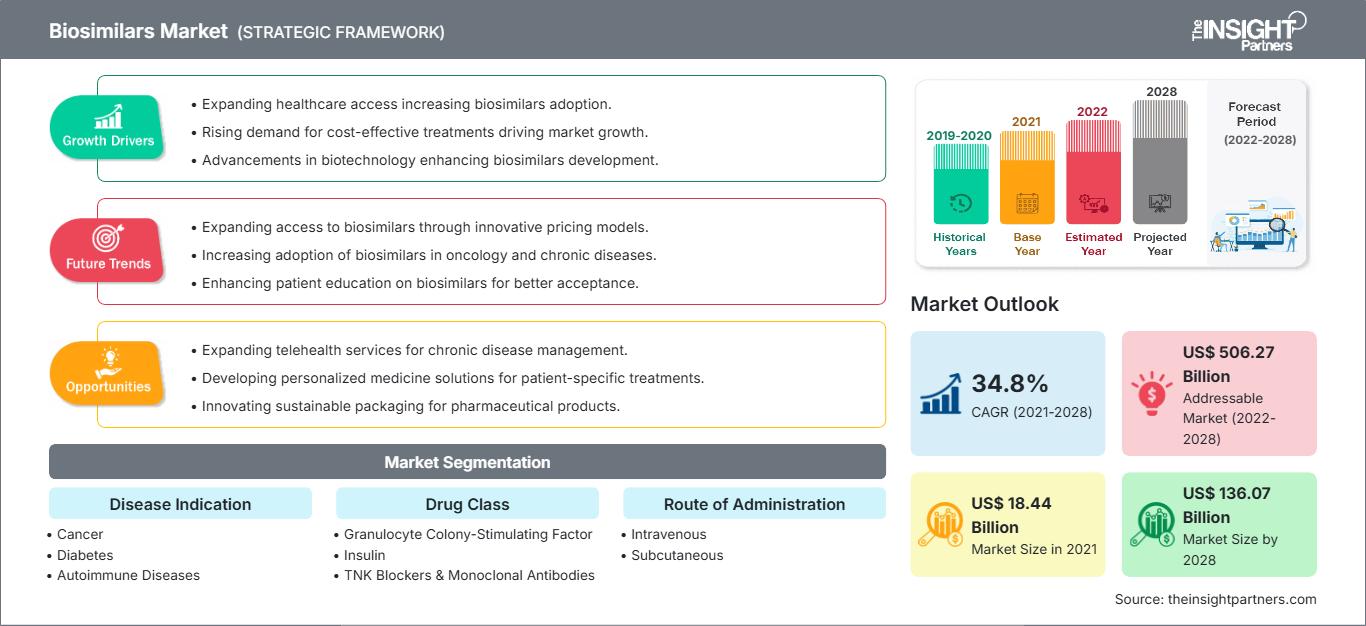
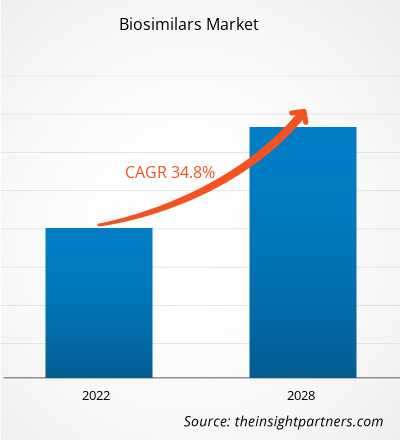
[Rapport de recherche] La taille du marché des biosimilaires devrait passer de 18 435,89 millions de dollars américains en 2021 à 1 36 069,53 millions de dollars américains en 2028 ; on estime qu'il enregistrera un TCAC de 34,8 % de 2022 à 2028.
