In-Vitro Diagnostic Market Growth, Size & Forecast by 2034
In-Vitro Diagnostic Market Size and Forecast (2021 - 2034), Global and Regional Share, Trend, and Growth Opportunity Analysis Report Coverage: By Product & Service (Reagents & Kits, Instruments, and Software & Services), Technology (Immunoassay/ Immunochemistry, Clinical Chemistry, Molecular Diagnostics, Microbiology, Blood Glucose Self-Monitoring, Coagulation and Hemostasis, Hematology, Urinalysis, and Others), Application (Infectious Diseases, Diabetes, Oncology, Cardiology, Autoimmune Diseases, Nephrology, and Others), End User (Hospitals, Laboratories, Home Care, and Others), and Geography (North America, Europe, Asia Pacific, the Middle East & Africa, and South & Central America)
Historic Data: 2021-2024 | Base Year: 2025 | Forecast Period: 2026-2034- Status : Data Released
- Report Code : TIPBT00002657
- Category : Life Sciences
- No. of Pages : 150
- Available Report Formats :


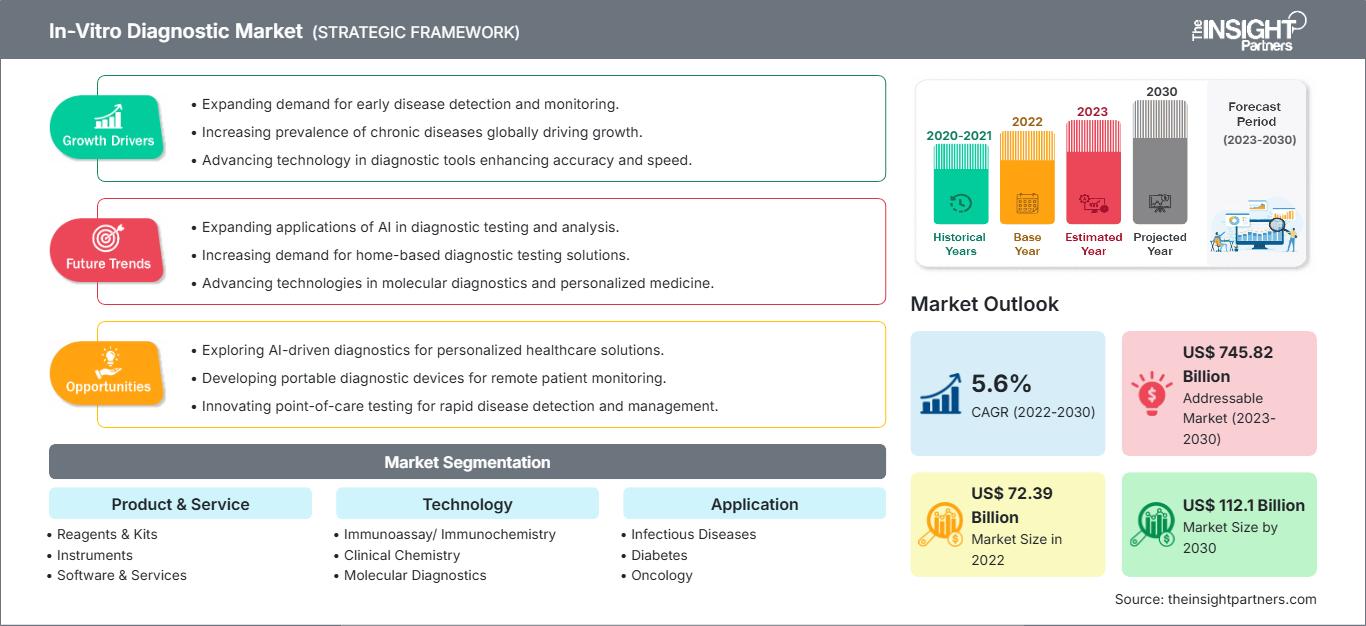
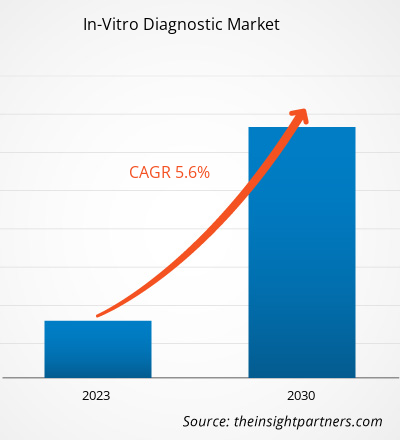
The global In-Vitro Diagnostic Market size is projected to reach US$ 139.95 billion by 2034 from US$ 88.71 billion in 2025. The market is anticipated to register a CAGR of 5.20% during the forecast period 2026–2034
Key market dynamics include an accelerating global shift toward precision medicine, the rapid integration of artificial intelligence in diagnostic workflows, and a rising clinical emphasis on early disease intervention. Additionally, the market is expected to benefit from the decentralization of healthcare through point-of-care (POC) testing, significant investments in genomic research, and the expanding application of molecular diagnostics in oncology and infectious disease management across both developed and emerging economies.
