Hospital Acquired Infection Treatment Market Segments and Growth by 2030
Hospital Acquired Infection Treatment Market Size and Forecasts (2020 - 2030), Global and Regional Share, Trends, and Growth Opportunity Analysis Report Coverage: By Drug Class (Antibacterial Drugs, Antiviral Drugs, Antifungal Drugs, and Others), Infection Type (Urinary Tract Infections, Ventilator-Associated Pneumonia, Surgical Site Infections, Bloodstream Infections, and Other Hospital Infections), Distribution Channel (Hospital Pharmacies, Retail Pharmacies, E-Commerce, and Others), and Geography (North America, Europe, Asia Pacific, South & Central America, and Middle East & Africa)
Historic Data: 2020-2022 | Base Year: 2022 | Forecast Period: 2022-2030- Status : Data Released
- Report Code : TIPRE00029976
- Category : Life Sciences
- No. of Pages : 171
- Available Report Formats :


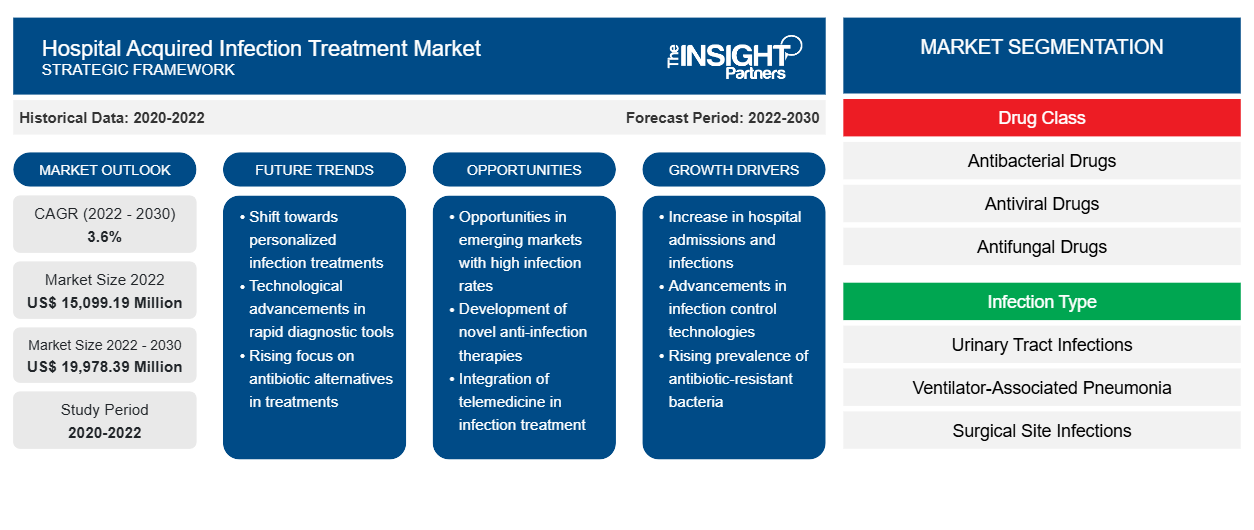
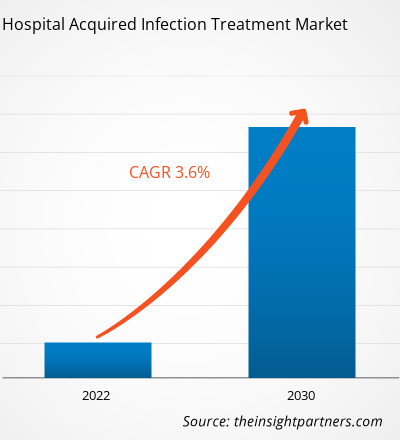
[Research Report] The hospital acquired infection treatment market is expected to grow from US$ 15,099.19 million in 2022 to US$ 19,978.39 million by 2030; it is anticipated to record a CAGR of 3.6% from 2022 to 2030.
Market Insights and Analyst View:
Hospital-acquired infections (HAIs), also known as nosocomially acquired infections, may develop in a variety of settings, including hospitals, long-term care institutions, and ambulatory settings, and they can also appear after discharge. HAIs can also involve occupational infections that affect healthcare workers. Key factors driving the hospital acquired infection treatment market growth are high prevalence of HAIs and growing focus on patient safety and quality care. However, the threat of antimicrobial resistance hinders hospital acquired infection treatment market growth.
Growth Drivers and Restraints:
Central line-associated bloodstream infections, catheter-associated urinary tract infections, and ventilator-associated pneumonia are a few examples of healthcare-associated infections (HAIs). These infections, known as surgical site infections, can also arise at surgical sites. According to the Centers for Disease Control and Prevention (CDC) report “2020 National and State Healthcare-Associated Infections Progress Report" published in 2021, cases of central line-associated bloodstream infections, Methicillin-resistant Staphylococcus aureus (MRSA) bacteremia, and ventilator-associated events increased by 24%, 35%, and 15%, respectively, in the US between 2019 and 2020.