Point de vue de l'analyste
Les principaux facteurs à l'origine de la croissance du marché des biosimilaires sont l'incidence croissante des maladies chroniques, telles que les cancers. Le fardeau croissant du cancer et l'augmentation des décès qui en découlent créent un besoin de traitements abordables et stimulent ainsi la croissance du marché des biosimilaires. Les principaux acteurs du marché ont également anticipé la croissance du marché au cours de la période de prévision grâce à diverses activités stratégiques, telles que les lancements de produits, les fusions et les acquisitions. L'augmentation de la prévalence des maladies auto-immunes telles que la spondylarthrite ankylosante et la polyarthrite rhumatoïde stimule la croissance de la taille du marché des biosimilaires. Français Par exemple, selon un article publié dans le « Scandinavian Journal of Rheumatology » en 2020, intitulé « Prévalence de la spondylarthrite ankylosante en Espagne », environ 7,3 % de la population a présenté un dépistage positif de la spondylarthrite ankylosante. Les biosimilaires, tels que l'infliximab-axxq (Avsola), l'infliximab-qbtx (Ixifi), l'infliximab-dyyb (Inflectra) et l'infliximab-abda (Renflexis) sont utilisés pour le traitement de la douleur chronique dans l'arthrite.
Aperçu du marché
Un biosimilaire est un médicament biologique très similaire à un autre médicament biologique déjà approuvé (le « médicament de référence »). Les biosimilaires sont approuvés selon les mêmes normes de qualité pharmaceutique, de sécurité et d'efficacité que tous les médicaments biologiques. Ils constituent des options thérapeutiques sûres et efficaces pour de nombreuses maladies, telles que les maladies chroniques de la peau et de l'intestin (comme le psoriasis, le syndrome du côlon irritable, la maladie de Crohn et la colite), l'arthrite, les maladies rénales et le cancer. Ils facilitent l'accès à des médicaments vitaux à des coûts potentiellement plus bas. Le principal moteur du marché des biosimilaires est l'augmentation de l'incidence des maladies chroniques.
Vous bénéficierez d’une personnalisation sur n’importe quel rapport - gratuitement - y compris des parties de ce rapport, ou une analyse au niveau du pays, un pack de données Excel, ainsi que de profiter d’offres exceptionnelles et de réductions pour les start-ups et les universités
Marché des biosimilaires: Perspectives stratégiques
-
Obtenez les principales tendances clés du marché de ce rapport.Cet échantillon GRATUIT comprendra une analyse de données, allant des tendances du marché aux estimations et prévisions.
Moteur du marché
L'augmentation des approbations de biosimilaires va stimuler la croissance du marché mondial des biosimilaires
La Food and Drug Administration (FDA) approuve les produits biosimilaires et fournit les conseils scientifiques et réglementaires nécessaires pour mettre sur le marché des biosimilaires sûrs et efficaces. L'approbation des produits biosimilaires peut améliorer les soins aux patients en augmentant le nombre d'options médicamenteuses à des coûts potentiellement inférieurs.
Quelques approbations récentes de produits biosimilaires sont mentionnées dans le tableau suivant.
Nom du biosimilaire |
Date d'approbation |
Produit de référence |
Alymsys (bevacizumab-maly) |
Avril 2022 |
Avastin (bevacizumab) |
Cimerli (ranibizumab-eqrn) |
Août 2022 |
Lucentis (ranibizumab) |
Fylnetra (pegfilgrastim-pbbk) |
Mai 2022 |
Neulasta (pegfilgrastim) |
Stimufend (pegfilgrastim-fpgk) |
Septembre 2022 |
Neulasta(pegfilgrastim) |
Vegzelma (bevacizumab-adcd) |
Septembre 2022 |
Avastin (bevacizumab) |
Idacio (adalimumab-aacf) |
Décembre 2022 |
Humira (adalimumab) |
Byooviz (ranibizumab-nuna) |
Septembre 2021 |
Lucentis (ranibizumab) |
Rezvoglar (insuline glargine-aglr) |
Décembre 2021 |
Lantus (insuline glargine) |
Semglee (insuline glargine-yfgn) |
Juillet 2021 |
Lantus (insuline glargine) |
Yusimry (adalimumab-aqvh) |
Décembre 2021 |
Humira (adalimumab) |
Hulio (adalimumab-fkjp) |
Juillet 2020 |
Humira (adalimumab) |
Riabni (rituximab-arrx) |
Décembre 2020 |
Rituxan (rituximab) |
Nyvepria (pegfilgrastim-apgf) |
Juin 2020 |
Neulasta (pegfilgrastim) |
Ainsi, l'augmentation des approbations de biosimilaires propulse la croissance du marché des biosimilaires.
