The vaccines market size is expected to grow from US$ 135.28 billion in 2022 to US$ 216.30 billion by 2030; the market is estimated to register a CAGR of 6.0% from 2022 to 2030.
Analyst’s Viewpoint
The vaccines market analysis explains market drivers such as the rising prevalence of infectious diseases and the increasing uptake of vaccines amid the COVID-19 pandemic. Further, innovation in vaccine development is expected to introduce new market trends during the forecast period. Based on type, the market is segmented into live attenuated vaccines, inactivated vaccines, toxoid vaccines, subunit & conjugate vaccines, and others. The live attenuated vaccines segment held the largest market share in 2022. Based on application, the vaccines market is segregated into human papillomavirus (HPV), cancer, MMR, DPT, and others. The HPV segment held the largest market share in 2022. By route of administration, the vaccines market is categorized into oral, nasal, and injectable. The injectable segment held the largest share of the market in 2022. By age, the pediatric segment will account for a larger share of the global vaccines market for the forecast period of 2022–2030. By end user, the vaccination centers & program segment will account for the largest share of the global vaccines market during 2022–2030.
A vaccine is a biological preparation that provides active acquired immunity to a particular infectious or malignant disease. A vaccine preparation typically contains an agent responsible for resembling a disease-causing microorganism is often made from a weakened or killed form of the microbe, toxin, or one of its surface proteins. Vaccines can be prophylactic or therapeutic.
Market Insights
Rising Infectious Diseases
According to the 2020 report of the Connecticut Department of Public Health, the number of cases of infectious diseases recorded in 2020 in Connecticut is given below.
Reported Cases of Connecticut Reportable Diseases in 2020 | |
Disease | Total |
Hepatitis A | 15 |
Influenza | 98 |
Malaria | 4 |
Tuberculosis | 54 |
SARS-CoV-2 | 200,379 |
Source: The Insight Partners Analysis
Additionally, the table below reveals notable infectious diseases as per the Center for Health Protection report in 2020:
Notable Infectious Disease | |
Disease | Total |
SARS-CoV-2 | 8,847 |
Chickenpox | 1,987 |
Community-associated methicillin-resistant Staphylococcus aureus infection | 813 |
Dengue Fever | 22 |
Tuberculosis | 3,656 |
Source: The Insight Partners Analysis
Among all the infectious diseases, SARS-CoV-2 was all-time high during the 2020 to 2022 period where other uptake of vaccines including Dengue, Chickenpox, Tuberculosis, and others were low. However, SARS-CoV-2 vaccine uptake was high due to the rising cases globally. According to the Public Health England report published in 2021, 12-month UK coverage for DTaP/IPV/Hib/HepB3 vaccination increased by 0.1% to 92.2% and Rotavirus by 0.3% to 90.6%. However, MenB2 decreased by 0.1%, accounting for 92.3% compared to the previous year. Also, the NHS England report revealed that, in December 2022, a total of 88.9% of all adult care home residents had been vaccinated with a COVID-19 booster dose. Therefore, due to the rising prevalence of infectious diseases, the demand for vaccination is high, resulting in overall market growth.
Market Trend
Innovation in Vaccine Development
The global vaccine development is subject to change due to the COVID-19 pandemic accelerating technological adoption. For example, mRNA vaccine innovation and new methods of administration, including intranasal, are the most innovative vaccine development methods. Additionally, manufacturers are trying different ways to improve these vaccines and deliver them in a way that makes them more productive and enhances the immunogenicity, durability, and overall efficacy.
Furthermore, more streamlined regulatory processes and standardization for clinical trial protocol played a crucial role in reducing the timeline for vaccine development from 10 years to 260 days for the COVID-19 vaccine, which is another example of vaccine development innovation. Such innovative vaccine delivery methods and favorable vaccine development innovation are responsible for the market growth for the forecast period of 2022–2030.
Customize Research To Suit Your Requirement
We can optimize and tailor the analysis and scope which is unmet through our standard offerings. This flexibility will help you gain the exact information needed for your business planning and decision making.
Vaccines Market: Strategic Insights
Market Size Value in US$ 135.28 billion in 2022 Market Size Value by US$ 216.30 billion by 2030 Growth rate CAGR of 6.0% from 2022 to 2030 Forecast Period 2022-2030 Base Year 2022