In-Vitro Diagnostic Market Analysis
The in-vitro diagnostic market analysis reveals a fundamental transition toward value-based healthcare, where diagnostic accuracy and speed are the primary metrics for clinical success. Strategic opportunities are increasingly concentrated in the liquid biopsy and companion diagnostics market, where IVD tools enable clinicians to tailor therapies to individual genetic profiles, particularly in oncology. Procurement trends indicate a move away from siloed instrument purchases toward integrated laboratory automation systems that combine high-throughput clinical chemistry with molecular capabilities. The analysis further suggests that competitive differentiation is now driven by connected diagnostics, platforms that offer seamless data integration with hospital information systems and remote monitoring capabilities. For manufacturers, the path to market leadership involves navigating complex global regulatory frameworks, such as the EU's IVDR, while simultaneously scaling cost-effective solutions for high-volume screening in developing regions.
In-Vitro Diagnostic Market Overview
In-vitro diagnostic (IVD) is evolving from a supplementary clinical service into the cornerstone of modern medical decision-making. Historically centered on routine blood and urine analysis in centralized laboratories, the sector has expanded into sophisticated domains including next-generation sequencing (NGS), digital pathology, and multiplexed immunoassays. The market landscape is defined by a diverse array of participants, ranging from multinational conglomerates providing end-to-end laboratory solutions to specialized biotech firms focused on niche biomarkers. As healthcare systems globally face the dual pressures of an aging population and the rising burden of chronic diseases like diabetes and cardiovascular disorders, IVD technologies are being leveraged to provide faster, more accurate results at the point of impact. The proliferation of specialized diagnostic franchises and the mainstreaming of at-home testing kits for wellness and chronic condition management further support this shift. For instance, the market in the US represents the most mature and influential segment of the global landscape, characterized by high healthcare expenditure and a robust culture of clinical innovation. The market is driven by a sophisticated reimbursement environment, the widespread adoption of automated laboratory systems, and a strong regulatory emphasis on diagnostic safety and efficacy.
Customize This Report To Suit Your Requirement
Get FREE CUSTOMIZATIONIn-Vitro Diagnostic Market: Strategic Insights
-
Get Top Key Market Trends of this report.This FREE sample will include data analysis, ranging from market trends to estimates and forecasts.
In-Vitro Diagnostic Market Drivers and Opportunities
Market Drivers:
- Rising Global Burden of Chronic and Infectious Diseases: The increasing prevalence of conditions such as cancer, diabetes, and cardiovascular diseases necessitates continuous monitoring and early detection. Additionally, the constant emergence of viral pathogens has cemented the need for robust, scalable infectious disease testing infrastructure.
- Technological Convergence with AI and Automation: The integration of machine learning algorithms into diagnostic software allows for the analysis of complex datasets with unprecedented speed. Laboratory automation reduces human error and enhances throughput, enabling facilities to manage rising test volumes efficiently.
- Shift Toward Personalized and Preventive Medicine: There is a growing clinical preference for diagnostic tests that can predict drug response or disease susceptibility. Companion diagnostics and genetic screening are becoming standard protocols, driving the demand for advanced molecular and tissue-based IVD solutions.
Market Opportunities:
- Expansion of Point-of-Care and Decentralized Testing: Significant growth potential exists in moving diagnostics closer to the patient, including retail clinics, physician offices, and home settings. Portable, user-friendly devices that deliver lab-quality results in minutes offer a major entry point for innovators.
- Advancements in Liquid Biopsy and Non-Invasive Screening: Developing IVD tools that can detect cancer or fetal abnormalities through simple blood draws presents a high-margin opportunity. These technologies are increasingly favored over invasive procedures, appealing to both patients and providers.
- Digitization and Cloud-Based Diagnostic Platforms: Strategic opportunities lie in the development of Diagnostic-as-a-Service models. By utilizing cloud-connected instruments, providers can offer real-time data analytics and remote interpretation services, particularly in regions with limited specialist availability.
