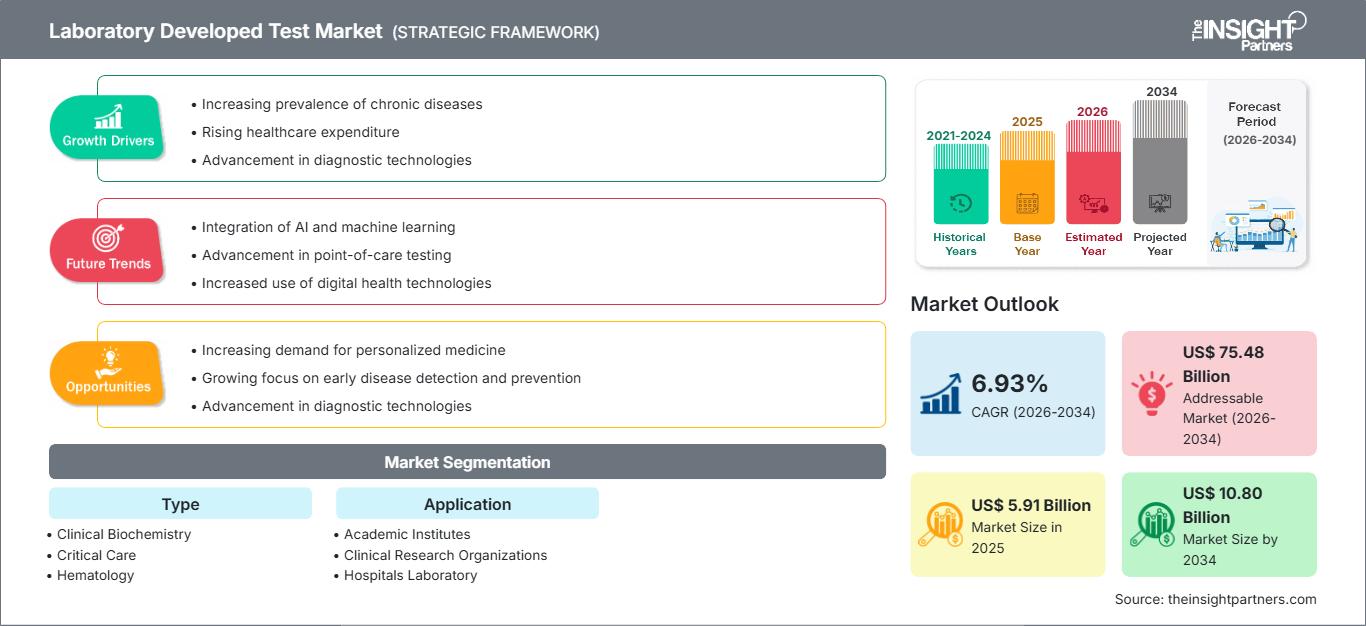
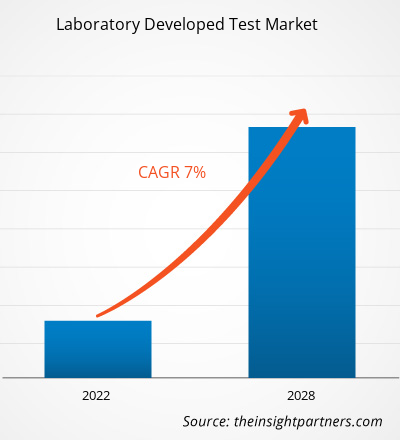
Se espera que el tamaño del mercado de pruebas desarrolladas en laboratorio (LDT) alcance los US$ 10.800 millones para 2034, desde los US$ 5.910 millones en 2025. Se anticipa que el mercado registre una CAGR del 6,93% durante el período 2026-2034.
Análisis del mercado de pruebas desarrolladas en laboratorio
El pronóstico del mercado de pruebas desarrolladas en laboratorio indica un crecimiento sostenido impulsado por la creciente demanda de medicina personalizada, la creciente carga de enfermedades crónicas y raras, los rápidos avances en las tecnologías de pruebas genéticas y moleculares, y el creciente papel de los laboratorios de diagnóstico en la atención médica de precisión. Las pruebas de diagnóstico por laboratorio (LDT) ofrecen velocidad, flexibilidad y capacidades de diagnóstico personalizables, lo que permite a los laboratorios desarrollar ensayos especializados donde no existen pruebas autorizadas por la FDA o disponibles comercialmente.