Akshay
Have a question?
Akshay will walk you through a 15-minute call to present the report’s content and answer all queries if you have any.
 Speak to Analyst
Speak to Analyst
Customize Research To Suit Your Requirement
We can optimize and tailor the analysis and scope which is unmet through our standard offerings. This flexibility will help you gain the exact information needed for your business planning and decision making.
Vaccines Market: Strategic Insights

| Market Size Value in | US$ 135.28 billion in 2022 |
| Market Size Value by | US$ 216.30 billion by 2030 |
| Growth rate | CAGR of 6.0% from 2022 to 2030 |
| Forecast Period | 2022-2030 |
| Base Year | 2022 |

Akshay
Have a question?
Akshay will walk you through a 15-minute call to present the report’s content and answer all queries if you have any.
 Speak to Analyst
Speak to Analyst
Report Segmentation and Scope
Market Opportunity
Therapeutic Vaccine Development
Traditional vaccines stimulate the production of antibodies and immune proteins that target specific pathogens like viruses and bacteria. Likewise, therapeutic vaccines stimulate the immune system to target cancer cells and control the progression of chronic infections like HIV. Further, therapeutic vaccines typically stimulate the recipient's immune system to slow or stop chronic disease progression or target cancer cells. Further, therapeutic vaccines for the treatment of noninfectious diseases encompass a wide variety of possible formulations, antigens, and mechanism of action. Therefore, therapeutic vaccines development will provide lucrative market opportunity for the forecast period 2022-2030.
Examples of Therapeutic Vaccines in Development | |
Indication | Development Phase |
Ovarian Cancer | Phase 1 |
Multiple Myeloma | Phase 1 |
Type 1 Diabetes | Preclinical |
Acute Myelogenous Leukemia | 3 |
Mesothelioma | 2 |
Source: The Insight Partners Analysis
Type-Based Insights
Based on type, the vaccines market is segmented into live attenuated vaccines, inactivated vaccines, toxoid vaccines, subunit & conjugate vaccines, and others. The live attenuated vaccines segment held the largest market share in 2022. Live attenuated vaccines are very effective vaccines used in the prevention of a variety of diseases, including influenza, chickenpox, measles, polio, and tuberculosis. As per the World Health Organization (WHO) report, there are nearly a billion cases of seasonal influenza, including 5 million cases of severe illness. Also, it causes ~290,000 to 650,000 respiratory deaths annually. Live-attenuated influenza vaccine (LAIV) proves advantageous in combating the infection among the population. For example, LAIV can provide up to 90% protection to adults under 65 years of age and up to 40% to adults over 65. The aforementioned factors are responsible for the market growth for the segment for the forecast period of 2022–2030.
Application-Based Insights
Based on application, the vaccines market is categorized into human papillomavirus (HPV), cancer, MMR, DPT, and others. The HPV segment held the largest market share in 2022. According to the WHO report, HPV is highly prevalent among women in Sub-Saharan Africa (24%), followed by Latin America and the Caribbean (16%), Eastern Europe (14%), and Southeast Asia (14%). Also, 625,600 women and 694,000 men have HPV-related cancer annually globally. Also, in 2020, cervical cancer accounted for 93% of HPV-related cancers among women. Therefore, there is a high demand for HPV vaccines among the population due to rising cases, as these vaccines protect against genital warts and most cases of cervical cancer. Also, HPV vaccines are highly approved by the US Food and Drug Administration (USFDA). "GARDASIL 9" is one such example of an HPV vaccine approved by the USFDA. The aforementioned factors are responsible for the market growth for the segment during the forecast period of 2022–2030.
Route of Administration-Based Insights
In terms of route of administration, the vaccines market is categorized into oral, nasal, and injectable. The injectable segment held the largest share of the market in 2022. The most common route of vaccine administration is through injectables, as administering a vaccine through a subcutaneous or intradermal route may cause local irritation, skin discoloration, inflammation, and many more.