In-Vitro Diagnostic Market Report Segmentation Analysis
The In-Vitro Diagnostic Market share is analyzed across various segments to provide a clearer understanding of its structure, growth potential, and emerging trends. Below is the standard segmentation approach used in most industry reports:
By Product & Service:
- Reagents & Kits: This segment represents the largest portion of the market, driven by the recurring demand for consumables required to perform a vast array of clinical tests across all diagnostic modalities.
- Instruments: Comprises the hardware and machinery used in laboratories and point-of-care settings, focusing on increasing automation, miniaturization, and high-throughput capabilities.
- Software & Services: A high-growth segment involving laboratory information systems, AI-driven analytical tools, and technical maintenance services essential for modern digital pathology and data management.
By Technology:
- Immunoassay/Immunochemistry: A foundational technology used for detecting proteins and antibodies, critical for infectious disease testing and oncology.
- Molecular Diagnostics: The fastest-growing tech segment, utilizing PCR and NGS to analyze genetic material for precision medicine and pathogen identification.
- Clinical Chemistry: Focused on routine biochemical analysis of body fluids, serving as the workhorse of hospital laboratories.
- Others: Includes microbiology, blood glucose self-monitoring, coagulation/hemostasis, hematology, and urinalysis.
By Application:
- Infectious Diseases: The leading application segment due to global screening programs for HIV, hepatitis, and respiratory infections.
- Oncology: Rapidly expanding through the use of biomarkers and genetic testing for cancer staging and therapy selection.
- Others: Encompasses diabetes, cardiology, autoimmune diseases, and nephrology.
By End User:
- Hospitals: The primary setting for acute diagnostic testing, requiring integrated and rapid turnaround solutions.
- Laboratories: Including standalone and reference labs that handle high-volume routine and specialized diagnostic processing.
- Others: Covers home care settings and point-of-care centers, reflecting the trend toward decentralized healthcare.
By Geography:
- North America
- Europe
- Asia Pacific
- South & Central America
- Middle East & Africa
In-Vitro Diagnostic Market Report Scope
| Report Attribute | Details |
|---|---|
| Market size in 2025 | US$ 88.71 Billion |
| Market Size by 2034 | US$ 139.95 Billion |
| Global CAGR (2026 - 2034) | 5.2% |
| Historical Data | 2021-2024 |
| Forecast period | 2026-2034 |
| Segments Covered |
By Product & Service
|
| Regions and Countries Covered |
North America
|
| Market leaders and key company profiles |
|
In-Vitro Diagnostic Market Players Density: Understanding Its Impact on Business Dynamics
The In-Vitro Diagnostic Market is growing rapidly, driven by increasing end-user demand due to factors such as evolving consumer preferences, technological advancements, and greater awareness of the product's benefits. As demand rises, businesses are expanding their offerings, innovating to meet consumer needs, and capitalizing on emerging trends, which further fuels market growth.
In-Vitro Diagnostic Market Share Analysis by Geography
Asia-Pacific is expected to grow fastest in the coming years. Emerging markets in South & Central America, the Middle East, and Africa also have many untapped opportunities for diagnostic manufacturers and clinical laboratory service providers to expand.
The in-vitro diagnostic market is undergoing a significant transformation, moving from a centralized laboratory model to a more distributed, patient-centric ecosystem. Growth is driven by the modernization of healthcare infrastructure, a surge in demand for rapid testing, and the expansion of the precision oncology industry. Below is a summary of market share and trends by region:
North America
- Market Share: Holds the largest global share, anchored by the United States' leadership in genomic research and diagnostic technology.
- Key Drivers:
- High adoption rates of premium molecular diagnostic platforms and NGS technologies.
- Favorable reimbursement policies for advanced screening and companion diagnostics.
- Presence of leading industry players and extensive R&D investments in AI-integrated pathology.
- Trends: A strong shift toward home-based testing and retail health diagnostic services, alongside the integration of digital health platforms for chronic disease management.
Europe
- Market Share: A major regional market characterized by high regulatory standards and a well-established network of public and private clinical laboratories.
- Key Drivers:
- Implementation of the In Vitro Diagnostic Regulation (IVDR), driving a focus on clinical evidence and quality.
- Strong government support for early cancer screening and infectious disease surveillance.
- An aging population requires frequent diagnostic monitoring for age-related conditions.