Analyse segmentaire
En fonction de l'indication de la maladie, le marché des biosimilaires est segmenté en cancer, diabète, maladies auto-immunes et autres indications de maladies. Le segment du cancer détenait la plus grande part de marché en 2021 et les maladies auto-immunes devraient enregistrer le TCAC le plus élevé de 36,1 % au cours de la période de prévision (2022-2028). En fonction de la classe de médicaments, le marché des biosimilaires est segmenté comme facteurs de stimulation des colonies de granulocytes, hormone de croissance humaine, insuline, anti-TNF et anticorps monoclonaux, et autres (ostéoporose, etc.). Le segment de la classe de médicaments des facteurs de stimulation des colonies de granulocytes détenait la plus grande part du marché en 2021. De plus, le segment des autres classes de médicaments devrait croître au TCAC le plus élevé au cours de la période de prévision. En fonction de l'application, le marché mondial des biosimilaires est divisé en applications intraveineuses, sous-cutanées et autres. Le segment intraveineux détenait la plus grande part du marché en 2021 et devrait croître au TCAC le plus élevé au cours de la période de prévision. Français Le marché des biosimilaires, par utilisateur final, est segmenté en hôpitaux, cliniques spécialisées, soins à domicile et autres utilisateurs finaux. Le segment des hôpitaux détenait la plus grande part du marché en 2021 et le segment des soins à domicile devrait enregistrer le TCAC le plus élevé du marché, soit 36,6 %, au cours de la période de prévision (2022-2028).
Analyse régionale
Le marché nord-américain des biosimilaires était évalué à 5 479,84 millions de dollars américains en 2021 et devrait atteindre 47 746,80 millions de dollars américains d'ici 2028 ; il devrait croître à un TCAC de 37,3 % au cours de la période de prévision. Le marché nord-américain des biosimilaires est segmenté entre les États-Unis, le Canada et le Mexique. En 2019, les États-Unis détenaient la plus grande part du marché nord-américain des biosimilaires. L'incidence du diabète et de l'infertilité a augmenté, et le développement de nouveaux produits sur ce marché a progressé. Selon le Comité de coordination des maladies auto-immunes des NIH, plus de 24 millions d'Américains souffraient de maladies auto-immunes en 2019. Huit millions de personnes présentent des auto-anticorps, des molécules sanguines qui indiquent le risque de développer une maladie auto-immune. Pour des raisons inconnues, les maladies auto-immunes touchent de plus en plus de personnes. Selon la branche de recherche clinique de l'Institut national des sciences de la santé environnementale (NIEHS), on observe une augmentation significative de la prévalence des anticorps antinucléaires (ANA), le biomarqueur le plus courant de l'auto-immunité aux États-Unis, en 2020. Cette étude est la première à évaluer l'évolution des ANA dans un échantillon représentatif de la population américaine au fil du temps. Elle comprend des hommes, des personnes blanches non hispaniques, des adultes de plus de 50 ans et des adolescents. Aux États-Unis, les biosimilaires sont utilisés pour traiter les patients atteints de cancers, de maladies rénales, de diabète et d'autres maladies auto-immunes telles que la polyarthrite rhumatoïde et la maladie de Crohn. Selon Cardinal Health, 33 biosimilaires ont été approuvés par la FDA aux États-Unis et 21 sont disponibles dans le commerce. Sur les 21 biosimilaires sur le marché, 17 sont utilisés pour des traitements associés aux cancers, trois pour traiter les maladies auto-immunes et un pour traiter le diabète.