Further, the CDC report reveals that DTaP, DT, HepA, HepB, Hib, HPV, IIV4, RIV4, ccIIV4, IPV*†, MenACWY, MenB, MMR‡ , PCV13, PPSV23*†, RZV, Td, Tdap, TT, VAR† are common vaccines administered through the injectable route of administration. Varied vaccines administered through injectable routes act as a standalone factor responsible for the market growth for the segment during the forecast period of 2022–2030.
Surgical Navigation Systems Market, by Type – 2022 and 2030
- Sample PDF showcases the content structure and the nature of the information with qualitative and quantitative analysis.
- Sample PDF showcases the content structure and the nature of the information with qualitative and quantitative analysis.
Regional Analysis
North America accounted for the largest share of the vaccines market. The market in North America is subsegmented into the US, Canada, and Mexico. The US holds the largest share of the vaccines market in this region.
The rising government support for vaccine development enhances vaccine production in the US alone. For example, in March 2023, the US Government announced an investment of US$ 31.9 billion in mRNA vaccine research and procurement. The study of vaccines is based on extensive analysis of US government research grants and procurement contracts related to mRNA vaccines and technologies. After the pandemic, US$ 29.2 billion (92%) of the US public funds announced vaccine procurement, with US$ 2.2 billion (7%) supporting clinical trials and US$ 108 million supporting manufacturing and science. Further, companies in the US get approval for vaccines favorably. For instance, in September 2023, Pfizer and BioNTech announced receiving USFDA approvals of "supplemental Biologics License Application (COMIRNATY 2023-2024 Formulation)" for individuals 12 years and older with emergency use authorization (EUA) for six months individuals. The newly approved vaccine is available in pharmacies, hospitals, and clinics across the US. Fast approvals and a rise in government support for vaccine development and manufacturing are responsible for the major market share of the US in the regional market.
Asia Pacific is expected to register the highest CAGR in the global vaccine market. Asia Pacific accounts for the largest share of R&D spending, with large publications and patents on vaccine research & development (R&D). Also, the region is home to state-owned and private pharmaceutical firms and contract research organizations (CROs) to conduct vaccine R&D. Further, countries in Asia Pacific utilize several supply-side and demand-side approaches to incentivize investment in vaccine R&D. For example, high-income countries in Asia Pacific are major contributors to product development partnerships launching vaccine development programs to boost innovative vaccine manufacturing and research.
During the COVID-19 pandemic, many high and middle-income countries in Asia Pacific established vaccine development manufacturing setups. Regional institutions and intergovernmental organizations in Asia Pacific helped promote and coordinate regional cooperation in vaccine R&D. Therefore, Asia Pacific favorable policies for conducting vaccine development programs to boost innovative vaccine manufacturing and research activities is a standalone factor responsible for accounting for the highest CAGR for the vaccine market during 2022-2030.
The report profiles leading players operating in the global vaccines market. These include GSK, Pfizer, Sanofi, Merck, Serum Institute of India (SII), AstraZeneca, BioNTech, Biological E, Sinovac, Sinopharm, and Bharat Biotech. In August 2022, GSK announced the acquisition of Affinivax, Inc., aiming to build a strong portfolio of specialty medicines and vaccines. The acquisition includes a next-generation 24 valent pneumococcal vaccine that is currently in Phase 2 development and is based on highly innovative Multiple Antigen Presenting System (MAPS) platform technology. The MAPS technology supports higher valency than conventional conjugational technologies and less broad coverage against prevalent pneumococcal serotypes. It generates higher antibody responses for many individual serotypes than current pneumococcal vaccines.
In January 2022, Pfizer Inc. announced a collaboration with BioNTech for new research, development, and commercialization to develop a potential first mRNA-based vaccine for the prevention of shingles. Also, the collaboration builds on the company's success in developing the first approved and most widely used mRNA vaccine to help prevent COVID-19.
Company Profiles
- GSK
- Pfizer
- Sanofi
- Merck
- Serum Institute of India (SII)
- AstraZeneca
- BioNTech
- Biological E
- Sinovac
- Sinopharm
- Bharat Biotech

