3D Printing Polymer Material Market Growth, Size & Forecast by 2034
3D Printing Polymer Material for Medical Application Market Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Material [Polyether Ether Ketone (PEEK), Polymethylmethacrylate (PMMA), Polylactic Acid (PLA), and Others] and Application (Dental and Hearing Aids)
Historic Data: 2021-2024 | Base Year: 2025 | Forecast Period: 2026-2034- Report Date : Apr 2026
- Report Code : TIPRE00027712
- Category : Chemicals and Materials
- Status : Upcoming
- Available Report Formats :


- No. of Pages : 150
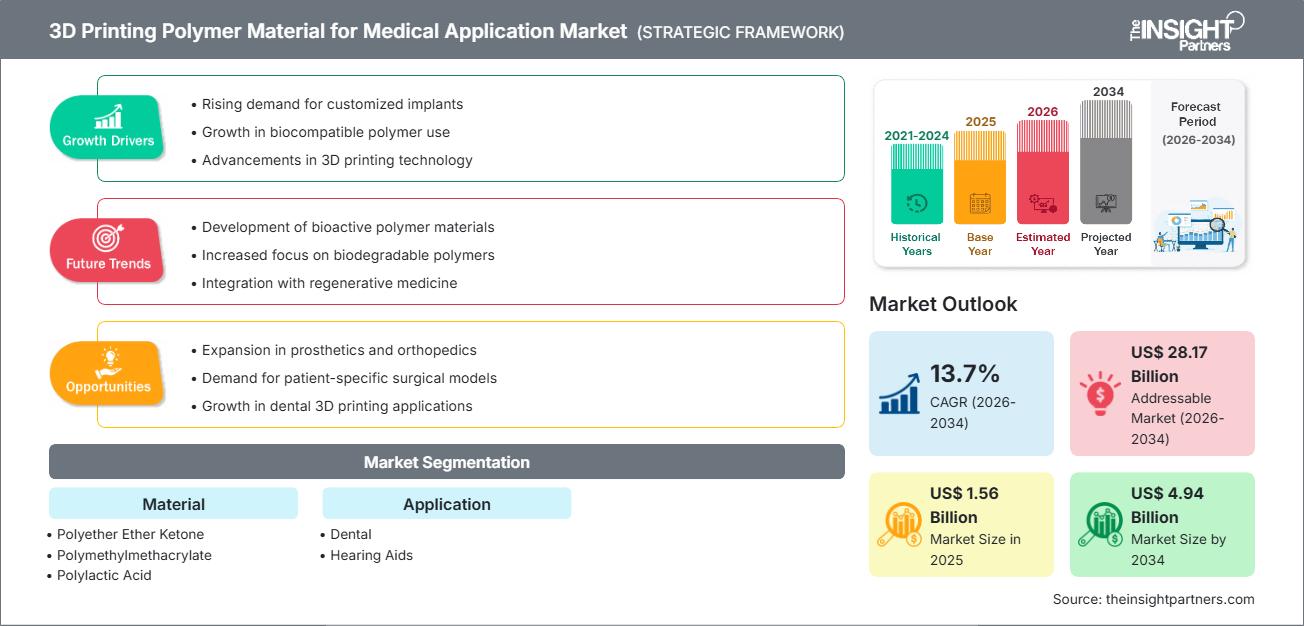
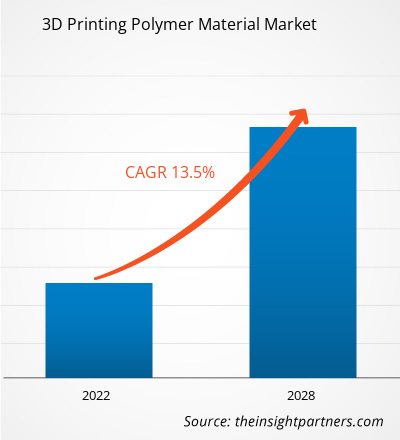
The global 3D Printing Polymer Material market size is projected to reach US$ 4.94 billion by 2034 from US$ 1.56 billion in 2025. The market is anticipated to register a CAGR of 13.7% during the forecast period 2026–2034. Key market dynamics include rapid adoption in dental workflows (surgical guides, aligners, crowns) and accelerating demand for hearing-aid shells, alongside material innovations in medical-grade PEEK, biocompatible photopolymers, and high-reuse PA powders that improve clinical performance and total cost of ownership. Additionally, the market is expected to benefit from the rapid clinical adoption of implant‑grade PEEK solutions, the expansion of digitally enabled dental and hearing‑care workflows across emerging healthcare systems, and the increasing integration of advanced polymer materials, such as radiopaque photopolymers and high‑reusability PA powders, into high‑value medical segments including patient‑specific implants, surgical guides, and CT‑accurate anatomical phantoms.