Les produits biologiques sont les médicaments les plus chers aux États-Unis, avec des coûts s'élevant à des dizaines de milliers de dollars par an et par patient. Le prix des biosimilaires devrait être inférieur de 15 à 30 % à celui de leur produit de référence. Rien qu'en 2020, les biosimilaires ont permis d'économiser 7,9 milliards de dollars, et ces économies devraient augmenter considérablement au cours des prochaines années avec l'arrivée de nouveaux biosimilaires sur le marché. Selon Cardinal Health, les biosimilaires devraient réduire les dépenses en médicaments aux États-Unis de 133 milliards de dollars d'ici 2025. Ainsi, aux États-Unis, les biosimilaires présentent un potentiel immense : ils permettent de réduire les coûts des médicaments biologiques, de rendre les soins plus accessibles aux patients et de créer de nouvelles innovations et avancées scientifiques, stimulant ainsi la croissance du marché des biosimilaires dans cette région.
Analyse des principaux acteurs
L'analyse du marché des biosimilaires porte sur des acteurs tels qu'Amgen Inc, Celltrion Inc, Sanofi SA, Biocon Ltd, Pfizer Inc, Samsung Bioepis Co Ltd, Coherus BioSciences Inc, Eli Lilly and Co, Sandoz AG, Teva Pharmaceutical Industries Ltd et Dr. Reddy's Laboratories Ltd. Parmi les acteurs du marché des biosimilaires, Pfizer Inc. et Novartis, Inc. sont les deux principaux acteurs en raison de la diversification de leur portefeuille de produits.
Aperçu régional du marché des biosimilaires
Les tendances régionales et les facteurs influençant le marché des biosimilaires tout au long de la période de prévision ont été analysés en détail par les analystes de The Insight Partners. Cette section aborde également les segments et la répartition géographique du marché des biosimilaires en Amérique du Nord, en Europe, en Asie-Pacifique, au Moyen-Orient et en Afrique, ainsi qu'en Amérique du Sud et en Amérique centrale.
Portée du rapport sur le marché des biosimilaires
| Attribut de rapport | Détails |
|---|---|
| Taille du marché en 2021 | US$ 18.44 Billion |
| Taille du marché par 2028 | US$ 136.07 Billion |
| TCAC mondial (2021 - 2028) | 34.8% |
| Données historiques | 2019-2020 |
| Période de prévision | 2022-2028 |
| Segments couverts |
By Indication de la maladie
|
| Régions et pays couverts |
Amérique du Nord
|
| Leaders du marché et profils d'entreprises clés |
|
Densité des acteurs du marché des biosimilaires : comprendre son impact sur la dynamique commerciale
Le marché des biosimilaires connaît une croissance rapide, porté par une demande croissante des utilisateurs finaux, due à des facteurs tels que l'évolution des préférences des consommateurs, les avancées technologiques et une meilleure connaissance des avantages du produit. Face à cette demande croissante, les entreprises élargissent leur offre, innovent pour répondre aux besoins des consommateurs et capitalisent sur les nouvelles tendances, ce qui alimente la croissance du marché.
- Obtenez le Marché des biosimilaires Aperçu des principaux acteurs clés
- Analyse historique (2 ans), année de base, prévision (7 ans) avec TCAC
- Analyse PEST et SWOT
- Taille du marché Valeur / Volume - Mondial, Régional, Pays
- Industrie et paysage concurrentiel
- Ensemble de données Excel
Rapports récents
Rapports connexes
Témoignages
Raison d'acheter
- Prise de décision éclairée
- Compréhension de la dynamique du marché
- Analyse concurrentielle
- Connaissances clients
- Prévisions de marché
- Atténuation des risques
- Planification stratégique
- Justification des investissements
- Identification des marchés émergents
- Amélioration des stratégies marketing
- Amélioration de l'efficacité opérationnelle
- Alignement sur les tendances réglementaires






















 Obtenez un échantillon gratuit pour - Marché des biosimilaires
Obtenez un échantillon gratuit pour - Marché des biosimilaires