Report Coverage
Revenue forecast, Company Analysis, Industry landscape, Growth factors, and Trends
Segment Covered
Type, Technology, Application, Route of Administration, End User, and Geography

Regional Scope
North America, Europe, Asia Pacific, Middle East & Africa, South & Central America
Country Scope
This text is related
to country scope.
Frequently Asked Questions
The growth of the market is attributed to the rising prevalence of infectious diseases, growing focus on implementation of immunization programs, and increasing support for vaccine development.
Pfizer Inc., GlaxoSmithKline plc, Merck & Co. Inc, Sanofi, Johnson & Johnson Services, Inc., Panacea Biotec Limited, Astellas Pharma Inc., NOVAVAX, INC., VBI Vaccines Inc., and Bavarian Nordic are some of the major players in the vaccines market.
A vaccine is a biological preparation that provides active acquired immunity against a particular disease. A vaccine consists of an agent that resembles or is a part of the disease-causing microorganism and is often made from weakened or killed forms of the microorganism, its toxins, or one of its surface proteins. The agent triggers the body's immune system to recognize the agent as a threat, destroy it, and to further recognize and destroy any of the microorganisms associated with that agent that it may encounter in the future.
The List of Companies - Vaccines Market
- GSK
- Pfizer
- Sanofi
- Merck
- Serum Institute of India (SII)
- AstraZeneca
- BioNTech
- Biological E
- Sinovac
- Sinopharm
- Bharat Biotech
The Insight Partners performs research in 4 major stages: Data Collection & Secondary Research, Primary Research, Data Analysis and Data Triangulation & Final Review.
- Data Collection and Secondary Research:
As a market research and consulting firm operating from a decade, we have published many reports and advised several clients across the globe. First step for any study will start with an assessment of currently available data and insights from existing reports. Further, historical and current market information is collected from Investor Presentations, Annual Reports, SEC Filings, etc., and other information related to company’s performance and market positioning are gathered from Paid Databases (Factiva, Hoovers, and Reuters) and various other publications available in public domain.
Several associations trade associates, technical forums, institutes, societies and organizations are accessed to gain technical as well as market related insights through their publications such as research papers, blogs and press releases related to the studies are referred to get cues about the market. Further, white papers, journals, magazines, and other news articles published in the last 3 years are scrutinized and analyzed to understand the current market trends.
- Primary Research:
The primarily interview analysis comprise of data obtained from industry participants interview and answers to survey questions gathered by in-house primary team.
For primary research, interviews are conducted with industry experts/CEOs/Marketing Managers/Sales Managers/VPs/Subject Matter Experts from both demand and supply side to get a 360-degree view of the market. The primary team conducts several interviews based on the complexity of the markets to understand the various market trends and dynamics which makes research more credible and precise.
A typical research interview fulfils the following functions:
- Provides first-hand information on the market size, market trends, growth trends, competitive landscape, and outlook
- Validates and strengthens in-house secondary research findings
- Develops the analysis team’s expertise and market understanding
Primary research involves email interactions and telephone interviews for each market, category, segment, and sub-segment across geographies. The participants who typically take part in such a process include, but are not limited to:
- Industry participants: VPs, business development managers, market intelligence managers and national sales managers
- Outside experts: Valuation experts, research analysts and key opinion leaders specializing in the electronics and semiconductor industry.
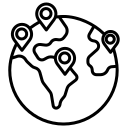
Below is the breakup of our primary respondents by company, designation, and region:
Once we receive the confirmation from primary research sources or primary respondents, we finalize the base year market estimation and forecast the data as per the macroeconomic and microeconomic factors assessed during data collection.
- Data Analysis:
Once data is validated through both secondary as well as primary respondents, we finalize the market estimations by hypothesis formulation and factor analysis at regional and country level.
- 3.1 Macro-Economic Factor Analysis:
