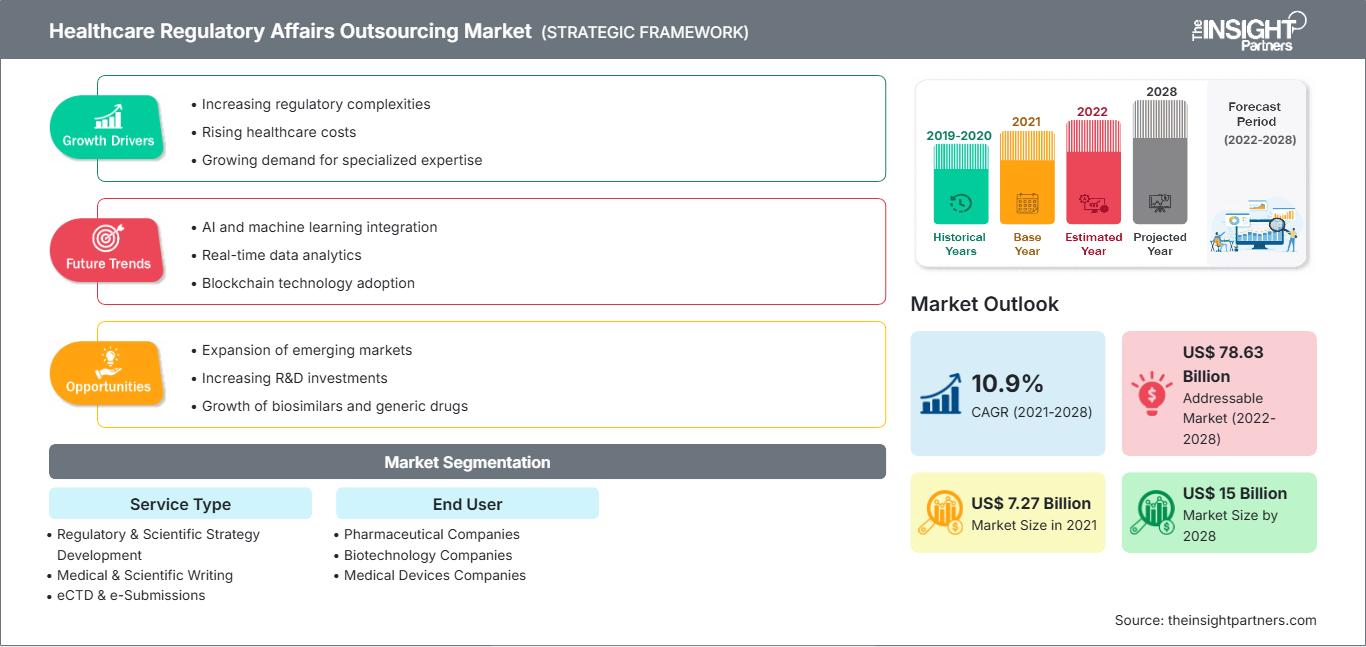
[Rapport de recherche] Le marché de l'externalisation des affaires réglementaires dans le secteur de la santé devrait atteindre 14 996,35 millions de dollars américains d'ici 2028, contre 7 274,73 millions de dollars américains en 2021 ; sa croissance devrait atteindre un TCAC de 10,9 % entre 2021 et 2028.
La pression réglementaire croissante sur les entreprises du secteur de la santé et la demande croissante d'approbation rapide des nouveaux produits freinent la croissance du marché de l'externalisation des affaires réglementaires dans le secteur de la santé. Cependant, le manque de professionnels qualifiés freine la croissance du marché. L'externalisation des affaires réglementaires est un service proposé aux industries pharmaceutiques, biotechnologiques et de fabrication de dispositifs médicaux. Les services d'externalisation des affaires réglementaires contribuent à obtenir des approbations réglementaires rapides. Les industries d'externalisation des affaires réglementaires aident à obtenir l'approbation de nouveaux produits, à préparer des protocoles pour la conduite d'un essai clinique, à publier des rapports, etc. Une augmentation de la demande pour divers services tels que la consultation réglementaire, la rédaction médicale et la publication de la documentation réglementaire, les demandes d'essais cliniques, ainsi que la consultation réglementaire et les représentations juridiques, les demandes de brevet, l'enregistrement de produits et les demandes d'essais cliniques a entraîné une augmentation de l'adoption des activités d'externalisation des affaires réglementaires des soins de santé.
Vous bénéficierez d’une personnalisation sur n’importe quel rapport - gratuitement - y compris des parties de ce rapport, ou une analyse au niveau du pays, un pack de données Excel, ainsi que de profiter d’offres exceptionnelles et de réductions pour les start-ups et les universités
Marché de l'externalisation des affaires réglementaires en matière de santé: Perspectives stratégiques
-
Obtenez les principales tendances clés du marché de ce rapport.Cet échantillon GRATUIT comprendra une analyse de données, allant des tendances du marché aux estimations et prévisions.