3D Printing Polymer Material Market Analysis
The 3D Printing Polymer Market analysis indicates a decisive shift from prototyping to regulated end-use medical devices, where biocompatibility evidence, sterilization compatibility, and validated workflows are the primary differentiators. Material strategies are converging on implant-grade PEEK for cranio‑maxillofacial and spine constructs, programmable photopolymers for radiology phantoms and surgical planning, and high‑reusability PA powders for cost-effective, repeatable series production. For suppliers, the most actionable plays include: (1) clinical validation with QMS-backed documentation (e.g., ASTM F2026 compliance for PEEK) to accelerate OEM submissions; (2) platform partnerships with printer OEMs and hospital point‑of‑care labs to embed materials into approved, closed workflows; and (3) portfolio risk balancing between permanent implants (higher regulatory burden, premium pricing) and short-term contact devices (faster adoption, volume). Market entry should prioritize dental/otology use‑cases and radiology education/testing, where turnaround time, anatomical fidelity, and radiopacity tuning are valued, and where buyers increasingly require repeatable, data-rich process windows tied to CFR/FDA and ISO 10993 evidence.
3D Printing Polymer Material Market Overview
3D Printing Polymer has evolved from early photopolymer models to biocompatible, sterilizable parts and patient-specific implants manufactured on certified workflows. Within materials, the industry is moving from standard PLA/ABS toward PEEK/PAEK family, medical photopolymers (e.g., surgical guide resins), and PA11/PA12 powders with improved sustainability and reuse. Dental applications have become mainstream, while hearing‑aid shells are routinely additively manufactured. Concurrently, hospitals and device OEMs leverage radiopaque photopolymers to create CT-accurate phantoms that reduce reliance on cadavers and standard phantoms. The maturing ecosystem features chemicals specialists (Evonik, Arkema), printer OEMs (Stratasys), and service providers co-developing validated materials and application-specific workflows. For instance, the market in the US emphasizes clinically validated, biocompatible polymers, with strong adoption in dental labs, radiology training, and point-of-care printing networks. Partnerships between materials suppliers, OEMs, and hospital systems drive rapid translation from prototyping to routine clinical tools and devices.
Customize This Report To Suit Your Requirement
Get FREE CUSTOMIZATION3D Printing Polymer Material for Medical Application Market: Strategic Insights
-
Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
3D Printing Polymer Material Market Drivers and Opportunities
Market Drivers:
- Clinical-Grade Biocompatibility and Workflow Validation
Hospitals and medtech OEMs demand materials with ISO 10993 data, sterilization compatibility, and audited QMS traceability. Implant-grade PEEK conforming to ASTM F2026 and radiopaque photopolymers with tunable HU values enable predictable, regulatory-ready outputs and accelerate time‑to‑market for patient-specific devices and imaging phantoms. - Shift From Prototyping to End‑Use, Patient-Specific Devices
Dental guides, aligners, and hearing‑aid shells exemplify high-volume, customized production where polymer AM excels. Repeatability, cleanability, and validated post-processing underpin the transition to clinical use, lifting demand for robust powders/resins with consistent mechanical and surface properties. - Cost and Sustainability Pressures in Healthcare Supply Chains
High‑reusability PA12 materials and bio-based PA11 options lower consumables cost and waste while maintaining surface finish and throughput, supporting hospital sustainability initiatives and device makers' cost‑per‑part targets.