- Trends: Strategic focus on laboratory consolidation to improve cost-efficiency and the rising use of digital pathology for cross-border specialist consultations.
Asia-Pacific
- Market Share: The fastest-growing region, with China and India acting as primary engines for market expansion due to massive population bases.
- Key Drivers:
- Rapid expansion of healthcare infrastructure and private hospital networks.
- Increasing disposable income is leading to higher demand for premium health check-ups and wellness screening.
- Government initiatives focused on local manufacturing and medical device self-sufficiency.
- Trends: Heavy investment in localized production of diagnostic reagents and the rapid adoption of smartphone-connected point-of-care devices in rural and semi-urban areas.
South and Central America
- Market Share: An emerging market with significant growth potential in countries like Brazil and Argentina.
- Key Drivers:
- Growing awareness of preventive healthcare and the importance of early diagnosis in reducing long-term treatment costs.
- Modernization of public health laboratories to manage endemic infectious diseases.
- Rising middle-class access to private health insurance, boosting demand for clinical laboratory services.
- Trends: Expansion of international diagnostic laboratory chains into the region and the introduction of low-cost, high-reliability rapid test kits for community health centers.
Middle East and Africa
- Market Share: A developing market with a focus on upgrading healthcare facilities and addressing the high burden of infectious diseases.
- Key Drivers:
- Strategic government Vision plans (e.g., in Saudi Arabia and the UAE) aimed at transforming the healthcare sector.
- High demand for advanced diagnostics for genetic disorders and metabolic diseases.
- Investment in Smart Hospitals that utilize automated and connected diagnostic systems.
- Trends: Implementation of national screening programs for non-communicable diseases and an increasing reliance on international partnerships for specialized molecular testing services.
High Market Density and Competition
Competition is intensifying due to the presence of established leaders such as Roche Diagnostics, Abbott Laboratories, and Danaher Corporation. Global giants like Siemens Healthineers and Thermo Fisher Scientific, alongside specialized innovators such as Qiagen and Sysmex, also contribute to a diverse and rapidly expanding market landscape.
This competitive environment pushes vendors to differentiate through:
- System Integration and Automation: Positioning IVD platforms as comprehensive total lab automation solutions that minimize manual intervention and maximize throughput for busy hospital environments.
- Expanding Assay Menus: Companies are racing to offer the broadest range of tests on a single platform, from routine chemistry to specialized genetic markers, providing a one-stop shop for clinical providers.
- Strategic Digitalization: Utilizing proprietary software and AI algorithms to provide clinical decision support, helping physicians interpret complex diagnostic data more accurately.
- Global Supply Chain Resilience: Managing the end-to-end production of reagents and instruments to ensure consistent availability, meeting ethical and regulatory standards across diverse international markets.
Opportunities and Strategic Moves
- Partner with Tech Giants: Collaborating with software and AI companies to enhance diagnostic accuracy and develop cloud-based data management solutions for global laboratory networks.
- Acquisition of Niche Biotech Firms: Acquiring smaller companies with proprietary biomarker technology or innovative point-of-care platforms to quickly expand product portfolios into high-growth segments like oncology or at-home testing.
Major Companies operating in the In-Vitro Diagnostic Market are:
- Abbott Laboratories
- F. Hoffmann-La Roche Ltd
- Danaher Corp
- Siemens AG
- Sysmex Corp
- Thermo Fisher Scientific Inc
- Becton Dickinson and Co
- bioMerieux SA
- Bio-Rad Laboratories Inc
- Qiagen NV
Disclaimer: The companies listed above are not ranked in any particular order.
In-Vitro Diagnostic Market News and Recent Developments
- In June 2025, Revvity, Inc. announced the launch of three Mimix™ reference standards for in In-Vitro Diagnostic use, specifically designed for the monitoring of next-generation sequencing (NGS) and droplet digital PCR (ddPCR) assays. These standards were developed to detect somatic mutations in genomic DNA from human samples, ensuring high-fidelity performance within a clinical In-Vitro Diagnostic framework. By undergoing rigorous design controls to meet U.S. FDA regulatory requirements, these cell line-derived standards enabled laboratories to seamlessly integrate them into existing workflows to monitor test performance, manage assay variation, and identify potential random or systemic errors.
- In April 2025, Roche announced that it had received the CE Mark for its new chest pain triage algorithm, a significant advancement in the in In-Vitro Diagnostic landscape. Developed in collaboration with Universitätsklinikum Heidelberg, the algorithm was designed to more quickly and accurately detect Acute Coronary Syndrome (ACS) in patients presenting with chest pain, which remained one of the most common reasons for emergency department visits. By integrating this software with established In-Vitro Diagnostic cardiac biomarkers, the company aimed to streamline clinical decision-making and improve patient outcomes in high-pressure emergency settings.