Analyses du marché
Pression réglementaire croissante sur les entreprises de santé Croissance du marché de l'externalisation des affaires réglementaires de santé
Les mises à niveau et les progrès continus des approches traditionnelles de développement de médicaments créent des défis importants dans le secteur de la santé. Les sociétés pharmaceutiques et le corps médical subissent une pression énorme pour réduire le coût des médicaments sur ordonnance, tandis que leurs coûts opérationnels explosent. La complexité des exigences réglementaires, la baisse des revenus due à l'expiration des brevets des médicaments phares et la pression des gouvernements et des assureurs maladie pour réduire les coûts des soins de santé ont posé des défis supplémentaires aux industries de la santé. Face à ces difficultés, les sociétés pharmaceutiques ont compris la nécessité de valoriser leurs ressources et l'expertise de sources externes spécialisées. De nombreux cabinets de conseil en réglementation haut de gamme proposent leur expertise tout au long du cycle de vie du produit. L'externalisation des affaires réglementaires peut permettre aux promoteurs d'acquérir de l'expérience, d'optimiser les coûts et d'améliorer la productivité. Les sociétés d'externalisation réglementaire sont mieux placées pour évaluer les exigences réglementaires, ce qui leur permet de sélectionner les meilleures solutions. Ils maîtrisent parfaitement la mise en œuvre, l'exploitation et la maintenance d'un système de publication réglementaire. La plupart des grandes entreprises pharmaceutiques et biotechnologiques recherchent des sociétés de conseil capables de proposer également des services d'accompagnement en matière de réglementation et de pharmacovigilance.
La complexité croissante des dépôts réglementaires souligne la demande d'expertise spécialisée en CRO. Il est essentiel de planifier des conseils et des stratégies réglementaires spécifiques à chaque produit, ainsi que des mesures de conformité réglementaire en matière de santé, dès les premières étapes du développement pour obtenir l'approbation réglementaire. L'absence de conformité dès les premières étapes du développement entraîne souvent des retards dans le processus d'approbation en raison de documents mal déposés, d'oublis de fabrication, d'études réglementaires omises et d'autres manquements aux exigences réglementaires. Les entreprises du secteur de la santé se concentrent désormais sur leurs compétences clés et externalisent les fonctions non essentielles afin d'améliorer leur productivité et leur efficacité opérationnelle. Elles externalisent généralement les fonctions réglementaires à des CRO opérant sur les marchés émergents, comme l'Asie-Pacifique et le Moyen-Orient et l'Afrique, ce qui leur permet également de réduire leurs coûts opérationnels et de se concentrer sur leurs fonctions clés, telles que la R&D et la commercialisation des produits existants. Ventes et distribution.
Informations basées sur le type de service
En fonction du type de service, le marché de l'externalisation des affaires réglementaires des soins de santé est segmenté en : élaboration de stratégies réglementaires et scientifiques, rédaction médicale et scientifique, eCTD et soumissions électroniques, services de gestion des données, services de gestion du cycle de vie, pharmacovigilance, services de fabrication et de contrôle de produits chimiques (CMC), étiquetage réglementaire et services d'illustration réglementaire. Le segment de la rédaction médicale et scientifique devrait détenir une part de marché plus importante en 2021, et le segment de la pharmacovigilance devrait en outre enregistrer un TCAC plus élevé au cours de la période de prévision.
Informations basées sur l'utilisateur final
En fonction de l'utilisateur final, le marché de l'externalisation des affaires réglementaires des soins de santé est segmenté en sociétés pharmaceutiques, sociétés de biotechnologie et sociétés de dispositifs médicaux. Le segment des sociétés pharmaceutiques représenterait une part de marché plus importante en 2021. Le marché du segment des sociétés pharmaceutiques devrait croître à un TCAC plus élevé de 2021 à 2028.
Les entreprises opérant sur le marché de l'externalisation des affaires réglementaires des soins de santé adoptent la stratégie d'innovation de produits pour répondre à l'évolution des demandes des clients dans le monde entier, ce qui leur permet également de maintenir leur nom de marque sur le marché mondial.