La expansión del mercado también se ve impulsada por mejoras en la secuenciación de nueva generación (NGS), la automatización del diagnóstico in vitro (IVD) y las plataformas bioinformáticas que permiten a los laboratorios diseñar, validar e implementar sofisticados ensayos de diagnóstico. Además, la evolución de los marcos regulatorios, especialmente en EE. UU. y Europa, impulsa a los laboratorios a adoptar flujos de trabajo de desarrollo que cumplan con las normativas e invertir en sistemas de gestión de calidad.
Descripción general del mercado de pruebas desarrolladas en laboratorio
Las pruebas desarrolladas en laboratorio (LDT) son ensayos de diagnóstico internos diseñados, fabricados y utilizados en un solo laboratorio clínico. Estas pruebas son esenciales para abordar necesidades de diagnóstico no cubiertas en áreas donde no existen pruebas disponibles comercialmente o donde se requiere una rápida personalización.
Las LDT facilitan la toma de decisiones clínicas en diversas especialidades, como oncología, enfermedades infecciosas, cardiología, trastornos genéticos y salud reproductiva. Permiten a los laboratorios ofrecer resultados diagnósticos oportunos, precisos y específicos para cada paciente, un requisito cada vez más importante en la medicina personalizada. Al permitir a los laboratorios innovar y responder rápidamente a enfermedades emergentes, las LDT se están convirtiendo en una parte integral de los ecosistemas de diagnóstico modernos.
Personalice este informe según sus necesidades
Obtenga PERSONALIZACIÓN GRATUITAMercado de pruebas desarrolladas en laboratorio: Perspectivas estratégicas
-
Obtenga las principales tendencias clave del mercado de este informe.Esta muestra GRATUITA incluirá análisis de datos, desde tendencias del mercado hasta estimaciones y pronósticos.
Impulsores y oportunidades del mercado de pruebas desarrollado en laboratorio
Factores impulsores del mercado:
- Creciente demanda de medicina personalizada y de precisión: La transición global hacia la medicina personalizada es un factor clave en la adopción de las LDT. Los médicos recurren cada vez más a herramientas de diagnóstico molecular y genético para adaptar los planes de tratamiento. Las LDT ofrecen la flexibilidad necesaria para diseñar ensayos especializados para una atención individualizada.
- Prevalencia creciente de enfermedades crónicas y raras: La creciente incidencia del cáncer, trastornos hereditarios, enfermedades autoinmunes y enfermedades infecciosas requiere plataformas de diagnóstico avanzadas. Las pruebas de diagnóstico por láser (LDT) permiten la detección temprana, la selección de terapias y el monitoreo, lo que impulsa la demanda del mercado.
- Avances en genómica, NGS y bioinformática: Los avances tecnológicos, especialmente en secuenciación, PCR multiplex y proteómica, permiten a los laboratorios crear pruebas de alta complejidad con una precisión superior. Estas innovaciones aceleran los ciclos de desarrollo de LDT.
- Panorama regulatorio en evolución que fomenta la mejora de la calidad: Marcos regulatorios como CLIA en EE. UU. e IVDR en Europa impulsan a los laboratorios a estandarizar los procesos de desarrollo y mejorar la calidad de las pruebas. Este enfoque en el cumplimiento normativo está incrementando la inversión en el desarrollo de LDT a nivel mundial.
Oportunidades de mercado:
- Expansión en mercados emergentes con infraestructura sanitaria en rápida evolución: Países como China, India, Brasil y la región del Golfo están invirtiendo fuertemente en capacidades de diagnóstico. Esto crea oportunidades para la adopción avanzada de LDT en oncología, genética y enfermedades infecciosas.
- Integración de IA/ML para la interpretación de resultados y la automatización del flujo de trabajo: El análisis basado en IA permite una interpretación más rápida y precisa de los datos de pruebas genómicas y moleculares. Esto transforma las capacidades de LDT al permitir análisis de alto rendimiento con uso intensivo de datos.
- Aumento del uso de LDT para la vigilancia de enfermedades infecciosas y la respuesta a brotes: durante emergencias sanitarias, como brotes virales, las LDT se pueden desarrollar e implementar rápidamente, proporcionando soluciones de diagnóstico más rápidamente que las pruebas disponibles comercialmente.