We analyse macroeconomic indicators such the gross domestic product (GDP), increase in the demand for goods and services across industries, technological advancement, regional economic growth, governmental policies, the influence of COVID-19, PEST analysis, and other aspects. This analysis aids in setting benchmarks for various nations/regions and approximating market splits. Additionally, the general trend of the aforementioned components aid in determining the market's development possibilities.
- 3.2 Country Level Data:
Various factors that are especially aligned to the country are taken into account to determine the market size for a certain area and country, including the presence of vendors, such as headquarters and offices, the country's GDP, demand patterns, and industry growth. To comprehend the market dynamics for the nation, a number of growth variables, inhibitors, application areas, and current market trends are researched. The aforementioned elements aid in determining the country's overall market's growth potential.
- 3.3 Company Profile:
The “Table of Contents” is formulated by listing and analyzing more than 25 - 30 companies operating in the market ecosystem across geographies. However, we profile only 10 companies as a standard practice in our syndicate reports. These 10 companies comprise leading, emerging, and regional players. Nonetheless, our analysis is not restricted to the 10 listed companies, we also analyze other companies present in the market to develop a holistic view and understand the prevailing trends. The “Company Profiles” section in the report covers key facts, business description, products & services, financial information, SWOT analysis, and key developments. The financial information presented is extracted from the annual reports and official documents of the publicly listed companies. Upon collecting the information for the sections of respective companies, we verify them via various primary sources and then compile the data in respective company profiles. The company level information helps us in deriving the base number as well as in forecasting the market size.
- 3.4 Developing Base Number:
Aggregation of sales statistics (2020-2022) and macro-economic factor, and other secondary and primary research insights are utilized to arrive at base number and related market shares for 2022. The data gaps are identified in this step and relevant market data is analyzed, collected from paid primary interviews or databases. On finalizing the base year market size, forecasts are developed on the basis of macro-economic, industry and market growth factors and company level analysis.
- Data Triangulation and Final Review:
The market findings and base year market size calculations are validated from supply as well as demand side. Demand side validations are based on macro-economic factor analysis and benchmarks for respective regions and countries. In case of supply side validations, revenues of major companies are estimated (in case not available) based on industry benchmark, approximate number of employees, product portfolio, and primary interviews revenues are gathered. Further revenue from target product/service segment is assessed to avoid overshooting of market statistics. In case of heavy deviations between supply and demand side values, all thes steps are repeated to achieve synchronization.
We follow an iterative model, wherein we share our research findings with Subject Matter Experts (SME’s) and Key Opinion Leaders (KOLs) until consensus view of the market is not formulated – this model negates any drastic deviation in the opinions of experts. Only validated and universally acceptable research findings are quoted in our reports.
We have important check points that we use to validate our research findings – which we call – data triangulation, where we validate the information, we generate from secondary sources with primary interviews and then we re-validate with our internal data bases and Subject matter experts. This comprehensive model enables us to deliver high quality, reliable data in shortest possible time.
Trends and growth analysis reports related to Vaccines Market

Nov 2023
Bioprocessing Market
Size and Forecast (2021 - 2031), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Product (Instruments, and Consumables and Accessories), Scale of Operation (Commercial Operations and Clinical Operations), Process (Downstream Bioprocess and Upstream Bioprocess), Application (Monoclonal Antibodies, Vaccines, Recombinant Protein, Cell and Gene Therapy, and Others), End User (Biopharmaceutical Companies, Contract Manufacturing Organization, and Others), and Geography (North America, Europe, Asia Pacific, South & Central America, and Middle East & Africa)

