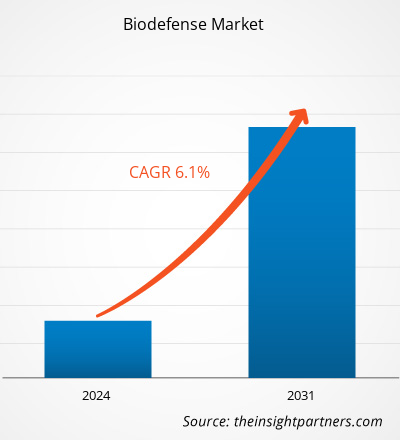
La taille du marché de la biodéfense en 2021 s'élevait à 12,37 milliards de dollars américains et devrait atteindre 21,62 milliards de dollars américains d'ici 2031. Le marché devrait enregistrer un TCAC de 6,1 % en 2023-2031. Les techniques de big data et d'intelligence artificielle offrent d'énormes possibilités pour réaliser des évaluations techniques du risque et de la possibilité de différentes menaces biologiques et une opportunité sans précédent pour cartographier les épidémies. Ainsi, l'analyse prédictive peut aider à remodeler l' industrie de la biodéfense , servant de tendance clé du marché de la biodéfense .
Analyse du marché de la biodéfense
Les virus mortels et naturels peuvent désormais être facilement modifiés pour causer encore plus de dégâts grâce aux récentes avancées scientifiques dans le domaine du génie génétique et de la biotechnologie . La reconnaissance croissante des effets potentiellement désastreux des attaques et des épidémies biologiques a accru la demande de solutions de biodéfense . Partout dans le monde, les gouvernements investissent massivement dans les capacités de biodéfense , ce qui soutient l'expansion du marché. Les développements technologiques et l'importance croissante accordée à la recherche et au développement ont également entraîné le lancement de solutions de biodéfense sophistiquées et innovantes . L'évolution des solutions de biodéfense est propulsée par des innovations technologiques telles que la biologie synthétique, la génomique et l'analyse de données, qui offrent des capacités améliorées d'identification, de caractérisation et d'atténuation des menaces biologiques. En outre, les partenariats entre les organisations gouvernementales, de défense et commerciales ont soutenu le développement de stratégies de biodéfense complètes et stimulé l'expansion du marché.
Aperçu du marché de la biodéfense
La biodéfense désigne l’ensemble des activités et des technologies qui visent à rétablir la biosécurité d’un pays qui est ou pourrait être exposé à des menaces biologiques ou à des maladies infectieuses. Elle permet de contrer les risques potentiels associés aux pandémies, au bioterrorisme et à d’autres urgences biologiques. Le gouvernement fédéral américain coordonne les programmes et établit des budgets pour prévenir, réagir et prendre des mesures contre les catastrophes biologiques. Le programme américain de défense biologique, également connu sous le nom de National Biodefense Strategy, implique des efforts collectifs de nombreux niveaux de gouvernement en association avec des organismes privés et d’autres parties prenantes pour la conduite d’ activités de biodéfense . En outre, le Centre de collaboration nationale des maladies infectieuses est une organisation canadienne qui vise à fournir des informations récentes sur les maladies infectieuses émergentes aux décideurs politiques, aux praticiens de la santé publique, aux inspecteurs de la santé et à d’autres par le biais de webinaires, de podcasts, de ressources en ligne et de publications. Ainsi, les efforts et les initiatives entrepris par le gouvernement pour renforcer le système de biodéfense sont susceptibles de soutenir la croissance du marché au cours de la période de prévision.
Personnalisez ce rapport en fonction de vos besoins
Vous bénéficierez d'une personnalisation gratuite de n'importe quel rapport, y compris de certaines parties de ce rapport, d'une analyse au niveau des pays, d'un pack de données Excel, ainsi que de superbes offres et réductions pour les start-ups et les universités.
Marché de la biodéfense : perspectives stratégiques

-
Obtenez les principales tendances clés du marché de ce rapport.Cet échantillon GRATUIT comprendra une analyse de données, allant des tendances du marché aux estimations et prévisions.
Facteurs moteurs et opportunités du marché de la biodéfense
Présence d' initiatives gouvernementales favorables pour stimuler la croissance du marché
Les gouvernements des pays développés prennent diverses mesures et adoptent des mesures préventives pour résister aux attaques bioterroristes . Le gouvernement maintient en permanence des stocks de vaccins et d’outils de détection des menaces biologiques pour protéger la population. Cela est dû à l’augmentation du financement fédéral, aux avancées techniques et à la sensibilisation croissante du public. La présence d’acteurs majeurs est également encourageante pour le marché. En 2018, le gouvernement américain a publié la stratégie nationale de biodéfense et son plan de mise en œuvre, la stratégie nationale de sécurité, la stratégie nationale de défense et la stratégie nationale de lutte contre le terrorisme , ainsi que la stratégie nationale de lutte contre les armes de destruction massive, qui incluent toutes la biosécurité et la biodéfense .