- Oportunidades para que los laboratorios de alta complejidad certificados por CLIA amplíen sus menús de pruebas: las grandes redes de laboratorios de diagnóstico pueden ampliar su oferta desarrollando paneles LDT patentados, mejorando la competitividad y las oportunidades de ingresos.
Análisis de segmentación del informe de mercado de pruebas desarrolladas en laboratorio
Se analiza la cuota de mercado de pruebas desarrolladas en laboratorio en varios segmentos para obtener claridad sobre la estructura, el potencial de crecimiento y las tendencias emergentes.
Por tipo:
- Bioquímica clínica
- Cuidados críticos
- Hematología
- Microbiología
- Diagnóstico molecular
- Inmunología
Por aplicación:
- Institutos académicos
- Organizaciones de investigación clínica
- Laboratorio de Hospitales
- Centros de diagnóstico especializados
Por geografía:
- América del norte
- Europa
- Asia Pacífico
- América del Sur y Central
- Oriente Medio y África
Perspectivas regionales del mercado de pruebas desarrolladas en laboratorio
Los analistas de The Insight Partners han explicado detalladamente las tendencias y los factores regionales que influyen en el mercado de pruebas desarrolladas en laboratorio durante el período de pronóstico. Esta sección también analiza los segmentos y la geografía del mercado de pruebas desarrolladas en laboratorio en América del Norte, Europa, Asia Pacífico, Oriente Medio y África, y América del Sur y Central.
Alcance del informe de mercado de pruebas desarrolladas en laboratorio
| Atributo del informe | Detalles |
|---|---|
| Tamaño del mercado en 2025 | 5.91 mil millones de dólares estadounidenses |
| Tamaño del mercado en 2034 | US$ 10.80 mil millones |
| CAGR global (2026-2034) | 6,93% |
| Datos históricos | 2021-2024 |
| Período de pronóstico | 2026-2034 |
| Segmentos cubiertos |
Por tipo
|
| Regiones y países cubiertos |
América del norte
|
| Líderes del mercado y perfiles de empresas clave |
|
Densidad de participantes en el mercado de pruebas desarrolladas en laboratorio: comprensión de su impacto en la dinámica empresarial
El mercado de pruebas desarrolladas en laboratorio está creciendo rápidamente, impulsado por la creciente demanda del usuario final debido a factores como la evolución de las preferencias de los consumidores, los avances tecnológicos y un mayor conocimiento de los beneficios del producto. A medida que aumenta la demanda, las empresas amplían su oferta, innovan para satisfacer las necesidades de los consumidores y aprovechan las tendencias emergentes, lo que impulsa aún más el crecimiento del mercado.
- Obtenga una descripción general de los principales actores clave del mercado de pruebas desarrolladas en laboratorio
Análisis de la cuota de mercado de las pruebas desarrolladas en laboratorio por geografía
América del norte
- Cuota de mercado: la mayor cuota a nivel mundial debido a la infraestructura de diagnóstico molecular avanzada y la alta adopción de la medicina de precisión.
-
Factores clave:
- Ecosistema de laboratorio ampliamente extendido con certificación CLIA
- Adopción temprana de NGS y pruebas de alta complejidad
- Fuerte enfoque en oncología y diagnóstico genético
- Tendencias: creciente adopción de flujos de trabajo LDT habilitados para IA y expansión de pruebas descentralizadas.
Europa
- Cuota de mercado: Participación significativa impulsada por estrictos estándares de calidad y la implementación de IVDR.
-
Factores clave:
- Demanda de ensayos de laboratorio de alta calidad y que cumplan con las normas
- Programas nacionales de genómica
- Crecimiento en el diagnóstico de enfermedades raras
- Tendencias: Creciente adopción de plataformas de diagnóstico interoperables para un intercambio fluido de datos de pacientes.
Asia Pacífico
- Cuota de mercado: Región de más rápido crecimiento.
-
Factores clave:
- Iniciativas de genómica y medicina de precisión apoyadas por el gobierno
- Creciente prevalencia del cáncer, trastornos genéticos y enfermedades infecciosas
- Rápida expansión de la infraestructura
- Tendencias: Diagnósticos moleculares impulsados por IA y soluciones de pruebas localizadas para diversas poblaciones.
América del Sur y Central
- Cuota de mercado: Creciendo con la modernización de los sistemas de laboratorio.
-
Factores clave:
- Asociaciones público-privadas
- Necesidad de soluciones de diagnóstico rentables.