Furthermore, According to the January 2022 report from the National Healthcare Safety Network (NHSN), urinary tract infections (UTIs) are the fifth most common type of healthcare-associated infection. UTIs account for more than 9.5% of acute care hospital conditions. Catheter-Associated Urinary Tract Infection (CAUTI) is caused by urinary catheters used to drain urine from the bladder. CAUTI is one of the most frequent infections among hospitalized UTIs patients. According to the Centers for Disease Control and Prevention (CDC), nearly 75% of infections are related to a urinary catheter. During their hospital stays, 15% to 25% of hospitalized patients need to use urinary catheters. Prolonged urinary catheter use is the most major risk factor for getting CAUTI. CAUTI complications entail patient suffering, prolonged hospital stays, increased treatment costs, and even death.
Furthermore, according to the CDC, UTIs are responsible for more than 13,000 deaths each year. Acute uncomplicated cystitis causes around six days of discomfort, resulting in ∼7 million office visits every year at a cost of US$ 1.6 billion. As a result, rising number of HAI cases places a significant financial burden on the healthcare system. The increasing prevalence of CAUTI, combined with the increased demand for drugs for treatment, is propelling the market.
However, antimicrobial resistance poses a significant challenge to HAI treatment, limiting the efficacy of existing antimicrobial agents and necessitating the development of novel treatment strategies and antimicrobial alternatives to combat resistant pathogens effectively.
Customize This Report To Suit Your Requirement
Get FREE CUSTOMIZATIONHospital Acquired Infection Treatment Market: Strategic Insights
-
Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
Report Segmentation and Scope:
The global hospital acquired infection treatment market is segmented on the basis of drug class, infection type, and distribution channel. Based on drug class, the hospital acquired infection treatment market is segmented into antibacterial drugs, antiviral drugs, antifungal drugs, and others. Based on infection type, the hospital acquired infection treatment is differentiated into urinary tract infections, ventilator-associated pneumonia, surgical site infections, bloodstream infections, and other hospital infections. Based on distribution channel, the market is segmented into hospital pharmacies, retail pharmacies, e-commerce, and others. The hospital acquired infection treatment market, based on geography, is segmented into North America (the US, Canada, and Mexico), Europe (Germany, France, Italy, the UK, Russia, and the Rest of Europe), Asia Pacific (Australia, China, Japan, India, South Korea, and the Rest of Asia Pacific), the Middle East & Africa (South Africa, Saudi Arabia, the UAE, and the Rest of Middle East & Africa), and South & Central America (Brazil, Argentina, and the Rest of South & Central America).
Segmental Analysis:
The hospital acquired infection treatment market, by drug class, is segmented into antibacterial drugs, antiviral drugs, antifungal drugs, and others. The antibacterial drugs segment held the largest market share in 2022 and is anticipated to register the highest CAGR during 2022–2030.
The hospital acquired infection treatment market, by infection type, is segmented into urinary tract infections, ventilator-associated pneumonia, surgical site infections, bloodstream infections, and other hospital infections. The urinary tract infections segment held the largest market share in 2022. However, the ventilator-associated pneumonia segment is anticipated to register the highest CAGR from 2022 to 2030.
The hospital acquired infection treatment market, by distribution channel, is segmented into hospital pharmacies, retail pharmacies, e-commerce, and others. In 2022, the hospital pharmacies segment held the largest market share, and the same segment is anticipated to register the highest CAGR during 2022–2030.
Regional Analysis:
Based on geography, the global hospital acquired infection treatment market is segmented into five key regions—North America, Europe, Asia Pacific, South & Central America, and the Middle East & Africa.
In 2022, North America held the largest share of the global hospital acquired infection treatment market size. The US has a high prevalence of HAI, which propels the hospital acquired infection treatment market growth. As per the CDC, on any given day, about 1 in 31 hospital patients has at least one HAI. Similarly, according to the Office of Disease Prevention and Health Promotion, HAIs and other infections can lead to sepsis, and it causes an ∼1.7 million illness cases and 270,000 deaths per year in the US. Thus, the high prevalence of HAI among people in the US fuels the hospital acquired infection treatment market growth.
Industry Developments and Future Opportunities:
Various initiatives taken by key players operating in the global hospital acquired infection treatment market are listed below:
- In May 2023, Innoviva Specialty Therapeutics announced that the US Food and Drug Administration (FDA) had approved XACDURO (sulbactam for injection; durlobactam for injection), co-packaged for intravenous use in patients aged 18 years and above for the treatment of hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP) caused by susceptible isolates of Acinetobacter baumannii-calcoaceticus complex (Acinetobacter). Innoviva Specialty Therapeutics stated that it is focused on delivering innovative therapies in critical care and infectious diseases.
- In May 2023, Researchers of the Indian Institute of Science Education and Research (IISER), Pune and Central Drug Research Institute (CSIR-CDRI), Lucknow announced that they had discovered a potential new antibiotic against Acinetobacter baumannii, which frequently causes HAIs.