En outre, dans les marchés émergents comme l’Inde, plusieurs institutions, comme l’Organisation de recherche et de développement de la défense, développent des mesures de défense pour lutter contre les attaques biologiques. Des efforts importants sont déployés pour préparer les forces militaires à une attaque biologique. Au cours des deux dernières décennies, on a assisté à une augmentation continue de l’attention portée à la sensibilisation au bioterrorisme dans toutes les nations, ainsi qu’à diverses propositions et réunions visant à lutter contre de telles menaces, ce qui est l’une des principales raisons de la demande en matière de biodéfense. Les initiatives des gouvernements du monde entier ouvrent une fenêtre d’opportunité aux entreprises impliquées dans le secteur de la biodéfense, stimulant ainsi la croissance du marché.
Adoption améliorée des technologies pour favoriser la croissance du marché
L'adoption croissante de technologies telles que les puces génétiques pour la détection des agents pathogènes dans les services médico-légaux, les systèmes de gestion de bases de données, le matériel de détection avancé dans le développement de médicaments liés à la biodéfense et la nanotechnologie est utilisée avec succès pour renforcer les techniques de défense biologique. La plupart de ces technologies sont utilisées soit indépendamment, soit conjointement pour concevoir et développer divers outils de biodéfense. La technologie qui est actuellement la plus importante pour la biodéfense est étroitement liée au génie génétique, une technologie basée sur la manipulation et le transfert artificiels de matériel génétique. Dans le même temps, la thérapie génique pourrait être utilisée efficacement pour guérir diverses maladies. Beaucoup prédisent que les armes biologiques du futur pourraient être des armes biologiques conçues sur mesure, et cette technologie pourrait jouer un rôle très important dans la découverte de remèdes pour de nombreuses maladies inconnues à l'heure actuelle. Les progrès récents en biologie moléculaire et en génie génétique ont conduit à de nouvelles stratégies de développement de vaccins et sont susceptibles de créer des opportunités de croissance considérables pour le marché de la biodéfense à l'avenir.
Analyse de segmentation du rapport sur le marché de la biodéfense
Les segments clés qui ont contribué à l’élaboration de l’analyse du marché de la biodéfense sont par produit.
- En fonction du produit, le marché de la biodéfense est divisé enAnthrax, variole, botulisme, radiation/nucléaire, autres. Le segment de l'anthrax détenait la plus grande part de marché en 2023 et devrait enregistrer le TCAC le plus élevé au cours de la période de prévision.
Analyse des parts de marché de la biodéfense par zone géographique
La portée géographique du rapport sur le marché de la biodéfense est principalement divisée en cinq régions : Amérique du Nord, Asie-Pacifique, Europe, Moyen-Orient et Afrique, et Amérique du Sud et centrale.
L'Amérique du Nord domine le marché de la biodéfense. Les menaces biologiques pesant sur les humains, les animaux et l'environnement comptent parmi les problèmes les plus graves auxquels sont confrontés les États-Unis et leur communauté nationale. Alors que la menace biologique continue de croître, les États-Unis se concentrent sur le renforcement de leurs capacités et considèrent la préparation aux menaces biologiques et au bioterrorisme comme l'un des aspects essentiels de la sécurité nationale. Une attention particulière portée à la sécurité nationale, un système de santé bien établi et de solides capacités de recherche et développement favorisent la croissance du marché dans la région. Grâce à des activités gouvernementales fortes et proactives, à des partenariats avec des acteurs industriels et à un degré élevé de préparation aux menaces bioterroristes, la région a connu des investissements importants dans la technologie de biodéfense, notamment dans les thérapies, les vaccins et les systèmes de détection.
Les stratégies de biodéfense mises en œuvre par des organisations telles que la BARDA, le NIAID, le CDC, l'ASPR et la FDA contribuent à la croissance de la région. Le financement important accordé par les agences militaires et civiles américaines à la biodéfense est également l'un des facteurs clés responsables de la part de revenus la plus élevée de la région.
L'Asie-Pacifique devrait connaître le taux de croissance annuel composé le plus élevé dans les années à venir. En raison de l'augmentation des investissements en R&D, de l'importance de la biodéfense et de la menace croissante des matières biologiques dangereuses qui provoquent des urgences, la croissance du marché dans la région est soutenue. La forte vulnérabilité de la région aux maladies infectieuses et sa population nombreuse ont également accru la demande de produits et de technologies de biodéfense, couplée à la présence d'infrastructures de soins de santé sophistiquées.
Aperçu régional du marché de la biodéfense
Les tendances régionales et les facteurs influençant le marché de la biodéfense tout au long de la période de prévision ont été expliqués en détail par les analystes d’Insight Partners. Cette section traite également des segments et de la géographie du marché de la biodéfense en Amérique du Nord, en Europe, en Asie-Pacifique, au Moyen-Orient et en Afrique, ainsi qu’en Amérique du Sud et en Amérique centrale.