Market Opportunities:
- PEEK Implants & Advanced Orthopedics
Emerging workflows for PEEK cranial and spinal implants, including FDA-cleared pathways and carbon‑fiber‑reinforced variants, open premium niches for suppliers who pair materials with process validation, documentation packs, and surgeon education. - Radiology Phantoms & Imaging R&D
Radiopaque, CT-accurate photopolymers enable tunable anatomy models for protocol optimization and AI dataset generation, creating a fast-growing materials niche tightly coupled with imaging OEM collaborations and academic centers. - Sustainable, High‑Reuse Powders for Series Production
Bio-based PA11 and high‑reuse PA12 S powders tailored for MJF and PBF platforms support cost-effective, continuous production of medical housings, guides, and fixtures, especially when combined with third-party post-processing automation.
3D Printing Polymer Material Market Report Segmentation Analysis
The 3D Printing Polymer Material Market share is analyzed across various segments to provide a clearer understanding of its structure, growth potential, and emerging trends. Below is the standard segmentation approach used in most industry reports:
By Material:
- Polyether Ether Ketone (PEEK): High-performance, implant-grade polymer valued for biocompatibility, radiolucency, and bone‑mimetic modulus; increasingly adopted for cranio‑maxillofacial, spinal, and trauma applications via extrusion and laser-based processes.
- Polymethylmethacrylate (PMMA): Used for dental models, temporary restorations, and anatomical visualization where rigidity, optical clarity, and chairside finishing are required.
- Polylactic Acid (PLA): Bio-derived filament preferred for educational models and low-risk contact components; valued for ease of processing and dimensional stability in lab environments.
- Others: Includes PA11/PA12 powders, photopolymers for surgical guides and radiology phantoms, and TPU/PEBA elastomers for soft‑tissue and orthotic applications.
By Application:
- Dental: Encompasses surgical guides, models, splints, and aligner workflows; adoption driven by speed, accuracy, and sterilization compatibility in lab‑to‑clinic processes.
- Hearing Aids: Custom shells and components produced at scale, leveraging polymer AM's fit precision and lightweight comfort for long-term wear.
By Geography:
- North America
- Europe
- Asia Pacific
- South & Central America
- Middle East & Africa
3D Printing Polymer Material for Medical Application Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2025 | US$ 1.56 Billion |
| Market Size by 2034 | US$ 4.94 Billion |
| Global CAGR (2026 - 2034) | 13.7% |
| Historical Data | 2021-2024 |
| Forecast period | 2026-2034 |
| Segments Covered |
By Material
|
| Regions and Countries Covered |
North America
|
| Market leaders and key company profiles |
|
3D Printing Polymer Material for Medical Application Market Players Density: Understanding Its Impact on Business Dynamics
The 3D Printing Polymer Material for Medical Application Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
3D Printing Polymer Material Market Share Analysis by Geography
Asia‑Pacific is expected to grow fastest in the coming years. Emerging markets in South & Central America, the Middle East, and Africa also have untapped potential for material suppliers and device makers to localize validated workflows and training. The market's transformation is fueled by the mainstreaming of dental and hearing‑care applications and the rise of imaging phantoms and point-of-care labs within hospitals.
The market is rapidly professionalizing, moving from generic polymers to medical-grade, evidence-backed materials integrated into end-to-end solutions. Growth is supported by the expansion of certified PEEK offerings, radiopaque photopolymers, and sustainable PA powders designed for repeatable production. Below is a summary of share dynamics and trends by region:
North America
- Market Share: A dominant, innovation-led region with deep ties between materials vendors, OEMs, and academic medical centers.
- Key Drivers:
- Strong demand for dental/orthodontic devices and hearing‑aid shells produced on validated polymer additive manufacturing workflows.
- Adoption of radiopaque photopolymers for CT‑accurate phantoms in training and algorithm development.
- Expanding PEEK implant programs supported by ASTM-compliant materials and FDA-aligned processes.
- Trends: Integration of point-of-care printing and OEM–hospital partnerships to standardize materials, sterilization, and QA around surgical applications.
Europe
- Market Share: A leading base for specialty chemicals and medical device standard‑setting, with strong sustainability orientation.