- In September 2020, Shionogi & Co Ltd announced that the FDA had approved a supplemental New Drug Application (sNDA) for FETROJA (cefiderocol) for the treatment of patients aged 18 years and above with hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP) caused by susceptible Gram-negative microorganisms such as Enterobacter cloacae complex, Klebsiella pneumoniae, Acinetobacter baumannii complex, Escherichia coli, Pseudomonas aeruginosa, and Serratia marcescens.
- In June 2020, Merck announced that the US Food and Drug Administration (FDA) had approved a supplemental New Drug Application (sNDA) for RECARBRIO (imipenem, cilastatin, and relebactam) for the treatment of patients aged 18 years and above suffering from hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia (HABP/VABP), caused by susceptible Gram-negative microorganisms such as Acinetobacter calcoaceticus-baumannii complex, Klebsiella oxytoca, Klebsiella pneumoniae, Enterobacter cloacae, Escherichia coli, Haemophilus influenzae, Klebsiella aerogenes, Pseudomonas aeruginosa, and Serratia marcescens.
Hospital Acquired Infection Treatment Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2022 | US$ 15,099.19 Million |
| Market Size by 2030 | US$ 19,978.39 Million |
| Global CAGR (2022 - 2030) | 3.6% |
| Historical Data | 2020-2022 |
| Forecast period | 2022-2030 |
| Segments Covered |
By Drug Class
|
| Regions and Countries Covered |
North America
|
| Market leaders and key company profiles |
|
Hospital Acquired Infection Treatment Market Players Density: Understanding Its Impact on Business Dynamics
The Hospital Acquired Infection Treatment Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
Competitive Landscape and Key Companies:
Merck & Co Inc, Pfizer Inc, AbbVie Inc, Paratek Pharmaceuticals Inc, Eugia US LLC, Abbott Laboratories, Cumberland Pharmaceuticals Inc, Innoviva Specialty Therapeutics Inc, Cipla Ltd, and Eli Lilly and Company are among the prominent players operating in the hospital acquired infection treatment market. These companies focus on new technologies, advancements in existing products, and geographic expansions to meet the growing consumer demand worldwide and increase their product range in specialty portfolios.
Frequently Asked Questions
Mrinal is a seasoned research analyst with over 8 years of experience in Life Sciences Market Intelligence and Consulting. With a strategic mindset and unwavering commitment to excellence, she has built deep expertise in pharmaceutical forecasting, market opportunity assessment, and developing industry benchmarks. Her work is anchored in delivering actionable insights that empower clients to make informed strategic decisions.
Mrinal’s core strength lies in translating complex quantitative datasets into meaningful business intelligence. Her analytical acumen is instrumental in shaping go-to-market (GTM) strategies and uncovering growth opportunities across the pharmaceutical and medical device sectors. As a trusted consultant, she consistently focuses on streamlining workflow processes and establishing best practices, thereby driving innovation and operational efficiency for her clients.
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Global, Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Testimonials
The Insight Partners' SCADA System Market report is comprehensive, with valuable insights on current trends and future forecasts. The team was highly professional, responsive, and supportive throughout. We are very satisfied and highly recommend their services.
RAN KEDEM Partner, Reali Technologies LTDsI requested a report on a very specific software market and the team produced the report in a few days. The information was very relevant and well presented. I then requested some changes and additions to the report. The team was again very responsive and I got the final report in less than a week.
JEAN-HERVE JENN Chairman, Future AnalyticaWe worked with The Insight Partners for an important market study and forecast. They gave us clear insights into opportunities and risks, which helped shape our plans. Their research was easy to use and based on solid data. It helped us make smart, confident decisions. We highly recommend them.
PIYUSH NAGPAL Sr. Vice President, High Beam GlobalThe Insight Partners delivered insightful, well-structured market research with strong domain expertise. Their team was professional and responsive throughout. The user-friendly website made accessing industry reports seamless. We highly recommend them for reliable, high-quality research services
YUKIHIKO ADACHI CEO, Deep Blue, LLC.This is the first time I have purchased a market report from The Insight Partners.While I was unsure at first, I visited their web site and felt more comfortable to take the risk and purchase a market report.I am completely satisfied with the quality of the report and customer service. I had several questions and comments with the initial report, but after a couple of dialogs over email with their analyst I believe I have a report that I can use as input to our strategic planning process.Thank you so much for taking the extra time and making this a positive experience.I will definitely recommend your service to others and you will be my first call when we need further market data.
JOHN SUZUKI President and Chief Executive Officer, Board Director, BK TechnologiesI wish to appreciate your support and the professionalism you displayed in the course of attending to my request for information regarding to infectious disease IVD market in Nigeria. I appreciate your patience, your guidance, and the fact that you were willing to offer a discount, which eventually made it possible for us to close a deal. I look forward to engaging The Insight Partners in the future, all thanks to the impression you have created in me as a result of this first encounter.
DR CHIJIOKE ONYIA MANAGING DIRECTOR, PineCrest Healthcare Ltd.Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends





 Get Free Sample For
Get Free Sample For