- Obtenez les données régionales spécifiques au marché de la biodéfense
Portée du rapport sur le marché de la biodéfense
| Attribut de rapport | Détails |
|---|---|
| Taille du marché en 2021 | 12,37 milliards de dollars américains |
| Taille du marché d'ici 2031 | 21,62 milliards de dollars américains |
| Taux de croissance annuel composé mondial (2023-2031) | 6,1% |
| Données historiques | 2021-2023 |
| Période de prévision | 2024-2031 |
| Segments couverts |
Par produit
|
| Régions et pays couverts |
Amérique du Nord
|
| Leaders du marché et profils d'entreprises clés |
|
Densité des acteurs du marché de la biodéfense : comprendre son impact sur la dynamique commerciale
Le marché de la biodéfense connaît une croissance rapide, tirée par la demande croissante des utilisateurs finaux en raison de facteurs tels que l'évolution des préférences des consommateurs, les avancées technologiques et une plus grande sensibilisation aux avantages du produit. À mesure que la demande augmente, les entreprises élargissent leurs offres, innovent pour répondre aux besoins des consommateurs et capitalisent sur les tendances émergentes, ce qui alimente davantage la croissance du marché.
La densité des acteurs du marché fait référence à la répartition des entreprises ou des sociétés opérant sur un marché ou un secteur particulier. Elle indique le nombre de concurrents (acteurs du marché) présents sur un marché donné par rapport à sa taille ou à sa valeur marchande totale.
Les principales entreprises opérant sur le marché de la biodéfense sont :
- Nordique bavarois
- Alnylam Pharmaceuticals, Inc.
- SIGA Technologies
- Emergent BioSolutions Inc.
- Laboratoires biologiques de Cleveland
- Dynavax Technologies
Avis de non-responsabilité : les sociétés répertoriées ci-dessus ne sont pas classées dans un ordre particulier.
- Obtenez un aperçu des principaux acteurs du marché de la biodéfense
Actualités et développements récents du marché de la biodéfense
Le marché de la biodéfense est évalué en collectant des données qualitatives et quantitatives issues de recherches primaires et secondaires, qui comprennent d'importantes publications d'entreprises, des données d'associations et des bases de données. Voici une liste des évolutions du marché de la biodéfense et des stratégies :
- En février 2022, Appili Therapeutics Inc. a annoncé un financement de plus de 10 millions de dollars US du ministère américain de la Défense, via le Joint Science and Technology Office de la Defense Threat Reduction Agency, pour faire progresser le candidat vaccin de biodéfense de la société , ATI-1701, un candidat vaccin potentiel de premier plan pour prévenir l'infection par Francisella tularensis. Le financement devrait renforcer la présence de l'entreprise et augmenter la croissance sur le marché de la biodéfense. (Source : Appili Therapeutics, communiqué de presse, 2022)
- En septembre 2022, Emergent BioSolutions Inc. a conclu son accord définitif avec Chimerix, Inc. pour acquérir les droits mondiaux exclusifs de Chimerix sur TEMBEXA (brincidofovir), le premier antiviral oral approuvé par la Food and Drug Administration (FDA) des États-Unis pour le traitement de la variole dans toutes les tranches d'âge. (Source : Emergent BioSolutions Inc., Communiqué de presse, 2022)
- En juillet 2023,Emergent BioSolutions Inc. a reçu l'approbation de la FDA américaine pour CYFENDUS (vaccin contre l'anthrax adsorbé, avec adjuvant), anciennement connu sous le nom d'AV7909, pour la prophylaxie post-exposition de maladies suite à une exposition suspectée ou confirmée à Bacillus anthracis chez les personnes âgées de 18 à 65 ans lorsqu'il est administré en association avec des médicaments antibactériens recommandés. (Source : Emergent BioSolutions Inc., communiqué de presse, 2023)
Rapport sur le marché de la biodéfense : couverture et livrables
Le rapport « Biodefense Market Size and Forecast (2021-2031) » fournit une analyse détaillée du marché couvrant les domaines ci-dessous :
- Taille du marché et prévisions aux niveaux mondial, régional et national pour tous les segments de marché clés couverts par le périmètre
- Dynamique du marché, comme les facteurs moteurs, les contraintes et les opportunités clés
- Principales tendances futures
- Analyse détaillée des cinq forces de PEST/Porter et SWOT
- Analyse du marché mondial et régional couvrant les principales tendances du marché, les principaux acteurs, les réglementations et les développements récents du marché
- Analyse du paysage industriel et de la concurrence couvrant la concentration du marché, l'analyse de la carte thermique, les principaux acteurs et les développements récents
- Profils d'entreprise détaillés
- Analyse historique (2 ans), année de base, prévision (7 ans) avec TCAC
- Analyse PEST et SWOT
- Taille du marché Valeur / Volume - Mondial, Régional, Pays
- Industrie et paysage concurrentiel
- Ensemble de données Excel
Rapports récents
Rapports connexes
Témoignages
Raison d'acheter
- Prise de décision éclairée
- Compréhension de la dynamique du marché
- Analyse concurrentielle
- Connaissances clients
- Prévisions de marché
- Atténuation des risques
- Planification stratégique
- Justification des investissements
- Identification des marchés émergents
- Amélioration des stratégies marketing
- Amélioration de l'efficacité opérationnelle
- Alignement sur les tendances réglementaires






















 Obtenez un échantillon gratuit pour - Marché de la biodéfense
Obtenez un échantillon gratuit pour - Marché de la biodéfense