementioned components aid in determining the market's development possibilities.
- 3.2 Country Level Data:
Various factors that are especially aligned to the country are taken into account to determine the market size for a certain area and country, including the presence of vendors, such as headquarters and offices, the country's GDP, demand patterns, and industry growth. To comprehend the market dynamics for the nation, a number of growth variables, inhibitors, application areas, and current market trends are researched. The aforementioned elements aid in determining the country's overall market's growth potential.
- 3.3 Company Profile:
The “Table of Contents” is formulated by listing and analyzing more than 25 - 30 companies operating in the market ecosystem across geographies. However, we profile only 10 companies as a standard practice in our syndicate reports. These 10 companies comprise leading, emerging, and regional players. Nonetheless, our analysis is not restricted to the 10 listed companies, we also analyze other companies present in the market to develop a holistic view and understand the prevailing trends. The “Company Profiles” section in the report covers key facts, business description, products & services, financial information, SWOT analysis, and key developments. The financial information presented is extracted from the annual reports and official documents of the publicly listed companies. Upon collecting the information for the sections of respective companies, we verify them via various primary sources and then compile the data in respective company profiles. The company level information helps us in deriving the base number as well as in forecasting the market size.
- 3.4 Developing Base Number:
Aggregation of sales statistics (2020-2022) and macro-economic factor, and other secondary and primary research insights are utilized to arrive at base number and related market shares for 2022. The data gaps are identified in this step and relevant market data is analyzed, collected from paid primary interviews or databases. On finalizing the base year market size, forecasts are developed on the basis of macro-economic, industry and market growth factors and company level analysis.
- Data Triangulation and Final Review:
The market findings and base year market size calculations are validated from supply as well as demand side. Demand side validations are based on macro-economic factor analysis and benchmarks for respective regions and countries. In case of supply side validations, revenues of major companies are estimated (in case not available) based on industry benchmark, approximate number of employees, product portfolio, and primary interviews revenues are gathered. Further revenue from target product/service segment is assessed to avoid overshooting of market statistics. In case of heavy deviations between supply and demand side values, all thes steps are repeated to achieve synchronization.
We follow an iterative model, wherein we share our research findings with Subject Matter Experts (SME’s) and Key Opinion Leaders (KOLs) until consensus view of the market is not formulated – this model negates any drastic deviation in the opinions of experts. Only validated and universally acceptable research findings are quoted in our reports.
We have important check points that we use to validate our research findings – which we call – data triangulation, where we validate the information, we generate from secondary sources with primary interviews and then we re-validate with our internal data bases and Subject matter experts. This comprehensive model enables us to deliver high quality, reliable data in shortest possible time.

May 2026
Anesthesia Machines Market
Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Product (Advanced Anesthesia Machines, Basic Anesthesia Machines, and Consumables), Customer (Small Hospitals & Nursing Home, Standalone Hospitals, Tertiary Care Hospitals, and Hospital Chain), End User (Anaesthesiologists, Critical Care Specialists, and Others), and Geography (North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America)

May 2026
Hospital at Home Market
Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Component (Equipment & Devices and Software & Services), Service Type (Acute Care at Home, Chronic Disease Management at Home, Post Acute or Transitional Care, Palliative Care and Hospice Care, and Rehabilitation Services), Delivery Mode (Remote Monitoring and Telehealth, In-person Home Visits, and Hybrid Models), Patient Demographic ( Adult Patients and Pediatric Patients), Indication (Cardiovascular Conditions, Respiratory Diseases, Orthopedic and Post-Surgical Recovery, Infectious Diseases, Neurological Rehabilitation, and Others), Care Provider (Hospitals and Health Systems, Home Health Agencies, Primary Care or Physician Groups, and Private HaH Providers), and Geography (North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America)

May 2026
MRI-guided Focused Ultrasound Therapy Market
Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Application (Breast Cancer, Prostate Cancer, Liver Cancer, Pancreatic Cancer, Breast Lifting and Aesthetic Application, Nipple and Areola Preservation, Post Surgical Applications, and Others), End User (Healthcare Facilities, Diagnostic Imaging Centers, and Research Centers), and Geography (North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America)

May 2026
Drug Delivery Devices Contract Manufacturing Market
Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Device Type (Infusion Devices and Administration Set, Syringes, Inhalers, Autoinjectors, and Pen Injectors), Service Type (Full-device Manufacturing, Sub-assembly and Components, and Materials-specific Services), and Geography (North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America)

May 2026
IVD Devices Contract Manufacturing Market
Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Service Type (Full-device Manufacturing, Sub-assembly and Components, and Materials-specific Services), Device Type (IVD consumables, IVD equipment), and Geography (North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America)

May 2026
Gene Therapy CDMO Market
Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Service Type (Drug Development and Manufacturing, Testing and Regulatory Services, and Other Service Types), End User (Pharmaceutical Companies, Biopharmaceutical Companies, and Other End Users), and Geography (North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America)

May 2026
RT-PCR Market
Size and Forecast (2021 - 2031), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Product (Reagents & Consumables, Instruments, and Software & Services), Application (Research Application, Clinical Application, and Forensic Application), End user (Hospitals and Diagnostic Centers, Pharmaceutical and Biotechnology Companies, Research Laboratories and Academic Institutes, Forensic Laboratories, and Clinical Research Organizations)

May 2026
dPCR Market
Size and Forecast (2021 - 2031), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Product (Reagents & Consumables, Instruments, and Software & Services), Application (Research Application, Clinical Application, and Forensic Application), End user (Hospitals and Diagnostic Centers, Pharmaceutical and Biotechnology Companies, Research Laboratories and Academic Institutes, Forensic Laboratories, and Clinical Research Organizations)


 Get Free Sample For
Get Free Sample For