Marché de l'externalisation des affaires réglementaires des soins de santé
Les tendances et facteurs régionaux influençant le marché de l'externalisation des affaires réglementaires de santé tout au long de la période de prévision ont été analysés en détail par les analystes de The Insight Partners. Cette section aborde également les segments et la répartition géographique du marché de l'externalisation des affaires réglementaires de santé en Amérique du Nord, en Europe, en Asie-Pacifique, au Moyen-Orient et en Afrique, ainsi qu'en Amérique du Sud et en Amérique centrale.Portée du rapport sur le marché de l'externalisation des affaires réglementaires des soins de santé| Attribut de rapport | Détails |
|---|---|
| Taille du marché en 2021 | US$ 7.27 Billion |
| Taille du marché par 2028 | US$ 15 Billion |
| TCAC mondial (2021 - 2028) | 10.9% |
| Données historiques | 2019-2020 |
| Période de prévision | 2022-2028 |
| Segments couverts |
By Type de service
|
| Régions et pays couverts |
Amérique du Nord
|
| Leaders du marché et profils d'entreprises clés |
|
Densité des acteurs du marché de l'externalisation des affaires réglementaires de santé : comprendre son impact sur la dynamique des entreprises
Le marché de l'externalisation des affaires réglementaires dans le secteur de la santé connaît une croissance rapide, portée par une demande croissante des utilisateurs finaux, due à des facteurs tels que l'évolution des préférences des consommateurs, les avancées technologiques et une meilleure connaissance des avantages du produit. Face à cette demande croissante, les entreprises élargissent leur offre, innovent pour répondre aux besoins des consommateurs et capitalisent sur les nouvelles tendances, ce qui alimente la croissance du marché.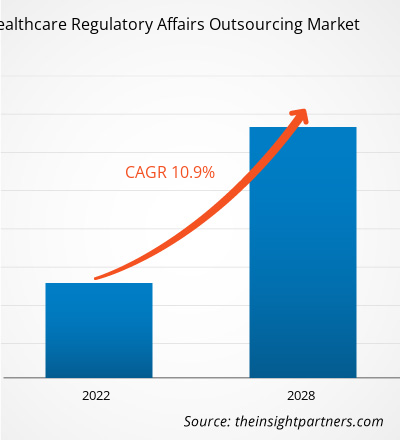
- Obtenez le Marché de l'externalisation des affaires réglementaires en matière de santé Aperçu des principaux acteurs clés
Marché de l'externalisation des affaires réglementaires des soins de santé - par type de service
- Développement de stratégies réglementaires et scientifiques
- Rédaction médicale et scientifique
- eCTD et soumissions électroniques
- Services de gestion des données
- Services de gestion du cycle de vie
- Pharmacovigilance
- Services de fabrication et de contrôle des produits chimiques (CMC)
- Étiquetage réglementaire
- Services d'illustration réglementaire
Marché de l'externalisation des affaires réglementaires des soins de santé - par utilisateur final
- Sociétés pharmaceutiques
- Sociétés de biotechnologie
- Sociétés de dispositifs médicaux
- Logiciels pour dispositifs médicaux (SaMD)
- Matériaux et composants pour dispositifs médicaux Biomatériaux
- Biomarqueurs de dispositifs médicaux et diagnostics in vitro (DIV)
- Dispositif médical à base de substances
- Dispositif médical de produit combiné (DDC)
Marché de l'externalisation des affaires réglementaires des soins de santé - par géographie
- Amérique du Nord
- États-Unis
- Canada
- Mexique
- Europe
- France
- Allemagne
- Italie
- Royaume-Uni
- Espagne
- Reste de l'Europe
- Asie-Pacifique (APAC)
- Chine
- Inde
- Corée du Sud
- Japon
- Australie
- Reste de l'Asie-Pacifique
- Moyen-Orient et Afrique (MOA)
- Afrique du Sud
- Arabie saoudite
- Émirats arabes unis
- Reste du Moyen-Orient et de l'Afrique
- Amérique du Sud et centrale (SCAM)
- Brésil
- Argentine
- Reste de l'Amérique du Sud et centrale
Profils d'entreprise
- KLIFO
- Groupe ProPharma
- Arriello Ireland Ltd.
- DRA CONSULTING OY
- Asphalion SL
- Parexel International Corporation
- IQVIA Inc.
- Pharmalex Gmbh
- Groupe ProductLife
- Voisin Consulting Life Sciences (VCLS)
- Azierta Contract Science Support Consulting
- Analyse historique (2 ans), année de base, prévision (7 ans) avec TCAC
- Analyse PEST et SWOT
- Taille du marché Valeur / Volume - Mondial, Régional, Pays
- Industrie et paysage concurrentiel
- Ensemble de données Excel
Rapports récents
Rapports connexes
Témoignages
Raison d'acheter
- Prise de décision éclairée
- Compréhension de la dynamique du marché
- Analyse concurrentielle
- Connaissances clients
- Prévisions de marché
- Atténuation des risques
- Planification stratégique
- Justification des investissements
- Identification des marchés émergents
- Amélioration des stratégies marketing
- Amélioration de l'efficacité opérationnelle
- Alignement sur les tendances réglementaires






















 Obtenez un échantillon gratuit pour - Marché de l'externalisation des affaires réglementaires en matière de santé
Obtenez un échantillon gratuit pour - Marché de l'externalisation des affaires réglementaires en matière de santé