Nov 2023
Bioproduction Market
Size and Forecasts (2020 - 2030), Global and Regional Share, Trends, and Growth Opportunity Analysis Report Coverage: By Product (Biologics and Biosimilars, Vaccines, Cell and Gene Therapies, Nucleic Acid Therapeutics, and Others), Application (Rheumatoid Arthritis, Hematological Disorders, Cancer, Diabetes, Cardiovascular Diseases, and Others), Equipment (Upstream Equipment, Downstream Equipment, Bioreactors, and Consumables and Accessories), End User (Biopharmaceutical Companies, Contract Manufacturing Organizations, and Others), and Geography (North America, Europe, Asia Pacific, Middle East & Africa, and South & Central America)

Nov 2023
ELISA Diagnostic Test Market
Size and Forecasts (2020 - 2030), Global and Regional Share, Trends, and Growth Opportunity Analysis Report Coverage: By Adoption (Human and Veterinary), Test Type (Sandwich ELISA, Indirect ELISA, Competitive ELISA, and Multiple & Portable ELISA), Application (Autoimmune Diseases, Infectious Diseases, Cancer Diagnosis, Protein Quantification, and Others), End User (Hospitals & Diagnostic Centers, Pharmaceutical & Biotechnology Companies, Veterinary Hospitals & Diagnostic Laboratories, and Others), and Geography (North America, Europe, Asia Pacific, South & Central America, and Middle East & Africa)

Nov 2023
Swine Diagnostics Market
Size and Forecasts (2022 - 2030), Global and Regional Share, Trends, and Growth Opportunity Analysis Report Coverage: By Product Type [Immunoassays Kits, PCR Kits, Hemagglutination Inhibition (HI), and Others], Sample Type (Blood, Oral Fluids, Nasal Swabs, Tissue Samples, and Others), Disease [African Swine Fever (ASF)/Classical Swine Fever (CSF), Respiratory Diseases, Porcine Circovirus, Porcine Epidemic Diarrhea, Swine Dysentery, and Others], End User (Veterinary Hospitals, Veterinary Clinics, and Other End Users), and Geography

Nov 2023
Microbial Identification Methods Market
Size and Forecasts (2020 - 2030), Global and Regional Share, Trends, and Growth Opportunity Analysis Report Coverage: By Method (Genotypic, Phenotypic, and Proteotypic), Type (Bacterial Identification System, Microbial Enumeration System, Bacterial Resistance Identification Systems, Microbiology Analyzer, and Others), and Geography (North America, Europe, Asia Pacific, Middle East & Africa, and Latin America)

Nov 2023
Biopharmaceuticals Market
Forecast to 2028 - COVID-19 Impact and Global Analysis By Product Type (Monoclonal Antibodies, Recombinant Vaccines, Conventional Vaccines, Recombinant Growth Factors, Purified Proteins, Recombinant Proteins, Recombinant Hormones, Recombinant Enzymes, Cell & Gene Therapies, Cytokines/Interferon/Interleukins, and Others) and Application (Oncology, Inflammatory & Infectious Disease, Autoimmune Disorders, Metabolic Disorders, Hormonal Disorders & Growth Failure, Cardiovascular Diseases, Neurological Diseases, and Others)

Nov 2023
GMP Cell Therapy Consumables Market
Forecast to 2028 - COVID-19 Impact and Global Analysis by Product (Kits, Reagents/Molecular Biology Reagents, Growth Factors/Cytokines and Interleukins, and Others), Cell Therapy (NK Cell Therapy, Stem Cell Therapy, T-Cell Therapy, and Others), Process (Cell Collection and Characterization/Sorting and Separation, Cell Culture and Expansion/Preparation, Cryopreservation, Cell Processing and Formulation, Cell Isolation and Activation, Cell Distribution/Handling, Process Monitoring and Control/Readministration/Quality Assurance, and Others), and End Use (Clinical, Commercial, and Research)

Nov 2023