- Key Drivers:
- Presence of specialty material suppliers advancing implant-grade PEEK portfolios and bio-based PA chains.
- Institutional focus on CE‑markable workflows and harmonized biocompatibility testing for dental/orthopedic devices.
- Consolidation and restructuring among material players are spurring portfolio refocus on high-value medical niches.
- Trends: Circularity and mass‑balance initiatives in polymer powders, plus collaborative testbeds for radiology phantoms and surgical planning materials.
Asia‑Pacific
- Market Share: Fastest‑growing region with strong adoption in dental labs and expanding hospital-based AM capabilities.
- Key Drivers:
- Rapid scaling of dental applications and custom devices for large urban populations.
- Ecosystem depth in powder‑bed and extrusion systems, enabling cost-effective series production with PA12/PA11 and elastomers.
- Academic‑industry consortia are pushing polymer AM for orthopedic models and training across major hubs.
- Trends: Localization of biocompatible materials and qualification services; increasing interest in PEEK for load-bearing applications as regulatory familiarity grows.
South & Central America
- Market Share: An emerging landscape centered on dental markets and medical education models.
- Key Drivers:
- Adoption of cost-optimized photopolymers and powders for labs and teaching hospitals.
- Growing private‑clinic investments in digital dentistry workflows.
- Partnerships with global suppliers to establish validated post-processing and sterilization protocols.
- Trends: Service‑bureau growth offering certified materials and QA to bridge capability gaps for clinics and medtech startups.
Middle East & Africa
- Market Share: Developing market with increasing interest in hospital-based printing and localized device customization.
- Key Drivers:
- Government-backed healthcare modernization, including training centers using radiology phantoms and anatomical models.
- Adoption of durable, sterilizable polymers for surgical instrumentation and guides.
- Strategic procurement of sustainable powders aligned with local industrial initiatives.
- Trends: Emerging qualification hubs to standardize materials handling in hot climates, and rising attention to point-of-care device traceability.
High Market Density and Competition
Competition is intensifying due to the presence of established leaders such as Evonik Industries AG, Arkema, Stratasys Ltd., Henkel AG & Co. KGaA, BASF SE, Formlabs, Roboze, Bionic Production GmbH, and Orion Additive Manufacturing GmbH, which also contribute to a diverse and rapidly expanding market landscape.
This competitive environment pushes vendors to differentiate through:
- Premiumization and Clinical Validation: Implant-grade PEEK portfolios (ASTM F2026), biocompatibility datasets, and sterilization evidence to support regulated submissions.
- Biotech‑Driven Portfolio Diversification: Radiopaque, digital‑anatomy photopolymers for imaging; carbon‑fiber‑reinforced PEEK for higher strength‑to‑weight implants.
- Vertical Integration and Ethical Sourcing: Mass‑balance and bio-based chains for PA11/PA12; service integration to deliver validated, end-to-end workflows.
- Advanced Delivery Technologies: Material–machine co-development for stable print windows, higher powder reuse, and automated post-processing to ensure repeatability.
Opportunities and Strategic Moves
- Co-developing Closed, Validated Workflows with Hospitals and OEMs: Bundle materials, parameters, and sterilization/post-processing SOPs for specific indications (dental guides, CMF implants, CT phantoms).
- Invest in Sustainable Polymers & Documentation Packs: Scale bio-based PA11 and high‑reuse PA12 S with lifecycle data to meet hospital sustainability KPIs and cost‑per‑procedure targets.
Major Companies operating in the 3D Printing Polymer Material Market are:
- Evonik Industries AG
- Arkema
- Stratasys Ltd.
- Henkel AG & Co. KGaA
- BASF SE
- Formlabs
- Roboze
- Bionic Production GmbH
- Orion Additive Manufacturing GmbH
- Solvay S.A
Disclaimer: The companies listed above are not ranked in any particular order.
3D Printing Polymer Material Market News and Recent Developments
- In April 2024, Evonik's VESTAKEEP® i4 3DF PEEK biomaterial, an implant-grade 3D Printing Polymer Material, expanded significantly post-regulatory and clinical validation, reflecting its growing role in advanced medical additive manufacturing.
