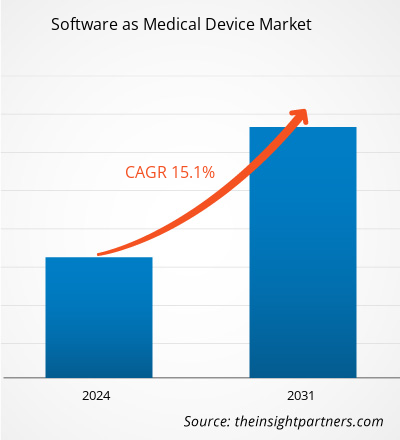
The software as medical device market size is projected to reach US$ 93.08 billion by 2031 from US$ 30.17 billion in 2023. The market is expected to register a CAGR of 15.1% during 2023–2031. The increasing number of wearable medical devices is likely to remain a key trend in the market.
Software as Medical Device Market Analysis
In recent years, software as a Medical Device (SaMD) has become a vital tool in the healthcare arsenal, and its growth is likely to accelerate in 2023. Factors like SaMD and other digital solutions are reshaping patient-provider interactions and revealing the power of data sharing, collection, and tracking. Other factors like the increasing prevalence of Virtual First Care, or V1C to the forefront, allowing healthcare providers to deliver care without risking physical contact. Thus, making remote diseases manageable.
Software as Medical Device Market Overview
Mexico is focused on advancing healthcare digitalization by encouraging more incorporation of software-based digital solutions in its infrastructure. The country is growing at an unprecedented rate. Within the past few decades, the Mexican government has put forth an ambitious initiative to improve the overall quality of healthcare by implementing digital tools in its healthcare settings. The development in overall digital healthcare in the country will also likely promote the market's growth and offer a conducive scenario over the years to come. Furthermore, Mexico’s geriatric population of 60 years and above would increase from 6.3% in 2010 to 23% by 2050. Thus, such factors as the rising geriatric population are expected to increase demand in the coming decade.
Customize This Report To Suit Your Requirement
You will get customization on any report - free of charge - including parts of this report, or country-level analysis, Excel Data pack, as well as avail great offers and discounts for start-ups & universities
Software as Medical Device Market: Strategic Insights

- Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
You will get customization on any report - free of charge - including parts of this report, or country-level analysis, Excel Data pack, as well as avail great offers and discounts for start-ups & universities
Software as Medical Device Market: Strategic Insights

- Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
Software as Medical Device Market Drivers and Opportunities
FDA Issues New Draft Guidance on SaMD to Favor Market
A recently released draft guidance update is clarifying and streamlining the process. According to the new guidance document, the Software as a Medical Device was classified into Clinical Decision Support Software (CDSS) and Patient Decision Support Software (PDS) for both the patients and caregivers. These classification rules consider several factors, including the degree of invasiveness in the body, duration of contact, body system affected, and local versus systemic effects. The draft guidance update is also an indication of increased SaMD development.
Developing regions with Emerging Economies
With the rise in the economy and per capita income of the people in emerging economies, the growth for enhanced medical services is also increasing. The growth of the medical industry is experienced due to the increasing automatization of healthcare services. The global market players consider the region for their business expansions in the emerging nations, which is further expected to grow the Software as Medical Device (SaMD) market during the forecast period. According to the Kingdom’s Communications and Information Technology Commission (CITC), it is estimated that most (54%) Saudi consumers use technology to manage their health, 40% use health apps on mobile devices, and the rest 14% use wearable technology.
Software as Medical Device Market Report Segmentation Analysis
Key segments that contributed to the derivation of the software as medical device market analysis are device type, application, and deployment type.
- Based on device type, the software as medical device market is divided into PCs/laptop, smartphones/tablets, and wearable devices. The PCs/laptop segment held a larger market share in 2023.
- By application, the market is segmented into screening and diagnosis, monitoring and alerting, and disease management. The screening and diagnostics segment held the largest share of the market in 2023.
- In terms of deployment type, the market is bifurcated into cloud and on-premises. The cloud segment held the largest share of the market in 2023.