- Ampliación de los laboratorios de diagnóstico privados
- Tendencias: software de interpretación basado en la nube y flujos de trabajo LDT rentables.
Oriente Medio y África
- Cuota de mercado: En desarrollo pero mejorando rápidamente.
-
Factores clave:
- Estrategias nacionales de medicina de precisión
- Crecimiento de las capacidades avanzadas de oncología y pruebas genéticas
- Inversiones en infraestructura de diagnóstico
- Tendencias: Integración de LDT en plataformas más amplias de vigilancia de la salud pública y de salud digital.
Densidad de participantes en el mercado de pruebas desarrolladas en laboratorio: impacto en la dinámica empresarial
Las estrategias competitivas se centran en:
- Ampliación de la cartera de productos propios de oncología y pruebas genéticas
- Integración de NGS, IA y bioinformática en el desarrollo de LDT
- Desarrollo rápido de ensayos de enfermedades infecciosas para necesidades de salud pública
- Asociaciones entre redes de laboratorios y proveedores de tecnología
Oportunidades y movimientos estratégicos
- Inversión en plataformas de secuenciación automatizada y pruebas moleculares
- Colaboración con empresas de biotecnología para desarrollar conjuntamente paneles de diagnóstico avanzados
- Expansión a regiones emergentes con creciente demanda de diagnóstico
Principales empresas que operan en el mercado de pruebas desarrolladas en laboratorio
- Quest Diagnostics Incorporated
- F. HOFFMANN-LA ROCHE LTD.
- QIAGEN
- Illumina, Inc.
- Eurofins Scientific
- Biodesix
- Adaptado
- Biotecnologías Bioteranósticas
- Rosetta Genomics Ltd.
Otras empresas analizadas durante el curso de la investigación:
- PerkinElmer Inc.
- Thermo Fisher Scientific Inc.
- Roche Diagnostics
- GeneDx
- 10x Genómica
- Natera Inc.
- Corporación de Ciencias Exactas
- Tecnologías Agilent
- Helix OpCo LLC
Noticias y desarrollos recientes del mercado de pruebas desarrolladas en laboratorio
- Quest Diagnostics amplió su cartera de oncología con el lanzamiento de nuevos paneles LDT basados en NGS para tumores sólidos y neoplasias hematológicas para respaldar iniciativas de oncología de precisión.
- LabCorp anunció la validación de múltiples LDT de patología asistidas por IA, mejorando la precisión del diagnóstico y los tiempos de respuesta en la detección y clasificación del cáncer.
- Los Laboratorios de Mayo Clinic presentaron nuevos paneles LDT para enfermedades raras, que permiten un diagnóstico rápido a través de métodos de secuenciación de alta complejidad.
- Fulgent Genetics amplió sus ofertas de LDT para enfermedades infecciosas, incluidos paneles virales y respiratorios rápidos, fortaleciendo sus capacidades de pruebas de salud pública.
Informe de mercado de pruebas desarrolladas en laboratorio: cobertura y resultados
El informe "Tamaño y pronóstico del mercado de pruebas desarrolladas en laboratorio (2021-2034)" ofrece un análisis detallado que abarca:
- Tamaño y pronóstico del mercado de LDT (global, regional y nacional)
- Tendencias del mercado, impulsores, restricciones y oportunidades
- Análisis PEST y FODA detallado
- Panorama competitivo, análisis de concentración y posicionamiento en el mercado
- Panorama regulatorio (CLIA, IVDR, marcos globales)
- Perfiles de empresas, carteras de productos y desarrollos estratégicos
- Análisis histórico (2 años), año base, pronóstico (7 años) con CAGR
- Análisis PEST y FODA
- Tamaño del mercado, valor/volumen: global, regional y nacional
- Industria y panorama competitivo
- Conjunto de datos de Excel
Informes recientes
Testimonios
Razón para comprar
- Toma de decisiones informada
- Comprensión de la dinámica del mercado
- Análisis competitivo
- Información sobre clientes
- Pronósticos del mercado
- Mitigación de riesgos
- Planificación estratégica
- Justificación de la inversión
- Identificación de mercados emergentes
- Mejora de las estrategias de marketing
- Impulso de la eficiencia operativa
- Alineación con las tendencias regulatorias






















 Obtenga una muestra gratuita para - Mercado de pruebas desarrollado en laboratorio
Obtenga una muestra gratuita para - Mercado de pruebas desarrollado en laboratorio