- In March 2024, HP Inc. officially introduced the PA 12 S material at the annual AMUG (Additive Manufacturing User Group) Conference. It is a high-performance 3D Printing Polymer Material developed in collaboration with Arkema. The PA 12 S material set a new benchmark for surface aesthetics and reduced production costs for users of the Jet Fusion 5200 Series. HP also confirmed that the reach of this material would expand further, as it was scheduled for availability on the Jet Fusion 5600 Series later in the spring, strengthening its role across HP's broader portfolio of polymer 3D printing solutions.
3D Printing Polymer Material Market Report Coverage and Deliverables
The 3D Printing Polymer Material Market Size and Forecast (2021–2034) report provides a detailed analysis of the market covering below areas:
- 3D Printing Polymer Material Market size and forecast at global, regional, and country levels for all the key market segments covered under the scope
- 3D Printing Polymer Material Market trends, as well as market dynamics such as drivers, restraints, and key opportunities
- Detailed PEST and SWOT analysis
- 3D Printing Polymer Material Market analysis covering key market trends, global and regional framework, major players, regulations, and recent market developments
- Industry landscape and competition analysis covering market concentration, heat map analysis, prominent players, and recent developments in the 3D Printing Polymer Material Market.
- Detailed company profiles
Frequently Asked Questions
Habi is a seasoned Market Research Analyst with 8 years of experience specializing in the Chemicals and Materials sector, with additional expertise in the Food & Beverages and Consumer Goods industries. He is a Chemical Engineer from Vishwakarma Institute of Technology (VIT) and has developed deep domain knowledge across industrial and specialty chemicals, paints and coatings, paper and packaging, lubricants, and consumer products. Habi’s core competencies include market sizing and forecasting, competitive benchmarking, trend analysis, client engagement, report writing, and team coordination—making him adept at delivering actionable insights and supporting strategic decision-making.
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Global, Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Testimonials
The Insight Partners' SCADA System Market report is comprehensive, with valuable insights on current trends and future forecasts. The team was highly professional, responsive, and supportive throughout. We are very satisfied and highly recommend their services.
RAN KEDEM Partner, Reali Technologies LTDsI requested a report on a very specific software market and the team produced the report in a few days. The information was very relevant and well presented. I then requested some changes and additions to the report. The team was again very responsive and I got the final report in less than a week.
JEAN-HERVE JENN Chairman, Future AnalyticaWe worked with The Insight Partners for an important market study and forecast. They gave us clear insights into opportunities and risks, which helped shape our plans. Their research was easy to use and based on solid data. It helped us make smart, confident decisions. We highly recommend them.
PIYUSH NAGPAL Sr. Vice President, High Beam GlobalThe Insight Partners delivered insightful, well-structured market research with strong domain expertise. Their team was professional and responsive throughout. The user-friendly website made accessing industry reports seamless. We highly recommend them for reliable, high-quality research services
YUKIHIKO ADACHI CEO, Deep Blue, LLC.This is the first time I have purchased a market report from The Insight Partners.While I was unsure at first, I visited their web site and felt more comfortable to take the risk and purchase a market report.I am completely satisfied with the quality of the report and customer service. I had several questions and comments with the initial report, but after a couple of dialogs over email with their analyst I believe I have a report that I can use as input to our strategic planning process.Thank you so much for taking the extra time and making this a positive experience.I will definitely recommend your service to others and you will be my first call when we need further market data.
JOHN SUZUKI President and Chief Executive Officer, Board Director, BK TechnologiesI wish to appreciate your support and the professionalism you displayed in the course of attending to my request for information regarding to infectious disease IVD market in Nigeria. I appreciate your patience, your guidance, and the fact that you were willing to offer a discount, which eventually made it possible for us to close a deal. I look forward to engaging The Insight Partners in the future, all thanks to the impression you have created in me as a result of this first encounter.
DR CHIJIOKE ONYIA MANAGING DIRECTOR, PineCrest Healthcare Ltd.Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends





 Get Free Sample For
Get Free Sample For