Software as Medical Device Market Share Analysis by Geography
The geographic scope of the Software as Medical Device market report is mainly divided into five regions: North America, Asia Pacific, Europe, Middle East & Africa, and South & Central America.
North America has dominated the market. The penetration of artificial intelligent, machine learning, Natural Language Processing (NLP) and its application in digital healthcare is likely to be accountable for the dominance of the market. Furthermore, presence of key market leaders contribution novel solutions for diagnostic and remote supervising of patients through smartphone and laptop based software solutions are also creating a rise in demand for these solutions as they are easy to operate, understand and analyse health conditions thereby creating a rise their demand. Asia Pacific is anticipated to grow with the highest CAGR in the coming years.
Software as Medical Device Market Regional Insights
The regional trends and factors influencing the Software as Medical Device Market throughout the forecast period have been thoroughly explained by the analysts at Insight Partners. This section also discusses Software as Medical Device Market segments and geography across North America, Europe, Asia Pacific, Middle East and Africa, and South and Central America.
- Get the Regional Specific Data for Software as Medical Device Market
Software as Medical Device Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2023 | US$ 30.17 Billion |
| Market Size by 2031 | US$ 93.08 Billion |
| Global CAGR (2023 - 2031) | 15.1% |
| Historical Data | 2021-2022 |
| Forecast period | 2024-2031 |
| Segments Covered |
By Device Type
|
| Regions and Countries Covered | North America
|
| Market leaders and key company profiles |
Software as Medical Device Market Players Density: Understanding Its Impact on Business Dynamics
The Software as Medical Device Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
Market players density refers to the distribution of firms or companies operating within a particular market or industry. It indicates how many competitors (market players) are present in a given market space relative to its size or total market value.
Major Companies operating in the Software as Medical Device Market are:
- Velentium LLC
- Tietronix Software, Inc.
- S3 Connected Health
- Zühlke Group
- Science Group
- Inzentiz
Disclaimer: The companies listed above are not ranked in any particular order.
- Get the Software as Medical Device Market top key players overview
Software as Medical Device Market News and Recent Developments
The software as medical device market is evaluated by gathering qualitative and quantitative data post primary and secondary research, which includes important corporate publications, association data, and databases. A few of the developments in the software as medical device market are listed below:
- CompliancePath and Ideagen have entered into a Product Validation partnership to extend their on-going validation accelerator program to include all life science-related software products. (Source: CompliancePath, Press Release, April 2020)
- US-based STAT Health company introduced a 24/7 in-ear wearable general wellness product that measures blood flow to. (Source: STAT Health, Press Release, June 2023)
Software as Medical Device Market Report Coverage and Deliverables
The “Software as Medical Device Market Size and Forecast (2021–2031)” report provides a detailed analysis of the market covering below areas:
- Software as Medical Device market size and forecast at global, regional, and country levels for all the key market segments covered under the scope
- Software as Medical Device market trends as well as market dynamics such as drivers, restraints, and key opportunities
- Detailed PEST/Porter’s Five Forces and SWOT analysis
- Software as Medical Device market analysis covering key market trends, global and regional framework, major players, regulations, and recent market developments
- Industry landscape and competition analysis covering market concentration, heat map analysis, prominent players, and recent developments for the Software as Medical Device market
- Detailed company profiles
Frequently Asked Questions
Which region dominated the Software as Medical Device market in 2023?
North America held the largest market for Software as Medical Device. The growth of the region is attributed to continuous rise in the use of Software as Medical Device, innovations in software designs, and increasing awareness among people regarding the benefits for digital healthcare applications.
What are the driving factors impacting the Software as Medical Device market?
The driving factors are increasing adoption of Internet of Things (IoT) & connected devices in healthcare and the advantages offered by Software as Medical Device (SaMD).
What are the future trends of the Software as Medical Device market?
Increasing number of wearable medical devices is likely to remain key trends in the market.
Which are the leading players operating in the Software as Medical Device market?
S3 Connected Health, BrightInsight, Inc. and CompliancePath are among the top players operating in the Software as Medical Device market.
What would be the estimated value of the Software as Medical Device market by 2031?