In-Vitro Diagnostic Market Report Coverage and Deliverables
The In-Vitro Diagnostic Market Size and Forecast (2021–2034) report provides a detailed analysis of the market covering below areas:
- In-Vitro Diagnostic Market size and forecast at global, regional, and country levels for all the key market segments covered under the scope
- In-Vitro Diagnostic Market trends, as well as market dynamics such as drivers, restraints, and key opportunities
- Detailed PEST and SWOT analysis
- In-Vitro Diagnostic Market analysis covering key market trends, global and regional framework, major players, regulations, and recent market developments
- Industry landscape and competition analysis covering market concentration, heat map analysis, prominent players, and recent developments in the In-Vitro Diagnostic Market.
- Detailed company profiles
Frequently Asked Questions
Mrinal is a seasoned research analyst with over 8 years of experience in Life Sciences Market Intelligence and Consulting. With a strategic mindset and unwavering commitment to excellence, she has built deep expertise in pharmaceutical forecasting, market opportunity assessment, and developing industry benchmarks. Her work is anchored in delivering actionable insights that empower clients to make informed strategic decisions.
Mrinal’s core strength lies in translating complex quantitative datasets into meaningful business intelligence. Her analytical acumen is instrumental in shaping go-to-market (GTM) strategies and uncovering growth opportunities across the pharmaceutical and medical device sectors. As a trusted consultant, she consistently focuses on streamlining workflow processes and establishing best practices, thereby driving innovation and operational efficiency for her clients.
- Historical Analysis (2 Years), Base Year, Forecast (7 Years) with CAGR
- PEST and SWOT Analysis
- Market Size Value / Volume - Global, Regional, Country
- Industry and Competitive Landscape
- Excel Dataset
Recent Reports
Testimonials
The Insight Partners' SCADA System Market report is comprehensive, with valuable insights on current trends and future forecasts. The team was highly professional, responsive, and supportive throughout. We are very satisfied and highly recommend their services.
RAN KEDEM Partner, Reali Technologies LTDsI requested a report on a very specific software market and the team produced the report in a few days. The information was very relevant and well presented. I then requested some changes and additions to the report. The team was again very responsive and I got the final report in less than a week.
JEAN-HERVE JENN Chairman, Future AnalyticaWe worked with The Insight Partners for an important market study and forecast. They gave us clear insights into opportunities and risks, which helped shape our plans. Their research was easy to use and based on solid data. It helped us make smart, confident decisions. We highly recommend them.
PIYUSH NAGPAL Sr. Vice President, High Beam GlobalThe Insight Partners delivered insightful, well-structured market research with strong domain expertise. Their team was professional and responsive throughout. The user-friendly website made accessing industry reports seamless. We highly recommend them for reliable, high-quality research services
YUKIHIKO ADACHI CEO, Deep Blue, LLC.This is the first time I have purchased a market report from The Insight Partners.While I was unsure at first, I visited their web site and felt more comfortable to take the risk and purchase a market report.I am completely satisfied with the quality of the report and customer service. I had several questions and comments with the initial report, but after a couple of dialogs over email with their analyst I believe I have a report that I can use as input to our strategic planning process.Thank you so much for taking the extra time and making this a positive experience.I will definitely recommend your service to others and you will be my first call when we need further market data.
JOHN SUZUKI President and Chief Executive Officer, Board Director, BK TechnologiesI wish to appreciate your support and the professionalism you displayed in the course of attending to my request for information regarding to infectious disease IVD market in Nigeria. I appreciate your patience, your guidance, and the fact that you were willing to offer a discount, which eventually made it possible for us to close a deal. I look forward to engaging The Insight Partners in the future, all thanks to the impression you have created in me as a result of this first encounter.
DR CHIJIOKE ONYIA MANAGING DIRECTOR, PineCrest Healthcare Ltd.Reason to Buy
- Informed Decision-Making
- Understanding Market Dynamics
- Competitive Analysis
- Identifying Emerging Markets
- Customer Insights
- Market Forecasts
- Risk Mitigation
- Boosting Operational Efficiency
- Strategic Planning
- Investment Justification
- Tracking Industry Innovations
- Aligning with Regulatory Trends





 Get Free Sample For
Get Free Sample For