The software as medical device market size is projected to reach US$ 93.08 billion by 2031.
What is the expected CAGR of the Software as Medical Device market?
The market is expected to register a CAGR of 15.1% during 2023–2031.
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Global, Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
- Parking Meter Apps Market
- eSIM Market
- Advanced Distributed Management System Market
- Online Exam Proctoring Market
- Electronic Data Interchange Market
- Barcode Software Market
- Maritime Analytics Market
- Cloud Manufacturing Execution System (MES) Market
- Robotic Process Automation Market
- Digital Signature Market
Testimonials
I wish to appreciate your support and the professionalism you displayed in the course of attending to my request for information regarding to infectious disease IVD market in Nigeria. I appreciate your patience, your guidance, and the fact that you were willing to offer a discount, which eventually made it possible for us to close a deal. I look forward to engaging The Insight Partners in the future, all thanks to the impression you have created in me as a result of this first encounter.
DR CHIJIOKE ONYIA, MANAGING DIRECTOR, PineCrest Healthcare Ltd.The Insight Partners delivered insightful, well-structured market research with strong domain expertise. Their team was professional and responsive throughout. The user-friendly website made accessing industry reports seamless. We highly recommend them for reliable, high-quality research services
Yukihiko Adachi CEO, Deep Blue, LLC.Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Strategic Planning
- Investment Justification
- Identifying Emerging Markets
- Enhancing Marketing Strategies
- Boosting Operational Efficiency
- Tracking Industry Innovations
- Aligning with Regulatory Trends
Yes! We provide a free sample of the report, which includes Report Scope (Table of Contents), report structure, and selected insights to help you assess the value of the full report. Please click on the "Download Sample" button or contact us to receive your copy.
Absolutely — analyst assistance is part of the package. You can connect with our analyst post-purchase to clarify report insights, methodology or discuss how the findings apply to your business needs.
Once your order is successfully placed, you will receive a confirmation email along with your invoice.
• For published reports: You’ll receive access to the report within 4–6 working hours via a secured email sent to your email.
• For upcoming reports: Your order will be recorded as a pre-booking. Our team will share the estimated release date and keep you informed of any updates. As soon as the report is published, it will be delivered to your registered email.
We offer customization options to align the report with your specific objectives. Whether you need deeper insights into a particular region, industry segment, competitor analysis, or data cut, our research team can tailor the report accordingly. Please share your requirements with us, and we’ll be happy to provide a customized proposal or scope.
The report is available in either PDF format or as an Excel dataset, depending on the license you choose.
The PDF version provides the full analysis and visuals in a ready-to-read format. The Excel dataset includes all underlying data tables for easy manipulation and further analysis.
Please review the license options at checkout or contact us to confirm which formats are included with your purchase.
Our payment process is fully secure and PCI-DSS compliant.
We use trusted and encrypted payment gateways to ensure that all transactions are protected with industry-standard SSL encryption. Your payment details are never stored on our servers and are handled securely by certified third-party processors.
You can make your purchase with confidence, knowing your personal and financial information is safe with us.
Yes, we do offer special pricing for bulk purchases.
If you're interested in purchasing multiple reports, we’re happy to provide a customized bundle offer or volume-based discount tailored to your needs. Please contact our sales team with the list of reports you’re considering, and we’ll share a personalized quote.
Yes, absolutely.
Our team is available to help you make an informed decision. Whether you have questions about the report’s scope, methodology, customization options, or which license suits you best, we’re here to assist. Please reach out to us at sales@theinsightpartners.com, and one of our representatives will get in touch promptly.
Yes, a billing invoice will be automatically generated and sent to your registered email upon successful completion of your purchase.
If you need the invoice in a specific format or require additional details (such as company name, GST, or VAT information), feel free to contact us, and we’ll be happy to assist.
Yes, certainly.
If you encounter any difficulties accessing or receiving your report, our support team is ready to assist you. Simply reach out to us via email or live chat with your order information, and we’ll ensure the issue is resolved quickly so you can access your report without interruption.





















 Get Free Sample For
Get Free Sample For